Research Articles
Validating Protein Molecular Weight Markers: A Complete Guide for Accurate Unknown Sample Analysis
This article provides researchers, scientists, and drug development professionals with a comprehensive guide to validating protein molecular weight markers against unknown samples.
SDS-PAGE vs. IEF-IPG: A Comprehensive Comparison for Advanced Proteomic Profiling
This article provides a detailed analytical comparison of SDS-PAGE and IEF-IPG techniques for proteomic profiling, addressing the critical needs of researchers and drug development professionals.
Gradient Gels vs. Fixed % Gels: A Strategic Guide for Optimizing Protein Separation in Biomedical Research
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on selecting and optimizing SDS-PAGE gels for protein analysis.
Sharp Bands, Reliable Results: Optimizing Buffer Composition and Freshness for Superior Electrophoresis
This article provides a comprehensive guide for researchers and drug development professionals on achieving sharp, well-defined bands in electrophoresis by optimizing buffer freshness and composition.
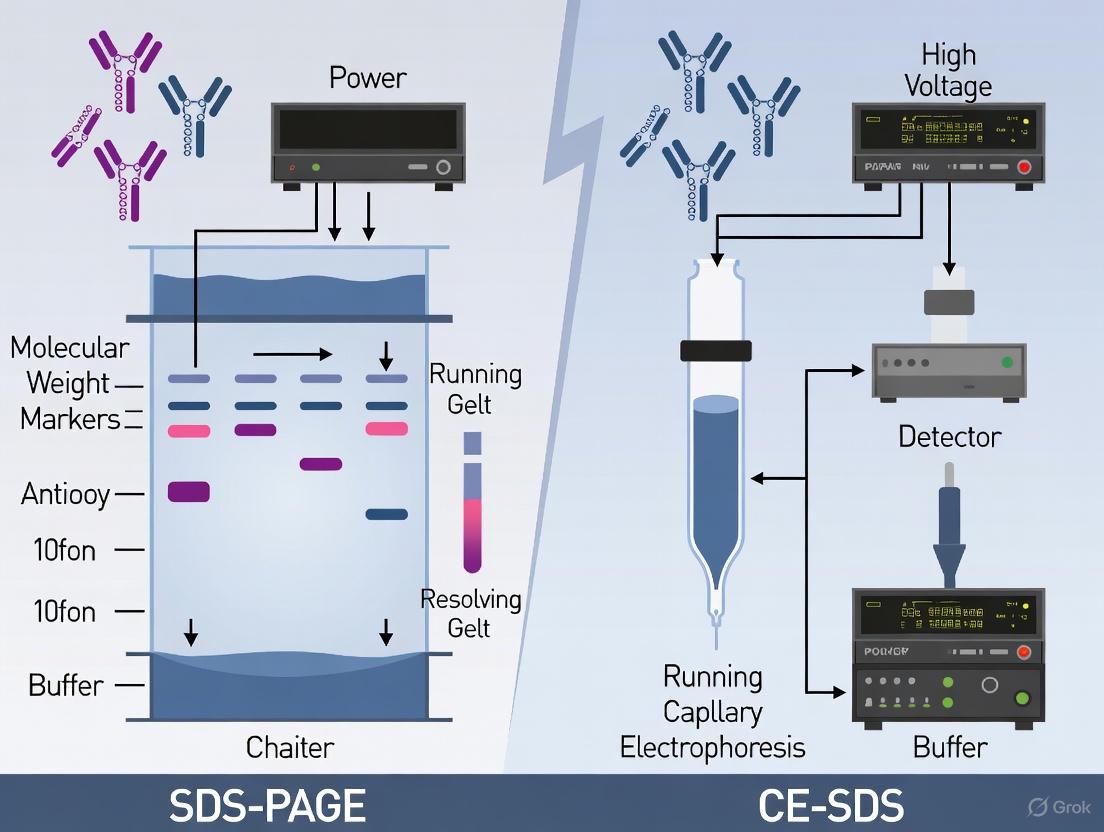
CE-SDS vs. SDS-PAGE: A Modern Guide to Antibody Purity Analysis for Biopharmaceutical Development
This article provides a comprehensive comparison of Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Capillary Electrophoresis-Sodium Dodecyl Sulfate (CE-SDS) for analyzing the purity of therapeutic monoclonal antibodies.
