SDS-PAGE vs Native PAGE: A Comprehensive Guide to Choosing the Right Protein Analysis Technique
This article provides a definitive comparison of SDS-PAGE and Native PAGE, two foundational electrophoresis techniques in biochemistry and molecular biology.
SDS-PAGE vs Native PAGE: A Comprehensive Guide to Choosing the Right Protein Analysis Technique
Abstract
This article provides a definitive comparison of SDS-PAGE and Native PAGE, two foundational electrophoresis techniques in biochemistry and molecular biology. Tailored for researchers, scientists, and drug development professionals, it explores the core principles, separation mechanisms, and distinct applications of each method. The content delivers practical, actionable guidance on experimental design, protocol optimization, and advanced troubleshooting to ensure reliable results. By synthesizing methodological insights with comparative analysis, this guide empowers scientists to select the optimal technique for their specific research goals, whether for determining molecular weight, studying protein complexes, or analyzing functional activity.
Core Principles of Protein Electrophoresis: Understanding SDS-PAGE and Native PAGE
Electrophoresis is a foundational laboratory technique used to separate macromolecules—such as DNA, RNA, and proteins—based on their size, electrical charge, and other properties [1]. The principle relies on the motion of dispersed particles or dissolved charged molecules, known as ions, relative to a fluid under the influence of a spatially uniform electric field [2]. In practice, an electric current is applied to move the molecules through a porous gel or other matrix. The pores in this matrix act like a molecular sieve, allowing smaller molecules to move faster and migrate farther than larger molecules [1]. This technique is an indispensable tool in molecular biology and biochemistry for analyzing complex mixtures of biological molecules.
The core principle can be summarized by the fact that any charged ion or molecule will migrate when placed in an electric field. The rate of this migration depends on the electric field strength, the net charge on the molecule, its size and shape, and the properties of the matrix through which it moves [3] [4]. For nucleic acids, which carry a inherent negative charge, and proteins, whose charge depends on the pH of their surroundings, electrophoresis provides a powerful method for separation, analysis, and purification.
The Polyacrylamide Gel Matrix
The polyacrylamide gel is a key component for high-resolution electrophoresis, particularly for protein separation and the analysis of small nucleic acids. It is created through a chemical reaction where acrylamide monomers are cross-linked by N,N'-methylenebisacrylamide (bis-acrylamide) to form a flexible three-dimensional mesh [3] [5]. This polymerization reaction is catalyzed by ammonium persulfate (APS) and TEMED (N,N,N',N'-tetramethylethylenediamine), which generates free radicals to initiate the chain formation [3].
The properties of the gel are defined by two main factors:
- Total acrylamide concentration (%T): This determines the average pore size of the gel. Lower percentages (e.g., 8%) create larger pores, suitable for separating high molecular weight proteins. Higher percentages (e.g., 15%) create smaller pores, ideal for resolving low molecular weight proteins [3].
- Cross-linking ratio (%C): This is the proportion of bis-acrylamide to total acrylamide, which affects the rigidity of the gel matrix [3].
Researchers can cast gels with a uniform concentration or as gradient gels, where the acrylamide concentration increases from top to bottom. Gradient gels provide a broader range of separation and can sharpen protein bands, effectively performing the function of a stacking gel [3]. The polyacrylamide gel is typically housed between two glass plates in a cassette, which is then placed in an electrophoresis apparatus filled with a running buffer that conducts the electric current [3] [5].
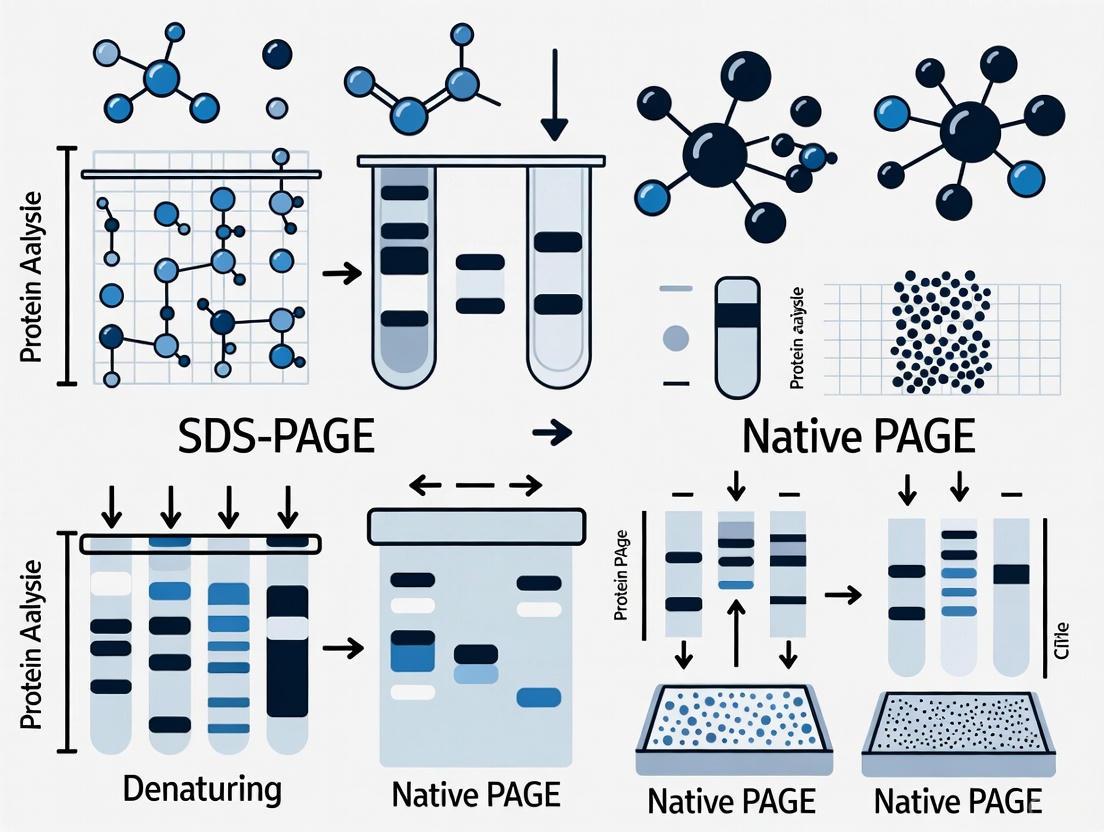
Comparative Analysis: SDS-PAGE vs. Native PAGE
Two of the most prevalent techniques using the polyacrylamide gel matrix are SDS-PAGE and Native PAGE. While both are forms of polyacrylamide gel electrophoresis, their methodologies and applications differ significantly, making each suitable for distinct research goals. The table below provides a structured, point-by-point comparison of these two fundamental methods.
Table 1: Key Differences Between SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight (mass) of polypeptide chains [6] [3] | Native size, overall charge, and 3D shape of the protein [6] [3] |
| Gel Conditions | Denaturing gel [6] | Non-denaturing gel [6] |
| Key Reagent (SDS) | Present: Denatures proteins and imparts uniform negative charge [6] [5] | Absent [6] |
| Sample Preparation | Heated with SDS and a reducing agent (e.g., DTT, β-mercaptoethanol) [6] [5] | Not heated; no denaturing or reducing agents [6] |
| Protein State | Denatured and linearized [6] [3] | Native, folded conformation [6] [3] |
| Protein Function Post-Separation | Destroyed [6] | Largely retained [6] [7] |
| Protein Recovery | Not typically recoverable in functional form [6] | Can be recovered for functional studies [6] |
| Primary Applications | Molecular weight estimation, purity assessment, protein expression analysis [6] [5] | Studying oligomeric structure, subunit composition, and enzymatic activity [6] [3] |
Detailed Experimental Protocols
Protocol for SDS-PAGE
- Sample Preparation: The protein sample is mixed with a loading buffer containing SDS, a reducing agent (like DTT or β-mercaptoethanol), glycerol, and a tracking dye (e.g., bromophenol blue). This mixture is heated at 70-100°C for 5-10 minutes to fully denature the proteins [6] [5].
- Gel Setup: A polyacrylamide gel, comprising a low-percentage stacking gel (pH ~6.8) on top of a higher-percentage resolving gel (pH ~8.8), is placed in an electrophoresis chamber filled with a running buffer (e.g., Tris-Glycine with SDS) [3] [5].
- Loading and Running: The denatured samples and molecular weight standards are loaded into the wells. The power supply is connected, and a constant voltage (e.g., 200V for a mini-gel) is applied for 30-60 minutes, or until the dye front reaches the bottom of the gel [7] [5].
- Visualization: After electrophoresis, the gel is stained with Coomassie Brilliant Blue, SYPRO Ruby, or silver stain to visualize the separated protein bands [3] [5].
Protocol for Native PAGE
- Sample Preparation: The protein sample is mixed with a non-denaturing sample buffer that lacks SDS and reducing agents. The sample is not heated to preserve native structure and function [6].
- Gel Setup: A polyacrylamide gel without SDS is cast. The running buffer also contains no SDS or denaturing agents. To maintain protein stability, the electrophoresis apparatus is often run at 4°C to dissipate heat [6].
- Loading and Running: The native samples are loaded, and a constant voltage is applied. Proteins migrate towards the electrode of opposite charge, with their mobility determined by their intrinsic charge and the sieving effect of the gel [3].
- Detection: Proteins can be visualized by staining. Furthermore, specific activity stains can be used directly on the gel to detect functional enzymes, or proteins can be electro-eluted for further functional studies [6] [3].
Experimental Data and Advanced Variations
Research has quantified the functional outcomes of these techniques. One study found that while standard SDS-PAGE resulted in only 26% retention of bound zinc in metalloproteins, a modified "Native SDS-PAGE" (NSDS-PAGE) protocol that omits heating and reduces SDS concentration achieved 98% metal retention [7]. Furthermore, this study demonstrated that seven out of nine model enzymes retained their activity after NSDS-PAGE, whereas all were denatured in standard SDS-PAGE [7]. This highlights the critical impact of protocol details on experimental outcomes.
Advanced variations of native PAGE have been developed for specific applications:
- Blue Native PAGE (BN-PAGE): Uses Coomassie dye to confer additional negative charge on native protein complexes, allowing their separation while preserving protein-protein interactions [6] [7].
- Clear Native PAGE (CN-PAGE): Separates proteins based on their native charge in a gradient gel without using Coomassie dye [6].
The following diagram illustrates the logical workflow and key decision points when choosing and executing these electrophoresis methods.
The Scientist's Toolkit: Essential Research Reagents
Successful electrophoresis relies on a suite of specialized reagents and materials. The table below details the core components of a typical SDS-PAGE workflow, which is one of the most common applications of the polyacrylamide gel matrix.
Table 2: Essential Reagents and Materials for SDS-PAGE
| Item | Function / Role in the Experiment |
|---|---|
| Acrylamide / Bis-acrylamide | Forms the cross-linked polymer network of the gel, creating the sieving matrix for separation [3] [5]. |
| Ammonium Persulfate (APS) & TEMED | Catalysts that initiate and accelerate the polymerization reaction of the polyacrylamide gel [3] [5]. |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and binds to them to impart a uniform negative charge [6] [5]. |
| Reducing Agent (DTT or BME) | Cleaves disulfide bonds in proteins, ensuring complete denaturation into individual subunits [6] [5]. |
| Tris-based Buffers | Provides the appropriate pH environment for gel polymerization and electrophoresis (e.g., Tris-HCl for gel components, Tris-Glycine for running buffer) [3] [5]. |
| Molecular Weight Standards | A mixture of proteins of known sizes run alongside samples to calibrate the gel and estimate the molecular weight of unknown proteins [3] [5]. |
| Coomassie Brilliant Blue / Silver Stain | Dyes that bind to proteins post-electrophoresis, allowing visualization of the separated bands [3] [5]. |
Electrophoresis using a polyacrylamide gel matrix is a cornerstone of modern biological research. The choice between SDS-PAGE and Native PAGE is fundamental and is dictated by the specific research question. SDS-PAGE is the go-to method for determining molecular weight, assessing purity, and analyzing protein composition under denaturing conditions. In contrast, Native PAGE is indispensable when the goal is to study proteins in their functional, native state—preserving enzymatic activity, protein-protein interactions, and tertiary structure. Understanding the principles, strengths, and limitations of each technique allows researchers to design robust experimental strategies for effective protein analysis in drug development and basic science.
Protein gel electrophoresis is a foundational laboratory technique in which charged protein molecules are transported through a porous matrix by an electrical field, enabling their separation based on physical and chemical properties [3]. This method provides a simple, rapid, and sensitive analytical tool for characterizing complex protein mixtures. The mobility of a molecule through an electric field depends on several factors: field strength, net charge, molecular size and shape, ionic strength, and the properties of the matrix through which migration occurs [3].
Polyacrylamide gel electrophoresis (PAGE) represents the most widely used approach for protein separation, with two primary variants serving distinct analytical purposes: SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis) and Native PAGE [6] [8]. These techniques differ fundamentally in their treatment of protein structure and the type of information they provide. SDS-PAGE employs denaturing conditions to separate proteins primarily by molecular weight, while Native PAGE maintains proteins in their native conformation, preserving biological activity and enabling separation based on size, charge, and shape [9] [3].
The selection between these methods depends entirely on the research objectives. SDS-PAGE is ideal for determining molecular weight, assessing purity, and studying protein expression levels, whereas Native PAGE is preferred for investigating protein-protein interactions, oligomeric states, enzymatic activity, and native structure [6] [9]. Understanding the fundamental principles, applications, and limitations of each technique is essential for designing appropriate experimental approaches in biochemistry, molecular biology, and drug development.
Fundamental Principles of SDS-PAGE
The Role of SDS in Protein Denaturation and Uniform Charge Conferment
SDS-PAGE separates proteins based almost exclusively on their molecular weight through a sophisticated denaturation process that masks intrinsic protein characteristics [3]. The key to this technique is sodium dodecyl sulfate (SDS), an anionic detergent that binds extensively to protein molecules in a constant weight ratio of approximately 1.4 grams of SDS per 1 gram of polypeptide [3]. This binding process unfolds the proteins into linear chains by wrapping around the polypeptide backbone, effectively disrupting hydrogen bonds, hydrophobic interactions, and other non-covalent forces that maintain secondary and tertiary structure [6] [3].
The bound SDS molecules impart a uniform negative charge to all proteins in direct proportion to their molecular mass, effectively overwhelming any inherent charge differences arising from the variable amino acid compositions of different proteins [8] [3]. Consequently, the charge-to-mass ratio becomes essentially identical for all SDS-coated polypeptides, eliminating charge as a significant variable in electrophoretic migration [3]. When an electric field is applied, all protein-SDS complexes migrate toward the positively charged anode with mobility determined primarily by molecular size as they sieve through the porous polyacrylamide matrix [9].
Molecular Sieving in Polyacrylamide Gels
The polyacrylamide gel matrix serves as a molecular sieve that regulates protein migration based on size [3]. Polyacrylamide gels are formed through the polymerization of acrylamide monomers cross-linked by N,N'-methylenebisacrylamide (bisacrylamide), creating a mesh-like network with defined pore sizes [3]. The porosity of the gel is controlled by adjusting the concentrations of acrylamide and bisacrylamide, with higher percentages creating smaller pores that provide better resolution for lower molecular weight proteins [3].
During electrophoresis, smaller proteins navigate through the gel matrix more easily than larger counterparts, resulting in faster migration rates [6] [3]. This molecular sieving effect enables the separation of polypeptides based on molecular weight, with the relationship between migration distance and log molecular weight being approximately linear under standardized conditions [3]. The discontinuous buffer system, incorporating stacking and resolving gels with different pore sizes and pH values, further enhances resolution by concentrating proteins into sharp bands before they enter the main separating phase of the gel [3].
Table 1: Key Components of SDS-PAGE and Their Functions
| Component | Function | Typical Composition |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins and confers uniform negative charge | 0.1-0.2% in buffers [3] |
| Polyacrylamide Gel | Provides molecular sieving matrix for separation | 5-20% acrylamide depending on target protein size [3] |
| Reducing Agents (DTT, BME) | Breaks disulfide bonds for complete denaturation | 1-5% in sample buffer [6] |
| Tris-based Buffers | Maintains appropriate pH for electrophoresis | Tris-HCl, Tris-Glycine, or Bis-Tris systems [3] |
| Tracking Dye | Visualizes migration progress | Bromophenol blue or similar dye [3] |
SDS-PAGE Experimental Methodology
Sample Preparation and Denaturation Protocol
Proper sample preparation is critical for successful SDS-PAGE separation. Protein samples are typically mixed with an SDS-containing sample buffer that includes a reducing agent such as dithiothreitol (DTT) or β-mercaptoethanol to break disulfide bonds [6] [3]. This buffer also contains glycerol to increase density, preventing diffusion from wells, and a tracking dye to monitor migration progress [8]. The sample mixture is then heated to 70-100°C for 5-10 minutes to ensure complete denaturation and SDS binding [6] [3].
Heating facilitates the unfolding of protein structures and promotes the reduction of disulfide linkages, ensuring that multimeric proteins dissociate into their individual subunits [3]. For non-reducing SDS-PAGE, the reducing agent is omitted from the sample buffer, preserving disulfide-bonded complexes while maintaining denaturation by SDS [10]. This variation can provide information about protein quaternary structure and disulfide connectivity, as proteins maintain their covalent associations while losing non-covalent interactions [10].
Gel Composition and Electrophoresis Conditions
SDS-PAGE is typically performed using discontinuous buffer systems in vertically oriented gel apparatuses [3]. The gel consists of two distinct regions: a stacking gel with larger pores (lower acrylamide concentration, typically 4-5%) and lower pH (approximately 6.8) that concentrates proteins into a sharp starting zone, and a resolving gel with smaller pores (higher acrylamide concentration, typically 8-20%) and higher pH (approximately 8.8) where separation occurs based on molecular size [3]. Gradient gels with increasing acrylamide concentration from top to bottom can enhance resolution across a broader molecular weight range [3].
The electrophoresis running buffer typically contains Tris-based buffers at alkaline pH, glycine as a leading ion, and SDS (0.1% or lower) to maintain protein denaturation during separation [3] [7]. Electrophoresis is performed at constant voltage (typically 100-200V for mini-gels) for 30-60 minutes, with temperature maintained at room temperature to prevent overheating that could cause diffusion or gel distortion [6]. The process is complete when the tracking dye front reaches the bottom of the gel, indicating that separation has occurred throughout the entire length of the matrix [3].
Table 2: Standard SDS-PAGE Protocol Components and Parameters
| Step | Key Components | Optimal Conditions | Purpose |
|---|---|---|---|
| Sample Preparation | Protein sample, SDS, reducing agent, buffer, glycerol | Heating at 70-100°C for 5-10 min [6] [3] | Denature proteins, disrupt disulfide bonds |
| Gel Casting | Acrylamide, bis-acrylamide, APS, TEMED, buffer | Stacking gel: 4-5%, pH 6.8; Resolving gel: 8-20%, pH 8.8 [3] | Create molecular sieve with appropriate pore sizes |
| Electrophoresis | Running buffer (Tris, Glycine, SDS), power supply | Constant voltage (100-200V), room temperature, 30-60 min [6] [3] | Drive protein migration through gel matrix |
| Detection | Coomassie Blue, Silver Stain, fluorescent dyes | Incubation with stain followed by destaining [3] | Visualize separated protein bands |
Native PAGE: Separation of Proteins in Their Native State
Principle of Native PAGE
Unlike SDS-PAGE, Native PAGE (also called non-denaturing PAGE) separates proteins while maintaining their native conformation, quaternary structure, and biological activity [6] [9]. In this technique, no denaturing agents are used, allowing proteins to retain their folded three-dimensional structure, subunit interactions, and enzymatic functions [8] [3]. Separation occurs based on the combined influence of the protein's intrinsic charge, size, and shape as it migrates through the polyacrylamide matrix [6] [3].
In Native PAGE, proteins migrate according to their charge-to-mass ratio and the frictional forces they encounter in the gel matrix [3]. The net charge at the pH of the running buffer determines electrophoretic mobility, with more highly charged proteins migrating faster, while larger proteins experience greater frictional resistance and migrate slower [3]. The gel pore size creates a sieving effect that depends on the protein's hydrodynamic volume and shape, enabling discrimination between different native protein configurations [9]. This multi-parameter separation can provide information about oligomerization states, protein-protein interactions, and conformational changes that would be disrupted under denaturing conditions [9].
Native PAGE Methodology and Variations
Native PAGE employs similar equipment to SDS-PAGE but with critical differences in buffer composition and sample treatment [6]. Sample preparation is minimal, typically involving mixture with a non-denaturing buffer that may contain glycerol to facilitate loading but omits SDS, reducing agents, and heating steps [6] [3]. The running buffers lack SDS and may be formulated at various pH values to maintain protein stability and function, with cooler operating temperatures (often 4°C) recommended to preserve labile proteins [6].
Two main variants of Native PAGE have been developed: Blue Native PAGE (BN-PAGE) uses Coomassie brilliant blue dye, which binds to proteins without denaturation and imparts negative charge, improving solubility and separation resolution [6] [7]. Clear Native PAGE (CN-PAGE) separates proteins based solely on their intrinsic charge in gradient gels without dye binding [6]. BN-PAGE generally provides higher resolution than CN-PAGE and is particularly valuable for studying membrane protein complexes and multiprotein assemblies [7].
Table 3: Comparative Analysis: SDS-PAGE vs. Native PAGE
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight primarily [6] [3] | Size, charge, and shape [6] [9] |
| Gel Conditions | Denaturing (SDS present) [6] [8] | Non-denaturing (no SDS) [6] [8] |
| Sample Treatment | Heating with SDS and reducing agents [6] | No heating, no denaturants [6] |
| Protein Structure | Denatured, linearized [9] [3] | Native, folded conformation [9] [3] |
| Protein Function | Lost during separation [6] [9] | Retained after separation [6] [9] |
| Protein Recovery | Not typically recoverable functional [6] [8] | Recoverable in functional form [6] [8] |
| Molecular Weight Determination | Accurate for polypeptide chains [3] | Approximate, requires standards of similar shape/charge [9] |
| Applications | MW determination, purity assessment, expression analysis [6] [3] | Oligomeric state, protein-protein interactions, enzymatic activity [6] [9] |
| Running Temperature | Room temperature [6] | Typically 4°C [6] |
Comparative Experimental Data and Case Studies
Direct Comparison of Separation Outcomes
Experimental data clearly demonstrate the differential separation profiles obtained with SDS-PAGE versus Native PAGE. In one illustrative case study, a protein sample isolated from a natural source displayed distinct migration patterns when analyzed by the two techniques [10]. When electrophoresed on non-reducing SDS-PAGE, the protein migrated as a band corresponding to 60 kDa, suggesting this was the molecular weight of its constituent polypeptides [10]. However, when the same protein was analyzed by Native PAGE, it migrated corresponding to a 120 kDa marker protein, indicating the native protein existed as a dimer of two 60 kDa subunits that were not linked by disulfide bonds [10].
This differential migration behavior provides valuable insights into protein quaternary structure. The dissociation into monomers under SDS treatment (even without reducing agents) indicates that the dimeric structure is maintained by non-covalent interactions (hydrophobic, electrostatic, or hydrogen bonding) rather than disulfide bridges [10]. Such information is crucial for understanding protein function, stability, and regulation, demonstrating how complementary use of both techniques can reveal different aspects of protein architecture.
Hybrid Approaches: Native SDS-PAGE
Innovative methodological adaptations have emerged to bridge the gap between the high resolution of SDS-PAGE and the functional preservation of Native PAGE. One such approach, termed Native SDS-PAGE (NSDS-PAGE), modifies standard SDS-PAGE conditions by significantly reducing SDS concentration in the running buffer (from 0.1% to 0.0375%), eliminating EDTA from buffers, and omitting the heating step during sample preparation [7].
This modified technique achieves remarkable preservation of protein function while maintaining high resolution separation. Experimental results demonstrate that zinc retention in proteomic samples increased from 26% with standard SDS-PAGE to 98% with NSDS-PAGE [7]. Furthermore, functional assays revealed that seven of nine model enzymes, including four zinc-containing proteins, retained activity after NSDS-PAGE separation, whereas all were denatured during standard SDS-PAGE [7]. All nine enzymes maintained activity following BN-PAGE, though with lower resolution than achieved with NSDS-PAGE [7]. This hybrid approach offers a valuable compromise when both high resolution and partial functional retention are desired.
Essential Research Reagent Solutions
Successful implementation of protein electrophoresis techniques requires specific reagent systems optimized for each method. The research reagent market offers comprehensive solutions for both SDS-PAGE and Native PAGE workflows, with key manufacturers including Thermo Fisher Scientific, Bio-Rad Laboratories, Merck KGaA, and Danaher Corporation [11] [12]. These companies provide integrated product portfolios spanning instrumentation, consumables, and detection systems designed to ensure reproducible results across diverse laboratory settings.
Table 4: Essential Research Reagents for Protein Electrophoresis
| Reagent Category | Specific Products | Applications | Key Features |
|---|---|---|---|
| Precast Gels | NuPAGE Novex Bis-Tris Gels (Thermo Fisher), Mini-PROTEAN TGX Gels (Bio-Rad) | SDS-PAGE, Native PAGE [3] [7] | Consistent performance, multiple percentages, gradient options |
| Electrophoresis Buffers | MOPS, MES, Tris-Glycine buffers with/without SDS | SDS-PAGE, Native PAGE [3] [7] | Optimized pH and ionic strength, premixed formulations |
| Protein Standards | Prestained/Unstained molecular weight markers, NativeMark standards | Molecular weight estimation, migration monitoring [3] | Precise molecular weight calibration, visible tracking |
| Staining Solutions | Coomassie Blue, Silver Stain, SYPRO Ruby, SimplyBlue SafeStain | Protein detection after electrophoresis [3] | varying sensitivity, compatibility with downstream analysis |
| Transfer Systems | Nitrocellulose/PVDF membranes, transfer buffers | Western blotting following SDS-PAGE [9] | Efficient protein transfer, minimal loss of resolution |
| Specialized Kits | BN-PAGE, CN-PAGE kits, 2D electrophoresis systems | Native PAGE, complex protein separation [7] | Integrated components for specific techniques |
SDS-PAGE remains an indispensable technique in molecular biology and biochemistry laboratories worldwide, providing reliable separation of complex protein mixtures based primarily on molecular weight under denaturing conditions [6] [3]. Its simplicity, reproducibility, and compatibility with downstream analytical methods like western blotting and mass spectrometry have established it as a cornerstone of proteomic analysis [9] [3]. The denaturing nature of SDS-PAGE, while destroying native protein structure and function, enables precise molecular weight determination and reveals information about protein subunit composition that would be obscured in native separations [10].
Native PAGE serves as a powerful complementary technique that preserves protein structure, function, and interactions, making it invaluable for studying oligomeric states, protein complexes, and enzymatic activities [9]. The choice between these techniques should be guided by specific research objectives, with SDS-PAGE preferred for molecular weight analysis and purity assessment, and Native PAGE selected for functional studies and native structure investigation [6] [9]. Emerging hybrid approaches like NSDS-PAGE demonstrate ongoing methodological innovation aimed at balancing the high resolution of denaturing methods with the functional preservation of native techniques [7].
For researchers in drug development and biotechnology, understanding the capabilities and limitations of each electrophoretic method is essential for designing appropriate characterization protocols for therapeutic proteins, vaccines, and diagnostic reagents. The complementary data obtained from both techniques can provide comprehensive insights into protein attributes relevant to efficacy, stability, and safety, supporting the development of biologically relevant and therapeutically effective products.
In the field of protein analysis, polyacrylamide gel electrophoresis (PAGE) serves as a fundamental tool for separating and characterizing complex protein mixtures. Among the various PAGE techniques, Native PAGE stands out for its unique ability to analyze proteins in their natural, functional state. Unlike its denaturing counterpart, SDS-PAGE, which unravels protein structures, Native PAGE preserves the intricate three-dimensional architecture of proteins, allowing researchers to study them with their biological activities intact. This capacity makes it indispensable for investigating protein complexes, conformational changes, and functional interactions—aspects that are completely lost in denaturing methods. This guide provides a comprehensive comparison between Native PAGE and SDS-PAGE, examining their principles, applications, and methodological details to help researchers select the optimal technique for their specific experimental needs.
Core Principles and Comparative Analysis
Native PAGE separates proteins based on their intrinsic charge, size, and three-dimensional shape under non-denaturing conditions [13] [9]. The gel operates without denaturing agents, preserving the protein's native conformation and, consequently, its biological activity [6] [8]. The separation mechanism depends on the protein's charge-to-mass ratio and the sieving effect of the gel matrix, where smaller proteins migrate faster than larger ones [13]. In alkaline running buffers, most proteins carry a net negative charge and migrate toward the positive anode [13]. For proteins with basic isoelectric points (pI) that would normally carry a positive charge, systems like NativePAGE Bis-Tris use Coomassie G-250 dye to bind and confer a net negative charge, ensuring all proteins migrate toward the anode [13].
SDS-PAGE employs the anionic detergent sodium dodecyl sulfate (SDS) to denature proteins and mask their intrinsic charges [14]. SDS binds uniformly to the protein backbone at a constant ratio (approximately 1.4g SDS per 1g of protein), linearizing the proteins and imparting a uniform negative charge [14]. This process eliminates the influence of native charge and protein shape, resulting in separation based almost exclusively on molecular weight [6] [15]. The use of reducing agents like DTT or beta-mercaptoethanol further breaks disulfide bonds, ensuring complete denaturation [6] [14].
Table: Fundamental Differences Between Native PAGE and SDS-PAGE
| Criteria | Native PAGE | SDS-PAGE |
|---|---|---|
| Separation Basis | Size, charge, and shape of native protein [6] [9] | Molecular weight only [6] [15] |
| Gel Condition | Non-denaturing [6] [8] | Denaturing [6] [8] |
| SDS Presence | Absent [6] | Present [6] [14] |
| Protein State | Native, folded conformation [6] [9] | Denatured, linearized [6] [14] |
| Protein Function | Retained post-separation [6] [9] | Lost post-separation [6] |
| Protein Recovery | Possible for functional studies [6] [8] | Not recoverable in functional form [6] |
| Primary Applications | Studying structure, subunit composition, function, and protein complexes [6] [9] | Determining molecular weight, checking protein expression, and purity analysis [6] [9] |
Experimental Protocols and Methodologies
Standard Native PAGE Protocol
The following protocol outlines a typical Native PAGE procedure using a Tris-Glycine gel system [13]:
- Sample Preparation: Mix the protein sample with a non-denaturing Native Sample Buffer (e.g., Tris-Glycine Native Sample Buffer). Crucially, do not heat the samples and avoid any denaturing or reducing agents [6] [13].
- Gel Selection: Choose an appropriate polyacrylamide gel concentration based on the target protein size. For example, Novex Tris-Glycine gels are suitable for proteins between 20-500 kDa [13].
- Electrophoresis: Load the prepared samples into the wells. Run the gel using a native running buffer (e.g., Tris-Glycine Native Running Buffer) at recommended voltages. To preserve labile protein complexes, the run is often performed at 4°C [6].
- Detection: After electrophoresis, proteins can be visualized using standard staining techniques (e.g., Coomassie Brilliant Blue or silver staining). For functional analysis, activity assays can be performed directly on the gel [6].
Advanced Native PAGE Systems
Beyond the traditional Tris-Glycine system, specialized Native PAGE systems have been developed for enhanced performance:
- Blue Native PAGE (BN-PAGE): This method, on which the NativePAGE Bis-Tris system is based, incorporates Coomassie G-250 dye in the cathode buffer [13]. The dye binds non-specifically to hydrophobic protein regions, conferring a negative charge while maintaining the protein's native state. This is particularly advantageous for membrane proteins and complexes with basic pI values, as it prevents aggregation and ensures all proteins migrate toward the anode [13].
- Clear Native PAGE (CN-PAGE): A variation that separates proteins based on their intrinsic charge in a gradient gel without using Coomassie dye [6].
- Native SDS-PAGE (NSDS-PAGE): A hybrid method developed to bridge the resolution gap. It uses significantly reduced SDS concentrations (e.g., 0.0375% in the running buffer) and omits the heating and reducing agents from the sample preparation [7]. This protocol was shown to retain 98% of bound Zn²⁺ in proteomic samples and preserve the activity of seven out of nine model enzymes, while still achieving high-resolution separation comparable to standard SDS-PAGE [7].
Table: Comparison of Native PAGE Gel Systems [13]
| Gel System | Operating pH Range | Key Features | Ideal Use Cases |
|---|---|---|---|
| Novex Tris-Glycine | 8.3 - 9.5 | Traditional Laemmli system | Keeping the native net charge of proteins; studying smaller proteins (20-500 kDa) |
| NuPAGE Tris-Acetate | 7.2 - 8.5 | Better resolution of larger proteins | Keeping the native net charge of proteins; studying larger proteins (>150 kDa) |
| NativePAGE Bis-Tris | ~7.5 | Uses G-250 dye; resolution by molecular weight regardless of pI | Membrane/hydrophobic proteins; separating by molecular weight under native conditions |
Data Presentation and Experimental Findings
The functional differences between these techniques are substantiated by quantitative experimental data. A key study evaluating the retention of metal ions and enzymatic activity underscores the practical implications of choosing a native versus denaturing approach.
Table: Experimental Comparison of Protein Function Retention Across PAGE Methods [7]
| Electrophoresis Method | Zn²⁺ Retention in Proteomic Samples | Enzymatic Activity Retention (Model Enzymes) | Key Modification |
|---|---|---|---|
| Standard SDS-PAGE | 26% | 0 out of 9 active | Sample heating, 0.1% SDS, reducing agent |
| BN-PAGE | Not Specified | 9 out of 9 active | No SDS, non-denaturing conditions |
| NSDS-PAGE | 98% | 7 out of 9 active | No sample heat, low SDS (0.0375%), no reducing agent |
This data highlights that modified protocols like NSDS-PAGE can offer a compelling compromise, providing high-resolution separation while maintaining a significant degree of protein functionality and bound metal ions [7].
Visualization of Experimental Workflows
The diagram below illustrates the key procedural differences and outcomes between Native PAGE and SDS-PAGE workflows.
The Scientist's Toolkit: Key Research Reagent Solutions
Successful protein separation relies on a set of essential reagents, each with a specific function. The table below details key materials for Native PAGE and SDS-PAGE.