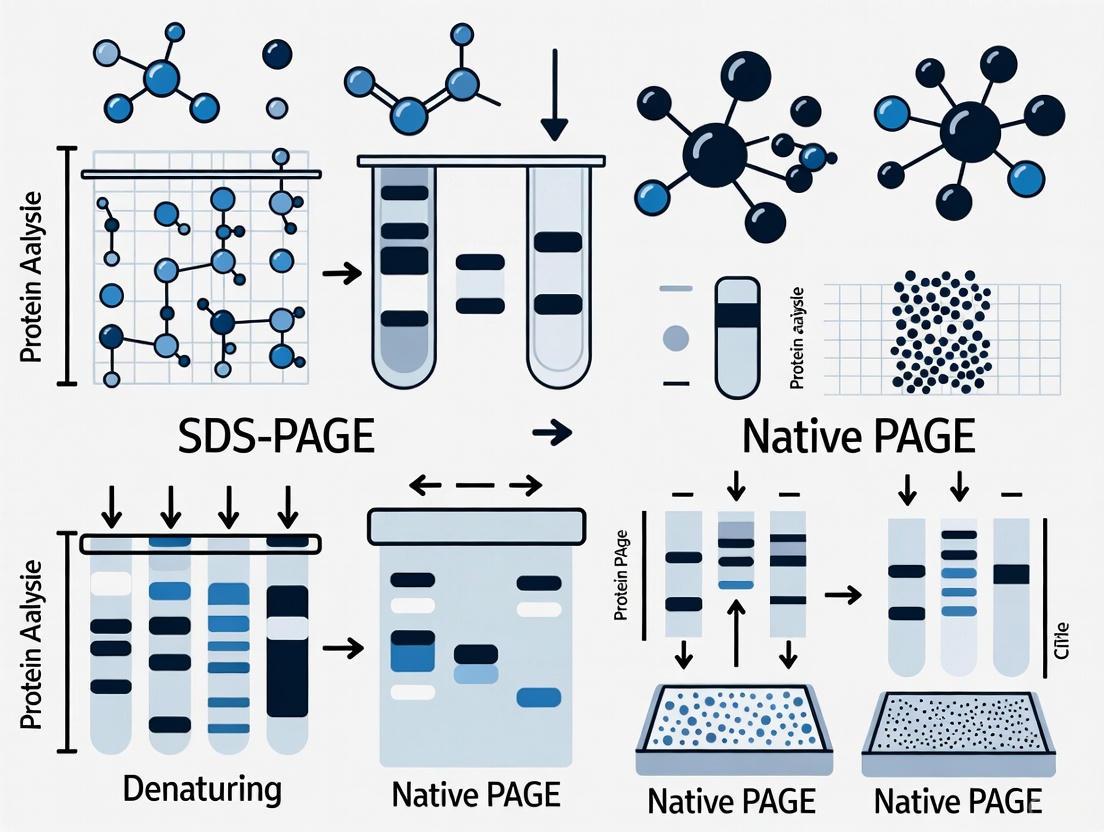
SDS-PAGE vs Native PAGE: A Comprehensive Guide to Choosing the Right Protein Analysis Technique
This article provides a definitive comparison of SDS-PAGE and Native PAGE, two foundational electrophoresis techniques in biochemistry and molecular biology.
Optimizing Sample Loading to Prevent Artifacts: A Strategic Guide for Robust Drug Development Data
This article provides a comprehensive guide for researchers and drug development professionals on preventing data-distorting overloading artifacts through strategic sample loading optimization.
Advanced Troubleshooting for Gel Polymerization: Solving Incomplete Setting in Biomedical Research
This article provides a comprehensive guide for researchers and drug development professionals facing gel polymerization challenges.
Solving Protein Aggregation in Electrophoresis: A Complete Guide from Prevention to Analysis
This article provides a comprehensive guide for researchers and drug development professionals tackling the pervasive challenge of protein aggregation during electrophoresis.
Defeating the Edge Effect: A Researcher's Guide to Preventing Distortion in Peripheral Gel Lanes
This article provides a comprehensive guide for researchers and drug development professionals on addressing the edge effect, a common phenomenon in gel electrophoresis where peripheral lanes exhibit distorted bands.