Table: Essential Reagents for PAGE Experiments
| Reagent / Material | Function | Native PAGE | SDS-PAGE |
|---|---|---|---|
| Polyacrylamide Gel | Sieving matrix for size-based separation [6] | Used at various concentrations [13] | Used at various concentrations [14] |
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins and confers negative charge [14] | Absent [6] | Present in sample and running buffers [6] [14] |
| Coomassie G-250 Dye | Imparts charge for consistent migration under native conditions [13] | Used in BN-PAGE systems [13] | Not used |
| Reducing Agent (DTT/BME) | Breaks disulfide bonds [14] | Absent [6] | Present [6] [14] |
| Glycerol | Increases sample density for well loading [8] | Used in sample buffer [7] | Used in sample buffer [7] |
| Tracking Dye | Visualizes migration front during run | e.g., Phenol Red [7] | e.g., Bromophenol Blue [16] |
| Blotting Membrane | For subsequent Western blot analysis | PVDF (recommended) [13] | Nitrocellulose or PVDF |
Native PAGE and SDS-PAGE are complementary, not competing, techniques in the protein researcher's arsenal. The choice between them is dictated by the fundamental question of the experiment: is the goal to understand protein identity, weight, and purity, or to probe its native structure, complex formation, and biological activity? SDS-PAGE offers a robust, standardized approach for determining molecular weight and analyzing protein composition under denaturing conditions. In contrast, Native PAGE provides a unique window into the functional proteome, preserving the delicate interactions and structures that define a protein's role in the cell. The development of hybrid methods like NSDS-PAGE further expands the toolkit, offering promising avenues for high-resolution separation without complete sacrifice of protein function. By understanding the principles, protocols, and applications detailed in this guide, researchers can make an informed decision to advance their protein analysis research effectively.
In protein analysis research, the choice of electrophoretic technique fundamentally shapes experimental outcomes. The selection between SDS-PAGE (Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis) and Native PAGE dictates whether proteins are studied in a denatured state for molecular weight determination or in their native conformation for functional analysis [6] [3]. This comparison guide objectively examines how key reagents—SDS, reducing agents, and buffer systems—orchestrate these divergent outcomes, providing researchers with the experimental data needed to inform protocol selection.
Core Principles and Separation Mechanisms
SDS-PAGE: Denaturing Separation by Mass
SDS-PAGE employs the anionic detergent sodium dodecyl sulfate (SDS) to denature proteins and impart a uniform negative charge. SDS binds to hydrophobic regions of proteins at a consistent ratio of approximately 1.4g SDS per 1g of protein, masking intrinsic charge differences and unfolding tertiary structures [3] [17]. When combined with reducing agents like dithiothreitol (DTT) or β-mercaptoethanol, which break disulfide bonds, proteins become linearized polypeptides that migrate strictly according to molecular weight [6] [17]. The polyacrylamide gel matrix serves as a molecular sieve, with smaller proteins migrating faster than larger ones [3].
Native PAGE: Non-Denaturing Separation by Charge and Size
Native PAGE preserves protein structure and function by omitting denaturing agents. Separation depends on the protein's intrinsic net charge, size, and three-dimensional shape [6] [3]. In the alkaline running buffers typically used, most proteins carry a net negative charge and migrate toward the anode. The gel matrix creates a sieving effect, regulating movement according to size and shape [3]. This technique maintains subunit interactions within multimeric proteins and preserves enzymatic activity [3].
Comparative Analysis of Key Reagents
The differential use of reagents creates distinct electrophoretic environments, as summarized in the table below.
Table 1: Comparative Roles of Key Reagents in SDS-PAGE vs. Native PAGE
| Key Reagent | SDS-PAGE | Native PAGE |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Present: Denatures proteins, imparts uniform negative charge, disrupts non-covalent interactions [6] [17]. | Absent: Proteins retain native conformation and intrinsic charge [6]. |
| Reducing Agents (DTT, BME) | Present in reducing SDS-PAGE: Breaks disulfide bonds, linearizes proteins [6] [18]. | Absent: Disulfide bonds and non-covalent interactions remain intact [6]. |
| Buffer System | Discontinuous (stacking & resolving gels) with SDS in running buffer; creates conditions for size-based separation [3] [17]. | Various non-denaturing buffers; may use Tris-glycine or imidazole systems without SDS [6]. |
| Sample Preparation | Samples heated (70-100°C) in SDS-containing buffer [6] [3]. | Samples not heated and prepared in non-denaturing buffer [6]. |
| Primary Separation Basis | Molecular weight [6] [3]. | Size, shape, and intrinsic charge [6] [3]. |
| Protein State Post-Separation | Denatured, inactive [6] [3]. | Native, folded, often functional [6] [3]. |
Experimental Data and Performance Comparison
Quantitative Proteomic Profiling Comparison
A comparative study of 1D SDS-PAGE versus nondenaturing 2DE for analyzing proteins from human bronchial smooth muscle cells revealed distinct performance characteristics [19]:
Table 2: Protein Identification by Electrophoresis Method with LC-MS/MS
| Separation Method | Number of Proteins Identified | Percent Abundance Range | Key Advantages |
|---|---|---|---|
| 1D SDS-PAGE-MS | 2,552 proteins | 3.5% to 2×10⁻⁴% | Superior for visualizing quantitative differences between samples [19]. |
| Nondenaturing 2DE-MS | 4,323 proteins | 3.6% to 1×10⁻⁵% | Superior for visualizing protein-protein interactions; higher sensitivity [19]. |
The study concluded these methods provide complementary information for cellular protein analysis, with nondenaturing 2DE showing increased sensitivity for detecting membrane proteins [19].
Enzyme Activity Retention Under Different Conditions
Research comparing standard SDS-PAGE, BN-PAGE, and a modified "Native SDS-PAGE" (NSDS-PAGE) demonstrated significant differences in functional retention [7]:
Table 3: Enzyme Activity and Metal Retention Across PAGE Methods
| Method | Zn²⁺ Retention in Proteomic Samples | Enzymatic Activity Retention (Model Enzymes) | Key Characteristics |
|---|---|---|---|
| Standard SDS-PAGE | 26% | 0/9 enzymes active | Complete denaturation, high resolution [7]. |
| BN-PAGE | Not specified | 9/9 enzymes active | Preserves function but with lower resolution [7]. |
| NSDS-PAGE | 98% | 7/9 enzymes active | High resolution with most native functions retained [7]. |
NSDS-PAGE achieved this by removing EDTA from sample buffers, omitting the heating step, and reducing SDS in the running buffer from 0.1% to 0.0375% [7].
Detailed Experimental Protocols
Protocol 1: Standard Denaturing SDS-PAGE
Based on Invitrogen NuPAGE specifications [7]:
- Sample Preparation: Mix 7.5μL protein sample with 2.5μL 4X LDS sample buffer (contains LDS detergent). Heat at 70°C for 10 minutes to denature proteins [7].
- Gel Composition: Use pre-cast NuPAGE Novex 12% Bis-Tris 1.0mm minigels [7].
- Running Buffer: 50mM MOPS, 50mM Tris Base, 1mM EDTA, 0.1% SDS, pH 7.7 [7].
- Electrophoresis Conditions: Run at constant voltage (200V) for approximately 45 minutes at room temperature until dye front reaches gel bottom [7].
Protocol 2: Blue Native PAGE (BN-PAGE)
Based on Invitrogen NativePAGE system [7]:
- Sample Preparation: Mix 7.5μL protein sample with 2.5μL 4X BN-PAGE sample buffer (no detergents). Do not heat samples [7].
- Gel Composition: Use pre-cast NativePAGE Novex 4-16% Bis-Tris gradient gels [7].
- Running Buffer:
- Cathode Buffer: 50mM BisTris, 50mM Tricine, 0.02% Coomassie G-250, pH 6.8
- Anode Buffer: 50mM BisTris, 50mM Tricine, pH 6.8 [7]
- Electrophoresis Conditions: Run at constant voltage (150V) at room temperature for 90-95 minutes [7].
Protocol 3: Modified Native SDS-PAGE (NSDS-PAGE)
Method optimizing for both resolution and function retention [7]:
- Sample Preparation: Mix 7.5μL protein sample with 2.5μL 4X NSDS sample buffer (100mM Tris HCl, 150mM Tris base, 10% glycerol, 0.0185% Coomassie G-250, 0.00625% Phenol Red, pH 8.5). No heating and no SDS or EDTA in sample buffer [7].
- Gel Composition: Use standard NuPAGE Novex 12% Bis-Tris 1.0mm mini-gels [7].
- Running Buffer: 50mM MOPS, 50mM Tris Base, 0.0375% SDS (reduced from standard 0.1%), no EDTA, pH 7.7 [7].
- Electrophoresis Conditions: Pre-run gels at 200V for 30 minutes in ddH₂O before loading samples. Run with NSDS running buffer at 200V for standard time [7].
Visualization of Method Workflows
Research Reagent Solutions
The following table details essential reagents and their specific functions in protein electrophoresis protocols:
Table 4: Essential Research Reagents for Protein Electrophoresis
| Reagent Category | Specific Examples | Function & Mechanism | Application Notes |
|---|---|---|---|
| Denaturing Detergents | Sodium Dodecyl Sulfate (SDS) | Binds proteins (~1.4:1 ratio), imparts uniform charge, disrupts non-covalent bonds [3] [17]. | Essential for SDS-PAGE; concentration critical (0.1% in standard protocols) [7]. |
| Reducing Agents | Dithiothreitol (DTT), β-mercaptoethanol, Tris(2-carboxyethyl)phosphine (TCEP) | Breaks disulfide bonds, linearizes proteins for accurate MW determination [6] [18]. | Used in reducing SDS-PAGE; omitted from native protocols [6]. |
| Buffer Components | Tris, MOPS, BisTris, Tricine, Imidazole | Maintains pH, provides ionic strength, facilitates electrophoresis [7]. | Discontinuous systems (stacking/resolving) enhance SDS-PAGE resolution [3] [17]. |
| Gel Matrix Components | Acrylamide, Bis-acrylamide, APS, TEMED | Forms cross-linked polyacrylamide mesh for molecular sieving [3]. | Concentration determines pore size (4-16% for native, 8-12% for SDS-PAGE) [7]. |
| Tracking Dyes | Bromophenol Blue, Phenol Red, Coomassie G-250 | Visualizes migration front during electrophoresis [7]. | Coomassie in BN-PAGE adds negative charge; phenol red used in NSDS-PAGE [7]. |
| Charge Modifiers | Coomassie G-250 (in BN-PAGE) | Imparts slight negative charge to proteins while maintaining native structure [7]. | Critical for BN-PAGE; concentration varies (0.02% in cathode buffer) [7]. |
The strategic application of SDS, reducing agents, and specialized buffer systems fundamentally determines the capabilities and limitations of both SDS-PAGE and Native PAGE. SDS-PAGE, with its complete denaturation and charge uniformity, provides unparalleled resolution for molecular weight determination and purity assessment. Native PAGE, through the preservation of native protein structures, enables functional studies and interaction analyses. The experimental data presented demonstrates that modified approaches like NSDS-PAGE can bridge these paradigms, offering high resolution while preserving substantial functionality. Researchers must align reagent selection with experimental goals, whether prioritizing structural characterization or functional analysis, to optimize protein study outcomes.
For researchers in biochemistry and drug development, selecting the appropriate electrophoretic technique is fundamental to accurately interpreting protein structure and function. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and native-PAGE (Native Polyacrylamide Gel Electrophoresis) are two foundational methods, yet they differ profoundly in their impact on protein conformation. SDS-PAGE denatures proteins, destroying higher-order structures to analyze subunits by molecular weight, while native-PAGE preserves the protein's native architecture, allowing the study of functional complexes [3] [9]. This guide provides an objective comparison of these techniques, supported by experimental data and detailed protocols, to inform your analytical strategy.
Core Principles and Separation Mechanisms
The fundamental difference between these methods lies in their treatment of protein structure, which directly dictates their separation mechanism and the type of information they yield.
Denaturing SDS-PAGE employs the anionic detergent sodium dodecyl sulfate (SDS) and often a reducing agent like β-mercaptoethanol or dithiothreitol (DTT). During sample preparation, proteins are heated to disrupt hydrogen bonds and van der Waals forces, while the reducing agent cleaves disulfide bonds [18] [20]. SDS binds uniformly to the polypeptide backbone in a constant weight ratio, masking the protein's intrinsic charge and imparting a uniform negative charge density. This process unfolds the protein into a rod-like shape, meaning separation through the polyacrylamide gel matrix occurs primarily based on molecular mass alone [3] [20].
In contrast, Native-PAGE is performed in the absence of denaturing agents. The sample buffer is SDS-free and non-reducing, which preserves the protein's secondary, tertiary, and quaternary structures. Consequently, separation is influenced by a combination of the protein's intrinsic charge, size, and three-dimensional shape [3] [9]. A protein's net charge at the running buffer's pH determines its migration direction and speed, while the gel matrix exerts a sieving effect based on the protein's overall bulk and shape.
The following diagram illustrates the key procedural differences and their direct impacts on protein structure.
Comparative Analysis: Key Characteristics
The choice between SDS-PAGE and native-PAGE involves trade-offs between structural preservation and analytical resolution. The table below summarizes their core characteristics.
Table 1: Core Characteristics of SDS-PAGE vs. Native-PAGE
| Feature | SDS-PAGE (Denaturing) | Native-PAGE (Non-Denaturing) |
|---|---|---|
| Protein State | Denatured and unfolded [9] [20] | Native, folded structure [9] [20] |
| Structure Impact | Destroys tertiary/quaternary structure; reduces disulfide bonds [18] [3] | Preserves tertiary/quaternary structure and disulfide bonds [3] |
| Separation Basis | Primarily molecular mass [3] | Net charge, size, and shape [3] |
| Biological Activity | Lost after separation [9] | Often retained after separation [3] [21] |
| Molecular Weight Determination | Accurate for polypeptide chains [3] | Not direct; requires comparison with native standards [3] |
| Key Reagents | SDS, reducing agent (e.g., DTT, β-mercaptoethanol) [18] [22] | No SDS or reducing agents; native buffers [7] [20] |
| Ideal For | Purity assessment, subunit composition, Western blotting [7] [20] | Studying oligomerization, enzyme activity assays, protein complexes [9] [21] |
Experimental Data and Interpretation
The different principles of each method lead to distinct experimental outcomes, as demonstrated by published data and case studies.
Case Study: Identifying a Non-Covalent Dimer
A classic example illustrating the power of using both techniques comes from the analysis of a protein isolated from a natural source.
- On non-reducing SDS-PAGE, the protein migrated as a single band corresponding to 60 kDa [10].
- On native-PAGE, the same protein migrated corresponding to a 120 kDa marker [10].
Inference: The protein exists as a dimer of 60 kDa subunits that are not linked by disulfide bonds. The non-covalent interactions holding the dimer together are disrupted by SDS in the non-reducing SDS-PAGE, but are preserved in native-PAGE, revealing the protein's true oligomeric state [10].
Table 2: Comparative Migration and Data Interpretation
| Experimental Observation | SDS-PAGE Interpretation | Native-PAGE Interpretation | Combined Conclusion |
|---|---|---|---|
| Single band at 60 kDa (non-reducing); Single band at 120 kDa (native) | Protein migrates as a 60 kDa polypeptide [10] | Protein migrates as a 120 kDa complex [10] | Protein is a non-covalent homodimer (2x 60 kDa subunits) [10] |
| In-gel activity staining possible after separation | Not applicable (proteins denatured) [9] | Enzymatic activity confirmed for MCAD tetramers [21] | Native-PAGE preserves function, allowing activity-based analysis. |
Advanced Application: In-Gel Activity Assay for Enzyme Analysis
A 2025 study on Medium-chain acyl-CoA dehydrogenase (MCAD) deficiency showcases a key advantage of native-PAGE. Researchers adapted a high-resolution clear native PAGE (hrCN-PAGE) method, followed by an in-gel colorimetric assay. After separation, the gel was incubated with the substrate octanoyl-CoA and nitro blue tetrazolium chloride. Active MCAD enzymes oxidized the substrate, producing a visible purple precipitate directly in the gel [21].
This method allowed the team to:
- Distinguish active tetramers from inactive, fragmented forms of clinically relevant MCAD variants.
- Correlate protein amount and FAD cofactor content with enzymatic activity in specific protein bands.
- Gain insights into how pathogenic variants affect protein conformation and function, which would be impossible with denaturing SDS-PAGE [21].
Detailed Experimental Protocols
To ensure reproducibility and high-quality results, follow these core methodologies.
Protocol 1: Standard Denaturing SDS-PAGE
This is the widely used Laemmli method for separating proteins by molecular weight [18].
Sample Preparation:
- Mix protein sample with SDS-containing sample buffer (e.g., Laemmli buffer). A common 4X buffer contains 250 mM Tris-HCl (pH 6.8), 8% SDS, 40% glycerol, 0.02% Bromophenol Blue [18].
- Add a reducing agent like 100 mM DTT or 5% β-mercaptoethanol to break disulfide bonds [18].
- Heat the samples at 70-100°C for 5-10 minutes to fully denature the proteins [3].
Gel Preparation and Electrophoresis:
- Cast a polyacrylamide gel with a stacking gel (lower % acrylamide, pH ~6.8) and a resolving gel (higher % acrylamide, pH ~8.8) for optimal resolution [3].
- Use a running buffer containing Tris, glycine (or MOPS), and 0.1% SDS (e.g., 25 mM Tris, 192 mM glycine, 0.1% SDS) [7].
- Load samples and molecular weight markers. Run at constant voltage (e.g., 100-200V) until the dye front reaches the bottom [7].
Protocol 2: Standard Native-PAGE
This protocol preserves protein structure and function.
Sample Preparation:
Gel Preparation and Electrophoresis:
- Cast a polyacrylamide gel without SDS. The same stacking/resolving gel system can be used, but with native buffers.
- Use a running buffer without SDS (e.g., Tris-glycine or Tris-Bicine at pH 8.3-8.8) [3].
- Load samples and run under constant voltage, typically at 4°C to further stabilize protein activity [3].
The Scientist's Toolkit: Essential Research Reagents
Successful execution of these electrophoretic techniques relies on specific reagents. The table below details key solutions and their functions.
Table 3: Key Reagent Solutions for Protein Electrophoresis
| Reagent Solution | Function | Critical Notes |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers uniform negative charge [3] [22]. | Core component of SDS-PAGE; omitted in native-PAGE. |
| Reducing Agents (DTT, β-mercaptoethanol) | Cleaves disulfide bonds between cysteine residues, fully unfolding proteins [18] [20]. | Used in reducing SDS-PAGE; omitted in non-reducing SDS-PAGE and native-PAGE. |
| Polyacrylamide Gel Matrix | Cross-linked polymer that acts as a molecular sieve; pore size determines resolution range [3]. | Acrylamide concentration is varied to separate different molecular weight ranges. |
| Tris-based Running Buffers | Conducts current and maintains stable pH during electrophoresis [18] [3]. | Common systems include Tris-glycine (SDS-PAGE) and Tris-Bicine (native-PAGE). |
| Coomassie Blue G-250 | Used in Blue Native (BN)-PAGE to impart negative charge to native protein complexes [7] [23]. | Allows migration of intact complexes without denaturation. |
| Native Sample Buffer (without SDS) | Maintains protein solubility and native conformation during loading and electrophoresis [7]. | Typically contains glycerol, a mild buffer, and a colored tracking dye. |
Emerging Techniques and Hybrid Approaches
Innovative methods are bridging the gap between high resolution and structural preservation.
- Native SDS-PAGE (NSDS-PAGE): A modified technique that uses minimal SDS (e.g., 0.0375%) in the running buffer and omits SDS and heating from the sample buffer. This approach retains enzymatic activity and metal cofactors in many proteins (e.g., Zn²⁺ retention increased from 26% to 98%) while maintaining high-resolution separation comparable to standard SDS-PAGE [7].
- Two-Dimensional BN/SDS-PAGE: This powerful hybrid method separates intact protein complexes in the first dimension by Blue Native (BN)-PAGE, then excises a lane of the gel, denatures it, and runs it in the second dimension by SDS-PAGE. This identifies the individual subunit composition of each native complex, providing deep insights into protein-protein interactions and complex assembly [23].
SDS-PAGE and native-PAGE are not interchangeable but complementary tools in the protein scientist's arsenal. The decision to use one over the other must be driven by the specific research question.
- For determining subunit molecular weight, assessing sample purity, or performing immunoblotting, SDS-PAGE is the robust and standardized method of choice.
- For investigating native oligomeric state, protein-protein interactions, or enzymatic function, native-PAGE is the indispensable technique.
Emerging methods like NSDS-PAGE and 2D BN/SDS-PAGE offer powerful ways to gain more comprehensive information. By understanding the fundamental impact each method has on protein structure, researchers can design experiments that yield the most accurate and biologically relevant results, ultimately accelerating progress in drug development and basic research.
Protocols and Practical Applications: When to Use Each Technique
In protein analysis research, selecting the appropriate electrophoretic technique is fundamental to obtaining meaningful results. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Native PAGE serve distinct purposes and provide complementary information about protein systems. SDS-PAGE, developed by Ulrich K. Laemmli in 1970, denatures proteins to separate them primarily by molecular weight [18] [6]. In contrast, Native PAGE maintains proteins in their native, folded state, preserving their biological activity and enabling separation based on size, charge, and shape [9] [6]. This comparison guide objectively evaluates both techniques' performance, providing researchers with the experimental data necessary to select the optimal approach for their specific applications in drug development and basic research.
The fundamental distinction lies in their treatment of protein structure. SDS-PAGE utilizes the anionic detergent SDS to linearize proteins and impart a uniform negative charge, effectively masking intrinsic charge differences and ensuring separation correlates with molecular weight alone [9] [24]. This process destroys functional properties, including enzymatic activity and non-covalently bound cofactors [7]. Native PAGE, including variants like Blue Native (BN)-PAGE and Clear Native (CN)-PAGE, avoids denaturants, allowing separated proteins to be recovered in their functional form for downstream activity assays or interaction studies [25] [9] [26]. The choice between these methods ultimately depends on the research question: whether the goal is to analyze protein size and purity (SDS-PAGE) or to investigate structure-function relationships and protein complexes (Native PAGE) [27] [6].
Principles and Comparative Analysis
Core Principles and Technical Specifications
The following table summarizes the fundamental differences between SDS-PAGE and Native PAGE:
Table 1: Core Principles and Technical Specifications of SDS-PAGE vs. Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight only [9] [6] | Size, overall charge, and shape [9] [6] |
| Gel Type | Denaturing [6] | Non-denaturing [6] |
| Key Reagents | SDS, reducing agents (DTT/BME) [28] [6] | Coomassie dye (BN-PAGE); no denaturants [25] [6] |
| Sample Preparation | Heated (95-100°C, 5 min) in SDS-containing buffer [28] [24] | Not heated; kept on ice to preserve native state [25] [6] |
| Protein State | Denatured and linearized [9] [24] | Native, folded conformation [9] [29] |
| Net Protein Charge | Uniformly negative [9] [6] | Dependent on native charge and buffer pH [29] [6] |
| Protein Function Post-Separation | Lost [7] [6] | Retained [7] [6] |
| Primary Applications | Molecular weight determination, purity check, western blotting [9] [24] | Studying oligomerization, protein-protein interactions, enzymatic activity [25] [9] |
Performance Comparison and Experimental Data
Quantitative comparisons highlight the practical trade-offs between these techniques. A key study analyzing human bronchial smooth muscle cells (HBSMC) found that 1D SDS-PAGE-MS enabled the assignment of 2,552 proteins from the supernatant fraction, while nondenaturing 2DE-MS assigned 4,323 proteins from the same fraction, suggesting higher sensitivity for the native method in this context [27]. However, SDS-PAGE was crucial for analyzing the insoluble precipitate fraction, which was inaccessible to nondenaturing 2DE, and was advantageous for comparative quantitation [27].
Research into metalloproteins demonstrates the functional consequences of each method. When analyzing Zn²⁺-proteins, standard SDS-PAGE retained only 26% of bound Zn²⁺, whereas a modified Native SDS-PAGE (NSDS-PAGE) protocol retained 98% [7]. In-gel activity assays showed that seven out of nine model enzymes, including four Zn²⁺ proteins, retained activity after NSDS-PAGE, and all nine were active after BN-PAGE, whereas all were denatured during standard SDS-PAGE [7]. This data underscores Native PAGE's superiority for functional studies.
Table 2: Experimental Performance Data: SDS-PAGE vs. Native PAGE
| Performance Metric | SDS-PAGE | Native PAGE | Experimental Context |
|---|---|---|---|
| Proteins Assigned | 2,552 proteins [27] | 4,323 proteins (2DE) [27] | Analysis of HBSMC supernatant fraction [27] |
| Metal Ion Retention | 26% Zn²⁺ retention [7] | 98% Zn²⁺ retention (NSDS-PAGE) [7] | Analysis of Zn²⁺-proteome and model proteins [7] |
| Enzymatic Activity Retention | 0 out of 9 model enzymes [7] | 7 out of 9 (NSDS-PAGE); 9 out of 9 (BN-PAGE) [7] | In-gel activity assay of nine model enzymes [7] |
| Analysis of Insoluble Fractions | Effective [27] | Not applicable/ineffective [27] | Analysis of HBSMC precipitate fraction [27] |
| Comparative Quantitation | Advantageous [27] | Less advantageous [27] | Comparison of protein quantity differences [27] |
| Information on Protein Interactions | Limited [27] | Advantageous [27] | Analysis of protein complexes in HBSMC [27] |
Step-by-Step Experimental Protocols
SDS-PAGE Protocol
Principle: SDS binds to proteins at a constant ratio, linearizing them and imparting a uniform negative charge. When an electric field is applied, proteins migrate through the polyacrylamide gel matrix toward the anode, separated based on molecular weight [24].
- Research Reagent Solutions:
- 30% Acrylamide/Bis Solution (37.5:1): Forms the gel matrix.
- Resolving Gel Buffer (1.5 M Tris-HCl, pH 8.8): Provides the pH for separation.
- Stacking Gel Buffer (0.5 M Tris-HCl, pH 6.8): Creates a low-pH environment to stack proteins.
- 10% Sodium Dodecyl Sulfate (SDS): Denaturing agent that binds and charges proteins.
- 10% Ammonium Persulfate (APS): Initiates gel polymerization.
- TEMED (N,N,N',N'-Tetramethylethylenediamine): Catalyst for gel polymerization.
- 5X SDS-PAGE Loading Buffer: Contains SDS, glycerol, bromophenol blue, and a reducing agent (e.g., DTT or β-mercaptoethanol).
- Running Buffer (25 mM Tris, 192 mM Glycine, 0.1% SDS, pH ~8.3): Conducts current and maintains pH during electrophoresis.
- Coomassie Brilliant Blue Staining Solution: Visualizes separated proteins.
- Destaining Solution (e.g., Methanol/Acetic Acid/Water): Removes background stain.
Gel Preparation:
- Resolving Gel: Combine acrylamide/bis-acrylamide, Tris-HCl (pH 8.8), SDS, and water. Degas briefly. Add APS and TEMED to initiate polymerization, then pour the solution between glass plates. Overlay with isopropanol or water to create a flat surface. Allow to polymerize for 30-60 minutes.
- Stacking Gel: After removing the overlay, prepare and pour the stacking gel mixture (acrylamide, Tris-HCl pH 6.8, SDS, APS, TEMED) on top of the polymerized resolving gel. Insert a comb and allow to polymerize for 30-45 minutes.
Sample Preparation: Mix protein samples with loading buffer (e.g., 4:1 ratio). Heat the samples at 95-100°C for 5 minutes to denature proteins. Centrifuge briefly at 12,000 × g to collect condensation [28] [24].
Electrophoresis: Assemble the gel in the electrophoresis tank filled with running buffer. Load prepared samples and a molecular weight marker into the wells. Run at constant voltage: 70-100 V until the dye front enters the resolving gel, then increase to 100-150 V until the dye front reaches the bottom (typically 1-2 hours total) [28].
Staining and Visualization: Carefully disassemble the apparatus and transfer the gel to a container. Submerge in Coomassie Brilliant Blue stain and incubate with gentle shaking for 15 minutes to several hours. Replace the stain with destaining solution and shake until protein bands are clear against a transparent background. Rinse with water and document the gel [24].
Native PAGE (BN-PAGE) Protocol
Principle: Proteins are kept in their native state using mild detergents and no heat. Coomassie dye binds superficially to impart a negative charge proportional to mass. Separation occurs based on the protein's native mass, charge, and shape, preserving complexes [25].
- Research Reagent Solutions:
- Aminocaproic Acid Buffer (0.75 M, 50 mM Bis-Tris, pH 7.0): Provides ionic strength and pH control.
- Mild Detergent (e.g., 10% n-Dodecyl β-D-maltoside): Solubilizes membrane protein complexes without denaturing.
- Protease Inhibitors (PMSF, Leupeptin, Pepstatin): Prevent protein degradation.
- Coomassie Blue G Dye (5% in 0.5 M aminocaproic acid): Imparts charge for electrophoresis.
- Cathode Buffer (50 mM Tricine, 15 mM Bis-Tris, 0.02% Coomassie G, pH 7.0): Upper chamber buffer.
- Anode Buffer (50 mM Bis-Tris, pH 7.0): Lower chamber buffer.
- Gradient Gel Solutions (e.g., 6% and 13% Acrylamide): For forming a linear gradient gel for superior resolution of complexes.
Sample Preparation: Resuspend sedimented mitochondria (e.g., 0.4 mg) in aminocaproic acid buffer. Add detergent (e.g., 7.5 μL of 10% dodecyl maltoside) and incubate on ice for 30 minutes. Centrifuge at high speed (e.g., 72,000 × g, 30 minutes) to remove insoluble material. Collect the supernatant and add Coomassie blue G dye and protease inhibitors.
Native Gel Preparation: Prepare a linear gradient acrylamide gel (e.g., 6-13%) using a gradient former. The gel solutions contain aminocaproic acid and Bis-Tris (pH 7.0). Pour the gel, overlay with isopropanol, and allow to polymerize. Prepare and add a stacking gel, then insert the comb.
Electrophoresis: Load the prepared samples into wells. Run the gel at a constant voltage of 150 V for approximately 2 hours at 4°C until the dye front nearly runs off the bottom.
Downstream Analysis:
- First-Dimension Analysis: Complexes can be immediately analyzed by western blotting using a PVDF membrane [25].
- Second-Dimension Analysis (BN/SDS-PAGE): For higher resolution, excise a lane from the first-dimension BN-PAGE gel, soak it in SDS denaturing buffer, and then place it horizontally on top of an SDS-PAGE gel. This resolves the individual subunits of each complex [25].
Research Reagent Solutions
The following table details key reagents essential for successfully performing SDS-PAGE and Native PAGE.
Table 3: Research Reagent Solutions for Protein Electrophoresis
| Reagent | Function | Key Considerations |
|---|---|---|
| Acrylamide/Bis-Acrylamide | Forms the porous gel matrix for molecular sieving [28] [29] | Concentration determines pore size (e.g., 8-15% for SDS-PAGE; gradients for BN-PAGE). Neurotoxin in monomer form [29]. |
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins and confers uniform negative charge [9] [24] | Critical for SDS-PAGE; omitted in Native PAGE. Purity is essential for consistent results. |
| Tris-HCl Buffers | Maintains pH during gel polymerization and electrophoresis [28] [29] | Different pH for stacking (pH 6.8) and resolving (pH 8.8) gels in SDS-PAGE [28]. |
| Ammonium Persulfate (APS) & TEMED | Catalyzes acrylamide polymerization [28] [29] | TEMED is highly corrosive. Fresh APS solution is required for efficient polymerization. |
| Coomassie Dye | Protein stain (G-250 for BN-PAGE charge; R-250 for staining) [25] [24] | G-250 in BN-PAGE buffer binds proteins without denaturing [25]. R-250 is common for post-electrophoresis staining [24]. |
| Dithiothreitol (DTT) / β-Mercaptoethanol | Reducing agents that break disulfide bonds [28] [18] | Used in reducing SDS-PAGE. Omitted in non-reducing SDS-PAGE and Native PAGE. |
| Mild Detergents (e.g., Dodecyl Maltoside) | Solubilizes membrane proteins while preserving complexes [25] | Critical for BN-PAGE of mitochondrial and membrane protein complexes. Harsher than those used in Native PAGE for soluble proteins. |
SDS-PAGE and Native PAGE are complementary pillars of protein analysis. SDS-PAGE is the unrivaled method for determining molecular weight, assessing purity, and preparing samples for western blotting, offering simplicity and robust performance for denatured proteins [9] [24]. In contrast, Native PAGE, particularly BN-PAGE, is indispensable for probing the native interactome, preserving enzymatic function, and analyzing the oligomeric state of protein complexes, albeit with greater technical complexity [25] [7] [27].
The choice between these techniques is not a matter of superiority but of strategic alignment with research objectives. For drug development professionals, this distinction is critical: SDS-PAGE is ideal for validating the expression and size of a recombinant protein therapeutic, while Native PAGE is essential for confirming the correct assembly of a multi-subunit complex or for isolating active enzymes for functional screening. By understanding their principles, performance trade-offs, and detailed protocols, researchers can leverage these powerful tools to advance our understanding of protein structure and function.
In protein analysis research, the choice between denaturing and native electrophoretic techniques represents a fundamental crossroads with significant implications for experimental outcomes. While SDS-PAGE has become the default method for determining protein purity and molecular weight, it systematically destroys the very structural features and functional properties that define biological activity. Native PAGE (Polyacrylamide Gel Electrophoresis) addresses this limitation by maintaining proteins in their folded, active state during separation, enabling researchers to study enzymatic activity, protein-protein interactions, and quaternary structures that are invisible to denaturing methods. This comparison guide objectively examines the performance of native PAGE against SDS-PAGE alternatives, providing supporting experimental data to inform methodological selection for research and drug development applications.
Fundamental Principles: How Native PAGE Preserves Protein Function
Core Mechanism of Native PAGE
Unlike SDS-PAGE, which uses sodium dodecyl sulfate to denature proteins and mask their intrinsic charge, native PAGE separates proteins based on their net charge, size, and shape in their native conformation [30] [9]. Without denaturing agents, proteins retain their secondary, tertiary, and quaternary structures, allowing multimeric complexes to remain intact during electrophoresis [9]. This preservation of structural integrity is what enables the retention of biological function post-separation.
The electrophoretic migration in native PAGE occurs because most proteins carry a net negative charge in alkaline running buffers, with higher charge density resulting in faster migration [30]. Simultaneously, the frictional force of the gel matrix creates a sieving effect that regulates movement according to protein size and three-dimensional shape [30]. This dual separation mechanism means that small proteins with high charge density migrate fastest, while larger complexes with lower charge density migrate more slowly.
Variants of Native Electrophoresis
Blue Native PAGE (BN-PAGE): Utilizes Coomassie Blue G-250 dye, which binds to hydrophobic protein surfaces and imposes a negative charge shift, forcing even basic proteins to migrate toward the anode while preventing aggregation [31] [25]. This method is particularly valuable for studying membrane protein complexes and oxidative phosphorylation systems [32] [31].
Clear Native PAGE (CN-PAGE): Replaces Coomassie dye with mixtures of anionic and neutral detergents in the cathode buffer to induce charge shifts while avoiding blue dye interference during downstream in-gel enzyme activity staining [21] [31].
High-Resolution CN-PAGE: An advanced variant that provides superior resolution for detecting enzymatic activities while offering qualitative insights into structural diversity [21].
Comparative Performance Analysis: Native PAGE vs. SDS-PAGE
Quantitative Experimental Data
Table 1: Direct Performance Comparison Between Electrophoresis Methods
| Performance Parameter | SDS-PAGE | BN-PAGE | NSDS-PAGE | Experimental Context |
|---|---|---|---|---|
| Enzyme Activity Retention | 0% (0/9 model enzymes) | 100% (9/9 model enzymes) | 77.8% (7/9 model enzymes) | Model enzyme study with Zn-proteins [7] |
| Metal Cofactor Retention | 26% | Not specified | 98% | Zn²⁺ retention in proteomic samples [7] |
| Resolution Capability | High | Moderate | High | Proteomic separation quality [7] |
| Structural Information | Primary structure only | Quaternary structure preserved | Native conformation maintained | Protein complex analysis [9] |
| Typical Run Time | 20-45 minutes [30] | 90-120 minutes [25] | Approximately 45 minutes [7] | Mini-gel format conditions |
Functional Advantages of Native PAGE
The preserved enzymatic activity following native electrophoresis enables direct in-gel activity assays that provide biological insights impossible to obtain with denaturing methods. In a recent study investigating medium-chain acyl-CoA dehydrogenase (MCAD) deficiency, researchers adapted a high-resolution clear native gel colorimetric assay that quantified the activity of MCAD tetramers separately from other protein forms [21]. This approach revealed novel insights into how pathogenic variants affect MCAD structure and function, distinguishing subtle differences in protein shape, enzymatic activity, and FAD content that would be undetectable using standard SDS-PAGE methodology [21].
The ability to detect these functional differences is particularly valuable for understanding the molecular basis of metabolic disorders like MCAD deficiency, where variants may either impair enzymatic activity directly or destabilize interactions between subunits, leading to protein aggregation [21]. Standard enzymatic assays measure overall activity but cannot differentiate between tetramers and other protein forms, information that is critical for understanding the impact of pathogenic variants on structure destabilization [21].
Detailed Native PAGE Protocol for Functional Analysis
Sample Preparation
Mitochondrial Isolation: For membrane protein complexes like oxidative phosphorylation systems, isolate mitochondria from cells before analysis. While whole tissue or cell extracts can be used, mitochondrial isolation typically yields stronger signals with less background interference [25].
Solubilization: Resuspend 0.4 mg of sedimented mitochondria in 40 μL of buffer containing 0.75 M aminocaproic acid and 50 mM Bis-Tris (pH 7.0) [25]. Add 7.5 μL of 10% n-dodecyl-β-D-maltopyranoside, mix, and incubate for 30 minutes on ice [25].
Clarification: Centrifuge at 72,000 × g for 30 minutes, though a bench-top microcentrifuge at approximately 16,000 × g may suffice for smaller volumes [25].
Sample Buffer Preparation: Collect supernatant and add 2.5 μL of 5% Coomassie Blue G solution in 0.5 M aminocaproic acid, along with protease inhibitors (1 mM PMSF, 1 μg/mL leupeptin, and 1 μg/mL pepstatin) [25].
Gel Casting and Electrophoresis
Table 2: Research Reagent Solutions for Native PAGE
| Reagent | Function | Example Formulation | Critical Notes |
|---|---|---|---|
| n-dodecyl-β-D-maltopyranoside | Mild nonionic detergent for membrane protein solubilization | 10% solution in appropriate buffer | Preserves protein-protein interactions; critical for complex integrity [25] |
| Coomassie Blue G-250 | Charge conferral dye for BN-PAGE | 5% solution in 0.5 M aminocaproic acid | Imparts negative charge proportional to mass; prevents aggregation [31] [25] |
| 6-Aminocaproic Acid | Zwitterionic salt | 0.75 M, 50 mM Bis-Tris/HCl, pH 7.0 | Supports extraction without affecting electrophoresis; zero net charge at pH 7.0 [31] [25] |
| Bis-Tris System | Buffering system | 50 mM Bis-Tris, pH 7.0 | Maintains appropriate pH throughout electrophoresis [25] |
| Protease Inhibitors | Prevents protein degradation | 1 mM PMSF, 1 μg/mL leupeptin and pepstatin | Essential for preserving native structures during processing [25] |
Gel Preparation: While single-concentration gels can be used (e.g., straight 10% acrylamide), linear gradient gels (e.g., 6-13% acrylamide) typically provide superior separation across a broad molecular weight range [25]. The gel recipe for a 13% acrylamide solution includes 14 mL 30% acrylamide, 0.2 mL dd water, 16 mL 1 M aminocaproic acid (pH 7.0), 1.6 mL 1 M Bis-Tris (pH 7.0), 200 μL 10% APS, and 20 μL TEMED [25].
Electrophoresis Conditions: Load 5-20 μL samples into wells and run at 150 V for approximately 2 hours or until the sample buffer blue dye has almost run off the bottom of the gel [25]. Use anode buffer (50 mM Bis-Tris, pH 7.0) and cathode buffer (50 mM Tricine, 15 mM Bis-Tris, 0.02% Coomassie Blue G, pH 7.0) [25].
Downstream Applications
In-Gel Activity Assays: Following electrophoresis, incubate the gel in appropriate reaction mixtures to detect enzymatic activity. For example, MCAD activity can be determined using a solution containing the physiological substrate octanoyl-CoA and nitro blue tetrazolium chloride (NBT), which forms an insoluble purple diformazan precipitate upon reduction [21]. This staining typically becomes visible after 10-15 minutes of incubation [21].
Two-Dimensional Analysis: For detailed subunit composition analysis, excise lanes from the first-dimension native gel and soak in SDS denaturing buffer before loading onto an SDS-PAGE gel for second-dimension separation [25].
Experimental Workflow Visualization
Advanced Applications and Research Implications
Metabolic Network Analysis
Blue Native PAGE has become instrumental in gaining insights into structure/function relationships of complex metabolic systems, particularly the mitochondrial oxidative phosphorylation (OXPHOS) system [32] [31]. The technique enables researchers to study: (1) the assembly pathways of protein complexes, (2) the composition of higher-order respiratory chain supercomplexes (respirasomes), and (3) pathologic mechanisms in patients with monogenetic OXPHOS disorders [31]. This application demonstrates how native electrophoresis facilitates the exploration of enzymatic activity in metabolic networks via in-gel assays that are quick, specific, and amenable to further studies [32].
Disease Mechanism Investigation
The functional proteomic capabilities of native PAGE make it particularly valuable for investigating molecular disease mechanisms. In MCAD deficiency research, the adaptation of high-resolution clear native electrophoresis enabled quantification of tetramer-specific activity separate from other protein forms, providing novel insights into how pathogenic variants affect protein structure and function [21]. This approach revealed that some MCAD variants induce conformational changes that alter electrophoretic mobility without affecting monomeric molecular mass, as confirmed by parallel SDS-PAGE analysis [21]. Such structural insights are inaccessible through denaturing electrophoretic methods.
Drug Discovery Applications
Native electrophoresis techniques provide valuable tools for drug discovery, particularly in studying protein-ligand interactions and compound binding. Semi-native PAGE has been successfully applied as a rapid screening method for studying metal complex-protein interactions, enabling high-throughput screening and optimization of complex binding without relying on spectral changes of the metal complex upon protein interaction [33]. This application demonstrates how native electrophoretic methods can accelerate pharmaceutical development by providing functional binding data that complements structural information.
The comparative data presented in this guide demonstrates that Native PAGE provides unique capabilities for functional protein analysis that complement the structural information obtained from SDS-PAGE. While SDS-PAGE remains the superior method for determining molecular weight and assessing protein purity, Native PAGE enables researchers to investigate enzymatic activity, protein-protein interactions, and quaternary structures that are fundamental to biological function. The choice between these techniques should be guided by research objectives: SDS-PAGE for structural characterization and Native PAGE for functional studies. For comprehensive protein analysis, many researchers employ both methods sequentially, using two-dimensional BN/SDS-PAGE to correlate functional complexes with their subunit composition. This integrated approach provides the most complete understanding of protein structure-function relationships, making it invaluable for basic research, drug discovery, and diagnostic applications.
In the realm of protein analysis, polyacrylamide gel electrophoresis (PAGE) is a foundational technique, with SDS-PAGE and Native PAGE serving as two pivotal, yet distinct, methodological branches. The choice between them is dictated by the research objective: SDS-PAGE is the benchmark for determining molecular weight and assessing sample purity, while Native PAGE is indispensable for probing protein function, oligomeric state, and native structure [6] [9] [3]. This guide provides a detailed, objective comparison of their performance, focusing on the core applications of SDS-PAGE to equip researchers in making an informed methodological selection.
Principles and Mechanisms: A Tale of Two Techniques
The fundamental divergence between SDS-PAGE and Native PAGE lies in their treatment of protein structure, which directly dictates their applications.
SDS-PAGE employs the anionic detergent sodium dodecyl sulfate (SDS) and heat to denature proteins. SDS binds uniformly to the polypeptide backbone, masking the protein's intrinsic charge and conferring a uniform negative charge density. Reducing agents like DTT or β-mercaptoethanol are often added to break disulfide bonds. This process results in fully denatured, linear proteins whose migration through the polyacrylamide gel matrix is determined primarily by molecular weight alone [6] [3] [15]. Smaller proteins migrate faster, while larger ones are retarded.
Native PAGE, in contrast, is run under non-denaturing conditions without SDS or reducing agents. Consequently, proteins retain their native conformation, higher-order structure (including quaternary assemblies), and biological activity. Their migration depends on a combination of the protein's inherent charge, size, and three-dimensional shape [6] [9] [15].
The workflow diagram below illustrates the distinct procedural steps and outcomes of each method.
Performance Comparison: Application-Based Analysis
The following table summarizes the performance characteristics of SDS-PAGE and Native PAGE across key application areas, helping researchers select the optimal technique.
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Principle | Molecular weight only [6] [3] [15] | Size, net charge, and 3D shape [6] [9] [3] |
| Protein State | Denatured and linearized [6] [3] | Native, folded, and functional [6] [9] [3] |
| Key Applications | Molecular weight determination, purity checks, Western blotting [6] [9] [3] | Studying oligomeric state, protein complexes, enzymatic activity [6] [9] [3] |
| Data on Purity Checks | Excellent for detecting contaminating polypeptides; single band suggests purity [9] [3] | Can separate complexed from uncomplexed protein, but charge variants complicate purity assessment [9] |
| Data on Molecular Weight | High accuracy; comparison to standard curve provides MW estimate [3] | Low accuracy; inferred MW can be unreliable due to charge/shape influence [9] [10] |
| Functional Analysis | Not possible; proteins are inactivated [6] [9] | Possible; proteins can be recovered for activity assays [6] [9] [3] |
| Typical Running Temperature | Room Temperature [6] | 4°C (to preserve native structure) [6] |
Experimental Protocols for Core SDS-PAGE Applications
Molecular Weight Determination
Objective: To estimate the molecular weight of an unknown protein by comparing its electrophoretic mobility to a calibrated protein ladder.
Detailed Methodology:
- Sample Preparation: Mix the protein sample with an SDS-containing loading buffer (e.g., Laemmli buffer). A standard protein ladder with known molecular weights must be loaded onto the same gel. Heat the samples at 70-100°C for 5-10 minutes to ensure complete denaturation [3].
- Gel Electrophoresis: Load samples onto a polyacrylamide gel (e.g., 4-20% gradient for broad range separation). Apply a constant voltage (~120-150V) until the dye front migrates to the bottom of the gel [3].
- Staining & Analysis: Stain the gel with Coomassie Blue or a fluorescent stain to visualize protein bands. Measure the migration distance of each protein in the ladder and the unknown sample(s). Plot the log(MW) of the standard proteins against their migration distance (Rf value) to generate a standard curve. The molecular weight of the unknown protein is interpolated from this curve [3].
Purity Checks
Objective: To assess the homogeneity of a protein preparation, typically after a purification step.
Detailed Methodology:
- Sample Preparation: Prepare the purified protein sample as described in 3.1.
- Gel Electrophoresis: Run the purified sample alongside a control (e.g., crude lysate) on an SDS-PAGE gel.
- Analysis: A single, tight band observed for the purified sample suggests a high degree of purity. The presence of multiple bands indicates contaminating proteins. The intensity of the bands, which correlates with protein abundance, can be used for semi-quantitative analysis of the major component versus contaminants [9] [3].
Western Blotting
Objective: To specifically detect a protein of interest using antibodies after SDS-PAGE separation.
Detailed Methodology:
- Separation and Transfer: Proteins are first separated by SDS-PAGE. The gel is then placed against a membrane (e.g., PVDF or nitrocellulose). An electric current is applied to electrophoretically transfer the proteins from the gel onto the membrane [3].
- Immunoblotting: The membrane is blocked with a protein solution (e.g., BSA or non-fat milk) to prevent non-specific antibody binding. It is then incubated with a primary antibody specific to the target protein, followed by a secondary antibody conjugated to an enzyme (e.g., Horseradish Peroxidase - HRP) that recognizes the primary antibody [3].
- Detection: A chemiluminescent substrate is added to the membrane. The enzyme conjugated to the secondary antibody catalyzes a light-emitting reaction upon contact with its substrate. The signal is captured using X-ray film or a digital imaging system, revealing the specific band corresponding to the protein of interest [3].
Case Study: Interpreting Conflicting Results
A classic example that underscores the importance of technique selection involves a protein that migrates at 60 kDa on a non-reducing SDS-PAGE but at 120 kDa on Native PAGE [10].
- Interpretation: The 60 kDa band in SDS-PAGE indicates the mass of the individual polypeptide chain. The 120 kDa band in Native PAGE reveals that in its native state, the protein exists as a dimer. The fact that this occurs even in non-reducing SDS-PAGE indicates that the subunits are held together by non-covalent interactions (e.g., hydrophobic forces, hydrogen bonds) and not by disulfide bridges, which would have been stable under these conditions [10].
- Takeaway: Relying solely on SDS-PAGE would have mischaracterized the protein's native quaternary structure. This highlights Native PAGE's unique utility in studying protein-protein interactions and oligomerization.
The Scientist's Toolkit: Essential Reagents and Materials
Successful execution of SDS-PAGE relies on a suite of specialized reagents and equipment. The table below details key solutions and their critical functions.
| Research Reagent / Material | Function in SDS-PAGE Protocol |
|---|---|
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers a uniform negative charge [6] [3]. |
| Acrylamide/Bis-acrylamide | Monomers that polymerize to form the porous gel matrix, which sieves proteins by size [3]. |
| Tris-based Buffers | Provides the ionic environment and maintains stable pH during electrophoresis (e.g., Tris-Glycine) [34] [3]. |
| Reducing Agents (DTT, BME) | Cleaves disulfide bonds to ensure complete protein unfolding and subunit dissociation [6] [3]. |
| Protein Molecular Weight Markers | A mixture of proteins of known sizes used to create a standard curve for molecular weight estimation [3]. |
| Ammonium Persulfate (APS) & TEMED | Catalysts that initiate and accelerate the polymerization of acrylamide to form the gel [3]. |
| Coomassie Brilliant Blue Stain | A dye that binds to proteins, allowing for visualization of separated bands post-electrophoresis [3] [35]. |
SDS-PAGE and Native PAGE are not competing techniques but rather complementary tools in the protein scientist's arsenal. SDS-PAGE is the unequivocal method of choice for determining molecular weight, assessing purity, and preparing samples for Western blotting, offering simplicity, robustness, and high resolution based primarily on polypeptide size. In contrast, Native PAGE is specialized for the functional analysis of proteins in their native state, preserving complexes and activity.
The experimental data and protocols outlined herein demonstrate that the selection between these methods should be a deliberate one, grounded in the specific research question. For routine analytical tasks in characterization and validation, SDS-PAGE remains an indispensable and powerful workhorse in research and drug development.
In the field of protein research, the choice of electrophoretic technique is fundamental to the biological questions a scientist can address. While SDS-PAGE (sodium dodecyl sulfate-polyacrylamide gel electrophoresis) serves as the ubiquitous workhorse for determining protein molecular weight and purity under denaturing conditions, Native PAGE (polyacrylamide gel electrophoresis) provides a complementary approach that preserves proteins in their natural, functional state [9] [3]. This distinction is crucial for researchers investigating higher-order protein structures and functions. Where SDS-PAGE denatures proteins into uniform linear chains, Native PAGE maintains the intricate three-dimensional architecture of proteins, allowing for the study of protein complexes, oligomeric states, and enzymatic activity directly within the gel matrix [9] [36]. This guide objectively compares the performance of Native PAGE against alternative methods, with a focus on its specialized applications, supported by experimental data and detailed protocols.
Core Principle: How Native PAGE Preserves Native Structure
The fundamental difference between Native PAGE and SDS-PAGE lies in sample preparation. In SDS-PAGE, proteins are heated in the presence of the anionic detergent SDS and a reducing agent, which denatures the protein, disrupts disulfide bonds, and confers a uniform negative charge. This process ensures separation is based almost exclusively on polypeptide chain length [9] [3] [37]. In contrast, Native PAGE omits these denaturing agents. Proteins are separated based on a combination of their intrinsic net charge, size, and shape as they migrate through the porous polyacrylamide gel in response to an electric field [3]. The higher the negative charge density and the smaller the size, the faster a protein will migrate. This preservation of native structure is the property that enables all subsequent functional analyses.
The following diagram illustrates the key procedural differences and resulting outcomes of each method.
Performance Comparison: Native PAGE vs. Alternative Techniques
The selection of an electrophoretic method involves trade-offs between resolution, structural preservation, and application suitability. The table below provides a comparative overview of Native PAGE against two other common techniques.
Table 1: Comparative Analysis of Protein Electrophoresis Techniques
| Feature | Native PAGE | SDS-PAGE | Blue Native (BN)-PAGE |
|---|---|---|---|
| Separation Basis | Net charge, size, & shape [3] | Molecular weight [9] [3] | Mass & charge in native state [7] [38] |
| Protein State | Native, folded [9] | Denatured, linearized [9] | Native, but bound to Coomassie dye [7] [38] |
| Functional Activity | Retained (enzymes active) [7] [36] | Destroyed [9] [7] | Often retained, but dye may inhibit [36] [38] |
| Oligomeric State | Preserved [38] | Dissociated into subunits [9] | Preserved [38] |
| Resolution | Moderate [7] | High [9] [7] | Lower than SDS-PAGE [7] |
| Primary Application | Protein complexes, oligomerization, enzymatic assays [9] [36] | Molecular weight determination, purity checks [9] [18] | Membrane protein complexes, protein-protein interactions [36] [38] |
Advanced Hybrid Techniques
To leverage the strengths of multiple methods, researchers often employ hybrid techniques. A powerful example is 2-D Native-PAGE/SDS-PAGE, where protein complexes are first separated in a native gel, after which a single lane is excised and laid horizontally on a second gel for denaturing SDS-PAGE. This resolves the individual subunits of each oligomeric complex, allowing researchers to visualize their composition at a sensitivity of 2-3 picomoles [39]. Another innovation is Native SDS-PAGE (NSDS-PAGE), a modified method that uses minimal SDS and no heating or EDTA. This approach achieves high-resolution separation similar to traditional SDS-PAGE while remarkably retaining the functional activity of many enzymes and preserving up to 98% of bound metal ions in metalloproteins [7].
Key Applications & Experimental Data
Studying Oligomerization and Protein Complexes
Native PAGE is indispensable for analyzing the quaternary structure of proteins, such as the formation of dimers, trimers, and higher-order oligomers. This is critical for understanding the function of many proteins, as their active form is often multimeric.
Experimental Protocol: Analyzing HIV-1 Reverse Transcriptase Homodimers by Blue Native Agarose Gel Electrophoresis (BN-AGE) [38]
- Gel Preparation: Prepare a 3% horizontal agarose gel using Native Agarose Gel Buffer (NAGB: 25 mM Tris, 19.2 mM glycine, pH 8.5).
- Sample Preparation: Mix 10 µL of purified p51 protein with 2.5 µL of sample buffer (NAGB containing 30% glycerol) and 0.3 µL of 5% Coomassie blue G-250.
- Electrophoresis: Load samples and run the gel in NAGB at 40 V for 4.5 hours at room temperature.
- Detection: After electrophoresis, stain the gel with a protein gel staining solution (e.g., EZ-Run) for 1-2 hours, followed by destaining in water.
Supporting Data: This protocol successfully separated monomeric and dimeric forms of HIV-1 p51 protein. The study demonstrated that wild-type p51 and a W401A mutant formed dimers in the presence of the drug Efavirenz, while a L234A mutant, deficient in dimerization, did not [38]. This application highlights Native PAGE's utility in screening for protein-protein interactions and the effects of ligands or mutations on oligomerization.
In-Gel Enzymatic Activity Assays
A unique advantage of Native PAGE is the ability to detect enzyme activity directly within the gel, providing a direct link between a specific protein band and its biological function.
Experimental Protocol: Continuous Monitoring of Mitochondrial Complex IV Activity [36] [40]
- Sample Preparation & Electrophoresis: Separate mitochondrial protein complexes from heart tissue (e.g., pig or rabbit) using a commercial BN-PAGE system (e.g., NativePAGE Novex 4-16% Bis-Tris Gels) according to the manufacturer's specifications.
- In-Gel Assay Setup: Post-electrophoresis, place the gel in a custom reaction chamber with continuous circulation and filtration of the assay medium.
- Reaction Medium: For Complex IV (Cytochrome c Oxidase), the medium contains 3,3'-diaminobenzidine (DAB) and cytochrome c. Complex IV oxidizes cytochrome c, which in turn catalyzes the oxidation of DAB to form an insoluble brown precipitate at the site of activity.
- Data Acquisition & Analysis: Use time-lapse digital imaging to continuously capture images of the gel (e.g., every 20 seconds). Process the images to obtain kinetic traces of the precipitate formation, which reflects enzymatic activity.
Supporting Data: This sophisticated system allowed researchers to quantify the kinetics of Complex IV activity with high temporal resolution. The initial linear phase of the reaction was used to calculate catalytic rates, demonstrating that the in-gel assay reports on multiple catalytic turnovers and is sensitive to classic inhibitors like cyanide [36] [40]. This method provides topological and kinetic information that is lost in standard endpoint assays.
Visualization of an Oligomer's Subunits
Combining native and denaturing techniques offers a comprehensive view of complex composition.
Experimental Protocol: 2-D Native-PAGE/SDS-PAGE for IgG Subunit Analysis [39]
- First Dimension (Native-PAGE): Electrophorese IgG samples (e.g., 2-3 pmol) in a native acidic polyacrylamide gel.
- Staining: Stain the gel with Coomassie R-250 or G-250 to visualize the IgG oligomer bands.
- Gel Excision: Excise the protein bands of interest from the native gel.
- Second Dimension (SDS-PAGE): Place the excised gel strip horizontally onto a standard SDS-PAGE gel.
- Electrophoresis & Detection: Run the second-dimension gel to separate the subunits. Visualize the resulting heavy and light chains of IgG using a sensitive permanganate silver stain.
Supporting Data: This method provided a sensitive (2-3 pmol) and robust way to confirm the subunit composition of the IgG oligomer. The study systematically evaluated different staining combinations, finding Coomassie staining in the first dimension followed by silver staining in the second dimension to be optimal [39].
Table 2: Summary of Key Native PAGE Applications and Experimental Findings
| Application | Experimental Model | Key Finding / Outcome | Reference |
|---|---|---|---|
| Oligomerization Analysis | HIV-1 Reverse Transcriptase p51 | BN-AGE cleanly separated monomeric and dimeric p51, confirming drug-enhanced dimerization. | [38] |
| In-Gel Enzymatic Assay | Mitochondrial Complex IV | Kinetic traces from continuous monitoring confirmed catalytic activity and sensitivity to inhibitors. | [36] [40] |
| Metal Ion Retention | Zn²⁺-Metalloproteins (e.g., Alcohol Dehydrogenase) | NSDS-PAGE retained 98% of bound Zn²⁺, compared to 26% in standard SDS-PAGE. | [7] |
| Subunit Visualization | IgG Oligomer | 2-D PAGE successfully resolved heavy and light chains from the native oligomer. | [39] |
The Scientist's Toolkit: Essential Reagents and Materials
Successful Native PAGE experiments require specific reagents to maintain protein native state and ensure proper separation.
Table 3: Key Research Reagent Solutions for Native PAGE
| Reagent / Material | Function / Purpose | Example & Notes |
|---|---|---|
| Native Gel Matrix | Porous medium for sieving proteins based on size and shape. | Polyacrylamide (for most proteins) or Agarose (for very large complexes) [3] [38]. |
| Native Sample Buffer | Prepares sample without denaturation; often includes glycerol for loading. | Tris-glycine or Bis-Tris-based buffers at neutral or mildly alkaline pH, without SDS or reducing agents [3]. |
| Running Buffer | Conducts current and maintains pH during electrophoresis. | Tris-glycine or MOPS/Tris-based buffers [7] [3]. |
| Coomassie G-250 Dye | Imparts negative charge to proteins for migration (BN-PAGE). | Used in sample buffer at 0.02-0.05% for BN-PAGE; binds hydrophobic protein surfaces [7] [38]. |
| Activity Assay Reagents | Enable detection of specific enzymatic functions in-gel. | Substrate-specific (e.g., DAB for oxidase, ATP/lead salts for ATPase) [36] [40]. |
| Detergents | Solubilize membrane proteins while maintaining native state. | Dodecyl maltoside, digitonin [36] [38]. |
Native PAGE is not a replacement for SDS-PAGE but a specialized, powerful technique that answers different biological questions. Its unparalleled ability to preserve native protein structures enables critical investigations into oligomerization, protein-protein interactions, and enzymatic function that are impossible with denaturing methods. While SDS-PAGE remains the gold standard for determining molecular weight and assessing sample purity, Native PAGE, along with its advanced derivatives like BN-PAGE and 2D-PAGE, is essential for any researcher aiming to understand protein function in a state that closely mirrors its physiological reality. The choice between these techniques should be guided by the research objective: use SDS-PAGE to analyze polypeptide chains, and Native PAGE to understand functional proteins.
Blue Native PAGE (BN-PAGE) and Clear Native PAGE (CN-PAGE)
In the analysis of proteins, the choice of electrophoretic technique dictates the type of information researchers can obtain. While SDS-PAGE denatures proteins to separate them by molecular weight alone, native PAGE techniques preserve proteins in their functional, folded states, enabling the study of complexes, interactions, and activity [6]. Among these, Blue Native PAGE (BN-PAGE) and Clear Native PAGE (CN-PAGE) are two critical specialized methods. This guide provides an objective comparison of their performance, supported by experimental data, to inform their application in research and drug development.
Principle and Separation Mechanism
The core difference between these techniques lies in how they impart charge to proteins for electrophoretic separation, which directly impacts resolution, applicability, and downstream analysis.
BN-PAGE utilizes the anionic dye Coomassie Blue G-250, which binds nonspecifically to hydrophobic protein surfaces and basic amino acid residues [41] [13]. This binding imposes a strong negative charge shift, ensuring that all proteins—including basic membrane proteins—migrate toward the anode. The charge imposed is roughly proportional to the protein's size, allowing separation based on native molecular mass and providing high resolution [42] [41].
CN-PAGE is a milder technique that does not use charged dyes like Coomassie Blue. Instead, proteins separate based on their intrinsic negative charge at the gel's pH [42] [41]. This means a protein's migration depends on both its inherent charge and the gel's pore size. A significant limitation is that proteins with an isoelectric point (pI) higher than the gel pH (e.g., >7.5) may not enter the gel or may migrate toward the cathode and be lost [41].
High-Resolution CN-PAGE (hrCNE) is a variant that addresses some limitations of basic CN-PAGE. Instead of Coomassie dye, the cathode buffer contains mixed anionic micelles of neutral and anionic detergents. These micelles bind to proteins, inducing a charge shift that improves resolution and helps basic proteins enter the gel, making it a preferred "clear" method over CN-PAGE for many applications [41].
The workflow below illustrates the procedural differences and key outcomes of BN-PAGE and CN-PAGE.
Comparative Analysis: BN-PAGE vs. CN-PAGE
The choice between BN-PAGE and CN-PAGE involves trade-offs between resolution and the preservation of delicate protein functions. The following table summarizes the key operational and performance differences.
Table 1: Direct comparison of BN-PAGE and CN-PAGE characteristics and performance.
| Criteria | BN-PAGE | CN-PAGE |
|---|---|---|
| Charge Agent | Coomassie Blue G-250 dye [41] [25] | Protein's intrinsic charge; no dye [42] [41] |
| Resolution | High [42] [41] | Lower than BN-PAGE [42] [43] |
| Mass Estimation | Reliable, due to charge shift proportional to mass [42] [41] | Complicated, depends on intrinsic charge and size [42] |
| Protein Compatibility | All proteins, including basic membrane proteins [41] [13] | Limited to acidic proteins (pI < ~7.5) [41] |
| Gentleness | Standard harshness | Milder than BN-PAGE [42] [43] |
| Key Advantage | Robust, high-resolution standard analysis [42] [41] | Retains labile assemblies; ideal for post-electrophoresis activity assays [42] [44] |
| Primary Limitation | Coomassie dye can interfere with downstream assays [42] [7] | Lower resolution; not suitable for basic proteins [42] [41] |
Supporting Experimental Data
Empirical studies highlight the practical consequences of the theoretical differences between these methods, particularly regarding the analysis of protein complexes and their enzymatic function.
Preservation of Labile Complexes: A comparative study demonstrated that the combination of digitonin solubilization and CN-PAGE successfully retained labile supramolecular assemblies of mitochondrial membrane protein complexes, which were dissociated under standard BN-PAGE conditions. Furthermore, enzymatically active oligomeric states of mitochondrial ATP synthase that were not detected with BN-PAGE were identified using CN-PAGE [42].
Interference with Catalytic Activity: Research into metabolic networks has confirmed that BN-PAGE is highly effective for separating native complexes and analyzing protein interactions. However, the bound Coomassie dye can inhibit catalytic function. Consequently, for in-gel enzymatic activity assays, CN-PAGE (or hrCNE) is the preferred and often essential method [32].
Quantitative Complexome Profiling: CN-PAGE has been effectively coupled with mass spectrometry for quantitative complexome profiling. This approach successfully established abundance profiles of protein complexes along the molecular weight gradient in Arabidopsis thaliana, demonstrating the technique's utility for studying changes in complex abundance and composition in response to metabolic states [44].
Table 2: Summary of key experimental findings comparing BN-PAGE and CN-PAGE.
| Experimental Context | BN-PAGE Outcome | CN-PAGE Outcome |
|---|---|---|
| Mitochondrial ATP Synthase Analysis [42] | Dissociated labile supramolecular assemblies. | Identified previously undetected, enzymatically active oligomeric states. |
| In-gel Catalytic Activity Assays [42] [32] | Coomassie dye can interfere with or inhibit enzyme function. | Enabled determination of catalytic activities post-separation. |
| Membrane Protein Complex Stability [42] | Standard conditions may disrupt weak interactions. | Milder conditions preserved labile supramolecular assemblies. |
Detailed Methodologies
BN-PAGE Protocol
The following protocol for BN-PAGE is adapted from established methodologies [41] [25].
Sample Preparation:
- Isolate mitochondria or other cellular fractions. Resuspend 0.4 mg of mitochondrial pellet in 40 µL of buffer A (0.75 M 6-aminocaproic acid, 50 mM Bis-Tris/HCl, pH 7.0) with protease inhibitors [25].
- Solubilize by adding 7.5 µL of 10% n-dodecyl-β-D-maltopyranoside (DDM). Mix and incubate on ice for 30 minutes [25].
- Centrifuge at 72,000 x g for 30 minutes (or ~16,000 x g in a microcentrifuge) to remove insoluble material. Collect the supernatant [25].
- Add 2.5 µL of a 5% Coomassie Blue G-250 suspension in 0.5 M aminocaproic acid to the supernatant to achieve the required charge shift [25].
Gel Casting and Electrophoresis:
- Cast a linear acrylamide gradient gel (e.g., 4–16% or 3–12%) using a gradient mixer. A 6–13% gradient is often recommended for broad separation [25]. The gel system uses a Bis-Tris buffer at ~pH 7.5 [13].
- Load 5–20 µL of the prepared sample per well.
- Run the gel with specialized anode and cathode buffers. The cathode buffer contains 0.02% Coomassie Blue G-250 [25].
- Perform electrophoresis at 150 V for approximately 2 hours or until the dye front approaches the bottom of the gel [25].
CN-PAGE Protocol
The CN-PAGE protocol differs primarily in the omission of Coomassie dye and potential adjustments to solubilization conditions [42] [41].
Sample Preparation:
Gel Casting and Electrophoresis:
Research Reagent Solutions
Successful execution of native PAGE requires specific reagents tailored to preserve protein complexes.
Table 3: Essential reagents for BN-PAGE and CN-PAGE and their functions.
| Reagent / Equipment | Function | Example Use Case |
|---|---|---|
| Coomassie Blue G-250 | Imparts negative charge to proteins for BN-PAGE; prevents aggregation [41] [13]. | Added to sample and cathode buffer in BN-PAGE. |
| n-Dodecyl-β-D-Maltoside (DDM) | Non-ionic detergent for solubilizing membrane proteins [25]. | Solubilizing individual mitochondrial complexes from heart homogenates [41]. |
| Digitonin | Mild, non-ionic detergent for solubilizing membrane proteins. | Preserving labile supramolecular assemblies in CN-PAGE [42] [41]. |
| 6-Aminocaproic Acid | Provides ionic strength; acts as a protease inhibitor [25]. | Key component of sample and gel buffers. |
| Bis-Tris | Buffering agent for near-neutral pH (~7.5), crucial for native conditions [25] [13]. | Standard buffer for BN-PAGE and CN-PAGE gel systems. |
| PVDF Membrane | Blotting membrane for western transfer after native PAGE [25] [13]. | Recommended for western blotting; nitrocellulose binds Coomassie dye too tightly [13]. |
| Linear Acrylamide Gradient Gels | Separates proteins across a wide mass range (10 kDa to 10 MDa) by molecular sieving [41] [25]. | Standard for both BN-PAGE (e.g., 3-12%) and CN-PAGE. |
BN-PAGE and CN-PAGE are complementary techniques within the native protein analyst's toolkit. BN-PAGE is the definitive choice for high-resolution separation, accurate native mass estimation, and standard characterization of protein complexes. In contrast, CN-PAGE serves a specialized niche where its milder nature is paramount, enabling the study of delicate supramolecular structures and the direct assessment of protein function via in-gel activity assays. The decision between them is not one of superiority but of aligning the technique's strengths with the specific experimental goals, whether that is detailed structural analysis or functional investigation of native protein complexes.
Industry Applications in Drug Development and Quality Control
In the fields of drug development and biopharmaceutical quality control, protein characterization is a fundamental requirement. Polyacrylamide Gel Electrophoresis (PAGE) is a cornerstone technique for this purpose, with Sodium Dodecyl-Sulfate PAGE (SDS-PAGE) and Native PAGE representing two principal methodologies with distinct applications [9] [6]. The choice between these techniques is not trivial; it dictates the type of information obtained and has profound implications for downstream decision-making. SDS-PAGE denatures proteins, separating them primarily based on molecular weight, which is indispensable for assessing purity and subunit composition [9] [15]. In contrast, Native PAGE separates proteins in their folded, native state, preserving their biological activity and higher-order structure, which is critical for studying functional protein complexes and enzymatic activity [9] [21]. This guide provides an objective comparison of these techniques, supported by experimental data and protocols, to inform their strategic application in industry settings.
Fundamental Principles and Technical Comparison
The core difference between these techniques lies in their treatment of protein structure. SDS-PAGE employs the anionic detergent sodium dodecyl sulfate (SDS) and often a reducing agent like β-mercaptoethanol. SDS binds to and denatures proteins, linearizing them and conferring a uniform negative charge. This masks the proteins' intrinsic charge and eliminates the influence of shape, resulting in separation based almost exclusively on molecular mass [9] [6] [15]. Sample preparation typically involves heating in a denaturing buffer [6].
Conversely, Native PAGE uses non-denaturing conditions without SDS or reducing agents. Proteins retain their native conformation, quaternary structure, and cofactors. Consequently, their migration through the gel depends on a complex interplay of intrinsic charge, size, and shape [9] [6]. This preservation of structure allows for the recovery of functional proteins post-separation [6].
Table 1: Core Technical Differences Between SDS-PAGE and Native PAGE
| Feature | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight/mass [6] [15] | Size, intrinsic charge, and 3D structure [9] [6] |
| Protein State | Denatured and linearized [9] | Native, folded conformation [9] |
| Key Reagents | SDS, reducing agents (e.g., DTT, BME) [6] | Non-denaturing buffers; Coomassie dye (in BN-PAGE) [7] [6] |
| Sample Preparation | Heating in denaturing buffer [6] | No heating; mild buffer conditions [6] |
| Biological Activity | Lost [9] | Preserved [9] [7] |
| Protein Recovery | Not functional [6] | Functional proteins can be recovered [6] |
Experimental Data and Performance Comparison
Quantitative data highlights the operational and performance characteristics of each method. The following table summarizes key parameters based on standard laboratory protocols and published findings.
Table 2: Quantitative Performance and Operational Comparison
| Parameter | SDS-PAGE | Native PAGE | Experimental Context |
|---|---|---|---|
| Typical Run Temperature | Room Temperature [6] | 4°C [6] | Maintains stability of labile native complexes. |
| Metal Cofactor Retention | ~26% [7] | ~98% (BN-PAGE) [7] | Measured for Zn²⁺ in pig kidney proteome fractions [7]. |
| Enzyme Activity Retention | Inactive (0/9 model enzymes) [7] | Active (7/9 model enzymes in NSDS-PAGE; 9/9 in BN-PAGE) [7] | Assay of model enzymes including Zn²⁺ proteins post-electrophoresis [7]. |
| Resolution | High resolution of complex protein mixtures by mass [7] | Lower resolution for complex mixtures; high for functional complexes [7] | Comparison of proteome separation quality [7]. |
| Apparent Mass Shift | Not applicable (proteins denatured) | Observed (e.g., MCAD variant R206C) [21] | Indicates conformational changes while monomeric mass remains unchanged [21]. |
A key experimental finding is the development of Native SDS-PAGE (NSDS-PAGE), a modified approach that demonstrates the possibility of achieving high-resolution separation while retaining significant native functionality. This method involves removing SDS and EDTA from the sample buffer, omitting the heating step, and reducing the SDS concentration in the running buffer. This protocol resulted in a dramatic increase in Zn²⁺ retention from 26% (standard SDS-PAGE) to 98%, with the majority of tested enzymes retaining activity [7].
Detailed Experimental Protocols
This protocol is designed for determining molecular weight and assessing protein purity.
- Sample Preparation: Mix protein sample with 4X LDS sample loading buffer (containing SDS). Heat the mixture at 70°C for 10 minutes to denature the proteins.
- Gel Setup: Load samples and molecular weight standards onto a precast polyacrylamide gel (e.g., 12% Bis-Tris).
- Electrophoresis: Submerge the gel in a running buffer containing SDS (e.g., 50 mM MOPS, 50 mM Tris Base, 0.1% SDS, 1 mM EDTA, pH 7.7). Run at a constant voltage of 200V for approximately 45 minutes at room temperature.
- Post-Run Analysis: Proteins are typically visualized by staining (e.g., Coomassie Blue) or transferred to a membrane for Western blotting.
This protocol is adapted for analyzing the activity of specific protein complexes, such as Medium-Chain acyl-CoA Dehydrogenase (MCAD).
- Sample Preparation: Mix protein sample (e.g., recombinant enzyme or mitochondrial-enriched fraction) with a non-denaturing sample buffer (e.g., 50 mM BisTris, 50 mM NaCl, 10% Glycerol, 0.001% Ponceau S, pH 7.2). Do not heat.
- Gel Setup: Load samples onto a high-resolution gradient gel (e.g., 4-16% Bis-Tris).
- Electrophoresis: Use anode and cathode running buffers without denaturants. Run at a constant voltage of 150V for 90-95 minutes, ideally at 4°C.
- In-Gel Activity Staining: After electrophoresis, incubate the gel in a reaction mixture containing the physiological substrate (e.g., octanoyl-CoA for MCAD) and an electron acceptor like nitro blue tetrazolium chloride (NBT). Active enzymes will reduce NBT, producing a purple-colored diformazan precipitate at the location of the active complex.
Decision Workflow for Method Selection
The following diagram outlines a logical workflow for choosing between SDS-PAGE and Native PAGE based on research goals.
Research Reagent Solutions
The following table details essential materials and their functions for implementing these electrophoretic techniques.
Table 3: Key Research Reagents and Materials for PAGE
| Item | Function/Purpose | Key Considerations |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins and confers uniform negative charge for mass-based separation in SDS-PAGE [6] [15]. | Purity is critical for consistent results. |
| DTT or β-Mercaptoethanol | Reducing agents that break disulfide bonds, ensuring complete protein unfolding in SDS-PAGE [6]. | Fresh preparation is often necessary. |
| Coomassie G-250 Dye | Used in Blue Native PAGE (BN-PAGE) to confer charge to proteins without significant denaturation [7]. | Different from Coomassie R-250 used for staining. |
| Tris-Based Buffers | Provides the necessary ionic environment and pH control during electrophoresis in both techniques [7] [34]. | Composition varies between SDS-PAGE (e.g., Tris-Glycine) and Native PAGE (e.g., BisTris) [7]. |
| Polyacrylamide Gels | Forms the porous matrix through which proteins migrate. Gradient gels (e.g., 4-16%) can enhance resolution in Native PAGE [7] [21]. | Precast gels offer convenience and reproducibility [45]. |
| Molecular Weight Standards | A set of proteins of known mass for calibrating SDS-PAGE gels and estimating sample protein size [7]. | Pre-stained or unstained formats available. |
| Nitro Blue Tetrazolium (NBT) | Electron acceptor in in-gel activity assays; reduction forms a visible purple precipitate [21]. | Used to localize enzymatic activity after Native PAGE. |
SDS-PAGE and Native PAGE are not competing but complementary techniques that serve distinct purposes in the drug development and quality control pipeline. SDS-PAGE remains the gold standard for routine analysis of protein purity, expression, and molecular weight, forming the backbone of quality control workflows [9] [45]. Native PAGE, including its BN-PAGE and hrCN-PAGE variants, is a powerful tool for functional analysis, enabling the study of protein complexes, enzymatic mechanisms, and the structural impact of genetic variants in disease [46] [21]. The emergence of hybrid techniques like NSDS-PAGE [7] further broadens the analytical toolkit. A deep understanding of their principles, performance data, and protocols allows scientists to make informed choices, ensuring the application of the most appropriate method to answer specific biological and quality-related questions.
Troubleshooting Common Issues and Optimizing Your Gel Electrophoresis
In protein analysis research, selecting the appropriate electrophoretic technique is fundamental to obtaining reliable and meaningful data. Two primary methods, SDS-PAGE and Native PAGE, offer divergent approaches with distinct advantages and limitations. SDS-PAGE provides high-resolution separation based primarily on molecular weight by denaturing proteins, making it indispensable for determining protein size, purity, and expression levels. In contrast, Native PAGE preserves proteins in their functional, folded state, enabling the study of native charge, protein complexes, and enzymatic activity. This guide objectively compares their performance in resolving common experimental challenges—including smeared bands, smiling effects, and poor resolution—by synthesizing standard methodologies with innovative adaptations, providing researchers with a clear framework for technique selection and troubleshooting.
Core Principles: SDS-PAGE vs. Native PAGE
The fundamental differences between these two techniques dictate their application in research. The table below summarizes their key characteristics:
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight only [6] [8] | Molecular size, overall charge, and shape [6] [9] |
| Gel Nature | Denaturing [6] [47] | Non-denaturing [6] [9] |
| SDS Presence | Yes (denatures proteins and imparts uniform negative charge) [6] [47] | No [6] [8] |
| Sample State | Denatured and linearized [9] [47] | Native, folded conformation [6] [9] |
| Protein Function Post-Separation | Lost [47] | Retained [9] |
| Primary Applications | Molecular weight determination, checking purity/expression [6] [47] | Studying protein structure, complexes, and enzymatic activity [6] [9] |
| Typical Running Temperature | Room Temperature [6] | 4°C [6] |
Troubleshooting Common SDS-PAGE Issues: Causes and Experimental Solutions
Even well-optimized SDS-PAGE protocols can encounter problems. The following section details the root causes and experimental remedies for three common issues.
Smeared Bands
Smeared bands appear as diffuse, poorly defined streaks rather than sharp, discrete bands, complicating analysis.
Primary Causes and Corrective Methodologies:
- Excessive Voltage: Running the gel at too high a voltage generates excessive heat, causing protein denaturation and diffusion [48] [49] [50].
- Incomplete Denaturation: If proteins are not uniformly coated with SDS and fully denatured, they may not migrate uniformly.
- Protein Overloading: Loading too much protein overwhelms the gel's sieving capacity [49] [52].
- High Salt Concentration: High ionic strength in samples distorts the electric field [49] [52].
- Experimental Protocol: Desalt samples using dialysis, spin desalting columns, or protein precipitation (e.g., TCA precipitation) followed by resuspension in a low-salt buffer [49].
Smiling Effects
The "smiling effect" describes upward-curving bands at the gel's edges, caused by uneven heating where the center becomes warmer than the edges, leading to faster migration in the middle [48] [50].
Primary Causes and Corrective Methodologies:
- Joule Heating: The primary cause is heat generated by electric current flowing through the gel [48] [50].
- Experimental Protocol: Reduce the running voltage as a first step. For critical separations, run the gel in a cold room or use a circulating water cooling system. Placing an ice pack in the tank buffer can also help dissipate heat [48].
- Inadequate Buffer Circulation: Stagnant buffer leads to temperature gradients.
- Experimental Protocol: Ensure the buffer level is sufficient to cover the entire gel and acts as a heat sink. Gently stirring the buffer in the outer chamber before starting the run can help equalize temperature [52].
Poor Band Resolution
Poor resolution results in blurry, overlapping bands that are difficult to distinguish.
Primary Causes and Corrective Methodologies:
- Incorrect Gel Concentration: Using a gel with a pore size unsuitable for your target protein's molecular weight prevents effective sieving [49] [50].
- Experimental Protocol: Use a gel percentage appropriate for your protein's size range. For most proteins (5-250 kDa), 8-12% gels are standard. For very high molecular weight proteins, use lower percentage gels (e.g., 6%); for small peptides, use higher percentages (e.g., 15-20%). Gradient gels (e.g., 4-20%) are excellent for resolving a broad size range [48] [49].
- Improper Running Buffer: Old, contaminated, or incorrectly prepared buffer alters pH and ionic strength, disrupting current flow [48] [50].
- Insufficient Run Time: The gel may not have been run long enough for adequate separation [48] [49].
- Experimental Protocol: A standard practice is to run the gel until the dye front is ~0.5-1 cm from the bottom. For high molecular weight proteins, a longer run time may be necessary even after the dye front has migrated off the gel [48].
Advanced Experimental Protocol: Native SDS-PAGE (NSDS-PAGE)
A hybrid method, Native SDS-PAGE (NSDS-PAGE), has been developed to bridge the gap between standard SDS-PAGE and Native PAGE. It aims to provide high-resolution separation while retaining some native protein properties, such as bound metal ions and enzymatic activity [7].
Objective: To separate proteins with high resolution while preserving native enzymatic activity and metal cofactors [7].
Methodology Overview (Adapted from [7]):
- Sample Preparation: Mix 7.5 µL of protein sample with 2.5 µL of 4X NSDS sample buffer. Do not heat the samples.
- Gel Preparation: Use standard precast Bis-Tris gels. Pre-run the gel in ddH₂O for 30 minutes at 200V to remove storage buffers and unpolymerized acrylamide.
- Running Buffer: Use a modified Tris-MOPS running buffer containing a reduced concentration of SDS (0.0375% instead of the standard 0.1%) and no EDTA [7].
- Electrophoresis Conditions: Run at a constant voltage of 200V for approximately 45 minutes, similar to standard SDS-PAGE [7].
Supporting Experimental Data: A study comparing these methods demonstrated that Zn²⁺ retention in proteomic samples increased from 26% in standard SDS-PAGE to 98% in NSDS-PAGE. Furthermore, seven out of nine model enzymes, including four Zn²⁺ proteins, retained their activity after NSDS-PAGE separation, whereas all were denatured in standard SDS-PAGE [7].
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and materials critical for successful SDS-PAGE and Native PAGE experiments.
| Reagent/Material | Function/Purpose |
|---|---|
| Sodium Dodecyl Sulfate (SDS) | Anionic detergent that denatures proteins and imparts a uniform negative charge, making separation based on molecular weight possible [6] [47]. |
| Acrylamide/Bis-Acrylamide | Forms the cross-linked porous gel matrix that acts as a molecular sieve for separating proteins [6]. |
| Reducing Agents (DTT, BME) | Breaks disulfide bonds in proteins, ensuring complete unfolding and linearization for accurate molecular weight determination in SDS-PAGE [6] [8]. |
| Coomassie Blue/Silver Stain | Dyes used to visualize separated protein bands on the gel post-electrophoresis [48] [6]. |
| Tris-based Buffers | Provides the necessary conductive ionic environment and maintains a stable pH during electrophoresis [48] [7]. |
| Ammonium Persulfate (APS) & TEMED | Catalyzes the polymerization reaction of acrylamide to form the polyacrylamide gel [49]. |
| Glycerol | Added to sample buffer to increase density, ensuring samples sink to the bottom of the wells during loading [8]. |
| Tracking Dye (Bromophenol Blue) | A small, visible molecule that migrates ahead of the proteins, allowing visualization of the run's progress [48]. |
The choice between SDS-PAGE, Native PAGE, and advanced hybrids like NSDS-PAGE is not a matter of which technique is superior, but which is most appropriate for the specific research question. SDS-PAGE remains the gold standard for determining molecular weight and analyzing denatured proteins, though it is susceptible to issues like smiling and smearing that require careful control of voltage and sample preparation. Native PAGE is indispensable for functional studies where preserving protein activity and complex structure is paramount. The development of NSDS-PAGE offers a promising compromise, providing high-resolution separations while retaining critical native properties like metal binding and enzymatic activity. By understanding the underlying causes of common artifacts and applying the detailed methodologies outlined in this guide, researchers can optimize their electrophoretic approaches to generate robust, reproducible, and high-quality data.
Polyacrylamide gel electrophoresis (PAGE) serves as a foundational technique in biochemistry and molecular biology for separating complex protein mixtures. Within this context, researchers must choose between two primary separation methodologies: sodium dodecyl sulfate-PAGE (SDS-PAGE), which denatures proteins to separate them primarily by molecular weight, and native PAGE, which preserves protein structure and function while separating by charge, size, and shape [53] [3]. This guide focuses on a critical optimization parameter within both systems—acrylamide gel composition—comparing the performance and applications of fixed-concentration gels versus gradient gels. The selection between these gel types directly impacts resolution, sensitivity, and accuracy in protein analysis, making this optimization crucial for researchers in basic research and drug development [54] [55].
The polyacrylamide matrix forms a molecular sieve through which proteins migrate under an electric field. The pore size of this matrix is inversely related to the acrylamide percentage; lower percentages (e.g., 4-8%) create larger pores suitable for separating high molecular weight proteins, while higher percentages (e.g., 15-20%) create smaller pores that better resolve low molecular weight proteins [56] [3]. Understanding how to manipulate this matrix through either fixed or gradient concentrations enables researchers to tailor their electrophoretic separations to specific experimental needs, from routine analysis to complex proteomic studies.
Core Principles: SDS-PAGE vs. Native PAGE
The fundamental distinction in gel electrophoresis lies in the state of the proteins during separation. SDS-PAGE utilizes the anionic detergent sodium dodecyl sulfate (SDS) to denature proteins, disrupt their secondary and tertiary structures, and impart a uniform negative charge. This process negates the influence of inherent protein charge and shape, ensuring separation occurs almost exclusively based on polypeptide chain length and molecular weight [57] [3]. The resulting linearized proteins migrate through the gel at rates inversely proportional to the logarithm of their molecular mass, allowing for precise size determination when compared to standard protein ladders.
In contrast, native PAGE is performed without denaturing agents, preserving the protein's higher-order structure, enzymatic activity, and multi-subunit interactions [53] [3]. Separation depends on a combination of the protein's intrinsic charge, its size, and its three-dimensional shape. This makes native PAGE ideal for studying functional aspects of proteins, such as enzyme activity post-separation, protein-protein interactions within complexes, and the composition of multi-subunit proteins [7] [3]. However, the resulting migration pattern is more complex than in SDS-PAGE and does not directly correlate with molecular weight alone.
The following diagram illustrates the key differences in workflow and protein state between these two fundamental techniques:
Fixed-Percentage Gels: Precision for Defined Targets
Fixed-percentage gels (or single-concentration gels) are formulated with a uniform concentration of acrylamide throughout the resolving gel, creating a matrix with consistent pore size [53]. These gels are the workhorse of many routine laboratory applications, offering high resolution for proteins within a narrow molecular weight range.
Applications and Performance Characteristics
Fixed-percentage gels excel in scenarios where the target protein's approximate size is known beforehand. They provide maximum resolution around a specific molecular weight, making them ideal for quantifying specific proteins, assessing the purity of a single protein preparation, or confirming the results of a cloning experiment [58]. When running multiple samples with similar-sized proteins across several lanes, fixed gels ensure consistent migration and comparable results. Their slightly faster run times and lower cost compared to gradient gels also make them efficient for high-throughput routines where the protein size range is predictable and constrained [54] [58].
Optimization Guidelines
Selecting the correct acrylamide percentage is paramount for effective separation. The table below provides a guideline for choosing a fixed gel percentage based on the molecular weight of the target protein(s) [54] [56].
Table 1: Optimizing Fixed-Percentage Gels for Target Protein Size
| Target Protein Size (kDa) | Recommended Gel Percentage (%) | Common Applications and Targets |
|---|---|---|
| >200 | 4-6 | Large complexes, fibronectin |
| 25-200 | 7.5-8 | Antibodies, large enzymes |
| 15-100 | 10 | HSP70, tubulin, many enzymes |
| 10-70 | 12-12.5 | Cytokines, transcription factors |
| 12-45 | 15 | Small enzymes, peptide hormones |
| 4-40 | 15-20 | Peptides, ubiquitin |
Gradient Gels: Broad-Range Separation and Enhanced Resolution
Gradient gels consist of a single, continuous layer of polyacrylamide in which the concentration increases progressively from top to bottom. This creates a corresponding pore size gradient, with larger pores at the top and smaller pores at the bottom [54] [53].
Applications and Performance Characteristics
The primary advantage of gradient gels is their ability to resolve a remarkably broad spectrum of protein sizes on a single gel [54] [58]. This is invaluable when analyzing complex mixtures of unknown composition, samples with a wide size distribution, or when investigating phenomena like protein degradation or post-translational modifications that create multiple products of different sizes [58]. Furthermore, gradient gels produce sharper bands. As a protein migrates, its leading edge enters a region of higher acrylamide and smaller pores, slowing down relative to its trailing edge. This compresses the protein band, resulting in sharper, more defined bands that are easier to visualize and quantify [54]. This band-sharpening effect also improves the separation of proteins with similar molecular weights.
Optimization Guidelines
Choosing the right gradient depends on the expected size range of the proteins in the sample. Standard gradients like 4-20% are excellent for general-purpose use, but narrower gradients can be chosen for specific applications to maximize resolution in a particular size window [54].
Table 2: Selecting a Gradient Gel for Different Experimental Needs
| Protein Size Range (kDa) | Example Gradient Range (%) | Primary Application Scenario |
|---|---|---|
| 4 - 250 | 4 / 20 | Discovery work; unknown samples |
| 10 - 100 | 8 / 15 | Targeted analysis of broad-range proteins |
| 50 - 75 | 10 / 12.5 | High-resolution separation of similarly sized proteins |
Comparative Analysis: Fixed vs. Gradient Gels
Direct comparison of fixed-percentage and gradient gels reveals a clear trade-off between high, targeted resolution and broad, versatile separation. The following table summarizes the key performance characteristics and optimal use cases for each gel type [54] [53] [58].
Table 3: Performance Comparison: Fixed vs. Gradient Gels
| Feature | Fixed-Percentage Gel | Gradient Gel |
|---|---|---|
| Pore Size | Uniform throughout the gel | Varies continuously from top to bottom |
| Effective Separation Range | Narrow, optimized for a specific size range | Very wide, capable of resolving many sizes on one gel |
| Band Sharpness | Good | Superior due to the band-stacking effect [54] |
| Resolution of Similar-sized Proteins | High within its optimal range | Enhanced, especially in the middle of the gradient |
| Best For | Samples with proteins of known, similar sizes; routine target analysis | Complex mixtures, unknown samples, very large or very small proteins |
| Run Time | Slightly faster | Slightly longer |
| Cost (Pre-cast) | Lower | Slightly higher |
| Ease of Preparation (Hand-cast) | Simpler to pour | Requires specialized equipment or techniques [54] |
Experimental Protocols and Methodologies
Protocol for Casting a Fixed-Percentage Resolving Gel
The following protocol is adapted for a standard 10% Tris-Glycine mini gel for SDS-PAGE, with a total volume of 10 mL [56] [3]. Caution: Acrylamide is a potent neurotoxin. Wear gloves and appropriate personal protective equipment throughout.
- Gel Casting Setup: Clean the glass plates and spacers thoroughly. Assemble the gel cassette according to the manufacturer's instructions and ensure it is properly sealed in the casting stand.
- Gel Solution Preparation: In a beaker or conical flask, combine the reagents in the following order:
- dH₂O: 4.0 mL
- 1.5 M Tris-HCl (pH 8.8): 2.5 mL
- 10% SDS: 100 µL
- 30% Acrylamide/Bis (29.2:0.8 ratio): 3.3 mL
- Polymerization: Mix gently. Immediately before pouring, add:
- 10% Ammonium Persulfate (APS): 50 µL
- TEMED: 5 µL Swirl gently to mix. Note: TEMED and APS initiate rapid polymerization; work quickly after their addition.
- Pouring and Overlaying: Pour the gel solution into the assembled cassette. Gently overlay the gel surface with water-saturated butan-1-ol or isopropanol to ensure a flat, even interface. Allow the gel to polymerize completely (typically 15-30 minutes).
- Pouring Stacking Gel: Once polymerized, pour off the overlay and rinse the top of the gel with dH₂O. Prepare the stacking gel solution (e.g., 5% acrylamide) without APS and TEMED. For a 5 mL stacking gel, mix:
- dH₂O: 3.05 mL
- 0.5 M Tris-HCl (pH 6.8): 1.25 mL
- 10% SDS: 50 µL
- 30% Acrylamide/Bis: 650 µL Add 25 µL of 10% APS and 10 µL of TEMED, mix, and pour on top of the resolving gel. Insert a comb without introducing air bubbles. Polymerize for 15-30 minutes.
Protocol for Casting a Gradient Gel Using a Gradient Maker
Casting a gradient gel requires more specialized equipment but provides unparalleled range [54].
- Setup: Set up a two-chamber gradient mixer with a connecting valve and an outlet tube. Place the gel cassette vertically on a level surface, with the outlet tube leading to the top of the cassette. Ensure the valve between the chambers is closed.
- Solution Preparation: Prepare low-percentage and high-percentage acrylamide solutions in separate beakers. For example, for a 4-20% gradient, prepare a 4% "light" solution and a 20% "heavy" solution. Do not add APS and TEMED at this stage.
- Loading the Gradient Maker:
- Place a small stir bar in the chamber furthest from the outlet (the "resolving" or "heavy" solution chamber).
- Close the inter-chamber valve. Pour the high-percentage solution into this chamber.
- Open the inter-chamber valve momentarily to allow a small amount of solution to flow through, clearing air from the channel, then close it.
- Pour the low-percentage solution into the other chamber (closer to the outlet).
- Initiating Polymerization and Pouring: Add APS and TEMED to both chambers and stir gently. Open the inter-chamber valve. Simultaneously, start the peristaltic pump (or open the gravity flow valve) and begin stirring the "heavy" solution chamber. The solutions will mix gradually as they are pumped into the gel cassette, with the low-percentage solution flowing first, followed by an increasing concentration of the high-percentage solution, creating a linear gradient.
- Overlay and Polymerize: Once poured, gently overlay the gel with water-saturated butanol or water. Allow the gel to polymerize fully. A stacking gel is typically not used with gradient gels, as the gradient itself performs the stacking function [3].
The Scientist's Toolkit: Essential Reagents and Materials
Successful gel electrophoresis relies on a suite of specialized reagents and equipment. The following table details key solutions and materials required for performing SDS-PAGE and native PAGE, along with their critical functions [54] [56] [3].
Table 4: Essential Research Reagent Solutions for Protein Gel Electrophoresis
| Reagent/Material | Function | Key Considerations |
|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked polymer matrix that acts as a molecular sieve. | Standard ratio is 29:1 (acrylamide:bis); concentration determines gel pore size; handle as a neurotoxin. |
| APS (Ammonium Persulfate) | Initiates the polymerization reaction by generating free radicals. | Prepare fresh 10% solution in water for optimal polymerization efficiency. |
| TEMED | Catalyzes the polymerization reaction by accelerating the formation of free radicals from APS. | Polymerization begins immediately upon addition; add last and work swiftly. |
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins and confers a uniform negative charge, enabling separation by size in SDS-PAGE. | Critical for denaturing gels; typically included in sample buffer and running buffer. |
| Tris-Based Buffers | Provides the conductive medium and maintains stable pH during electrophoresis (e.g., Tris-Glycine, Bis-Tris). | Bis-Tris gels (neutral pH) offer superior stability and resolution for complex samples vs. Tris-Glycine (alkaline pH) [55]. |
| Sample Loading Buffer | Contains dye to visualize migration, glycerol to density the sample, and SDS/reducing agents for denaturation. | For native PAGE, SDS and reducing agents are omitted to preserve protein structure [53] [7]. |
| Protein Molecular Weight Marker | Contains proteins of known sizes to estimate the molecular weight of unknown proteins in the sample. | Essential for accurate molecular weight determination and monitoring run progress. |
| Coomassie/Silver Stain | Used for post-electrophoresis visualization of separated protein bands. | Coomassie for general use; silver stain for high-sensitivity detection of low-abundance proteins [57]. |
Advanced Applications and Hybrid Techniques
The landscape of gel electrophoresis continues to evolve with methodologies that bridge the gap between denaturing and native techniques. One significant advancement is Native SDS-PAGE (NSDS-PAGE), a modified protocol that reduces the SDS concentration in the running buffer, omits EDTA, and eliminates the sample heating step [7]. This approach aims to balance the high resolution of traditional SDS-PAGE with the retention of certain native functional properties. Experimental data shows that this method can preserve metalloprotein metal content with high efficiency (e.g., 98% Zn²⁺ retention in proteomic samples versus 26% with standard SDS-PAGE) and maintain the activity of many enzymes post-separation [7].
For the most complex separation challenges, Two-Dimensional Gel Electrophoresis (2D-PAGE) remains the gold standard. This technique combines two orthogonal separation principles: proteins are first separated by their native isoelectric point (pI) using isoelectric focusing (IEF), followed by separation by molecular weight using SDS-PAGE in the second dimension [57] [3]. The result is a high-resolution map where thousands of protein spots can be resolved from a single sample, making it an indispensable tool in proteomics for analyzing protein expression, post-translational modifications, and isoform diversity.
In protein analysis research, the choice between SDS-PAGE and native PAGE represents a fundamental methodological crossroads. This decision dictates whether proteins are separated solely by molecular weight under denaturing conditions or by a combination of size, charge, and shape while preserving their native structure and function. The buffer conditions—specifically pH, ionic strength, and temperature—are not merely supporting parameters but are critical determinants of experimental success. Proper management of these conditions ensures optimal protein stability, migration, and resolution, directly impacting the reliability and interpretability of results. This guide provides an objective comparison of how these buffer conditions differ between the two techniques and their effect on protein separation, supported by experimental data and detailed protocols.
Fundamental Principles and Comparative Analysis
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) is a discontinuous electrophoretic system that separates proteins based primarily on their molecular mass, typically in the range of 5 to 250 kDa [59]. The technique employs the anionic detergent sodium dodecyl sulfate (SDS), which denatures proteins by binding to the polypeptide backbone in a constant weight ratio (approximately 1.4 g SDS per 1 g of protein) and confers a uniform negative charge [59] [3]. This process masks the proteins' intrinsic charge, ensuring separation is based almost exclusively on molecular size as proteins migrate through the polyacrylamide gel matrix [9] [3].
In contrast, native PAGE separates proteins in their folded, functional state based on a combination of factors including their intrinsic charge, size, and three-dimensional shape [9] [3]. Without denaturing agents, proteins retain their biological activity, enzymatic function, and complex quaternary structures, allowing researchers to study protein-protein interactions, oligomerization states, and functional activities [9]. The migration occurs because most proteins carry a net negative charge in alkaline running buffers, with higher charge density leading to faster migration, while the gel matrix provides a sieving effect that regulates movement according to size and shape [3].
Table 1: Core Principles and Applications of SDS-PAGE vs. Native PAGE
| Analysis Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Primary Separation Basis | Molecular mass/weight [6] [3] | Net charge, size, and shape of native structure [6] [3] |
| Protein State | Denatured/unfolded [9] [6] | Native/folded [9] [6] |
| Biological Function | Not retained [9] | Retained [9] [3] |
| Key Buffer Additives | SDS, reducing agents (DTT, β-ME) [59] [6] | No denaturing agents; may include Coomassie dye (BN-PAGE) [7] [23] |
| Ideal Applications | Molecular weight determination, protein purity assessment, western blotting [9] [18] | Study of protein complexes, enzymatic activity, oligomerization state [9] [3] |
Buffer Condition Parameters: A Detailed Comparison
The effectiveness of both SDS-PAGE and native PAGE is heavily dependent on the precise management of buffer conditions, which differ significantly between the two techniques.
pH Environment and Buffer Composition
The pH of the electrophoretic environment is meticulously controlled to ensure predictable protein behavior and effective separation.
In SDS-PAGE, a discontinuous buffer system is used with different pH values in the stacking and resolving gels to create a stacking effect that sharpens protein bands. The stacking gel typically has a neutral pH (around 6.8), while the separating gel has a basic pH (around 8.8) [59]. This pH gradient is crucial for the stacking effect, where glycinate ions transition from trailing ions to leading ions as they move from the stacking to the separating gel, compressing the protein samples into sharp bands before separation begins [59]. Common buffer systems include Tris-glycine-chloride in traditional Laemmli systems [59], or Bis-tris methane with MOPS or MES in commercially available pre-cast gels for a continuous, more stable system near neutral pH [59].
For native PAGE, the pH must be carefully selected to maintain protein stability and activity. Running buffers are typically alkaline to ensure most proteins carry a net negative charge and migrate toward the anode [3]. However, the specific pH must be chosen to avoid the isoelectric point (pI) of the proteins of interest, as proteins precipitate at their pI [3]. Blue Native (BN)-PAGE, a common variant, uses BisTris-based buffers at neutral pH (e.g., pH 7.0) to preserve protein complexes, with Coomassie G-250 dye providing the necessary charge for migration [7] [23].
Ionic Strength and Conductivity
Ionic strength directly affects current flow and heating during electrophoresis and must be optimized for each technique.
In SDS-PAGE, the running buffer contains ions like Tris, glycine, and SDS to conduct current. The ionic strength is balanced to provide sufficient conductivity without excessive heat generation [59]. The presence of SDS at concentrations above 1 mM ensures complete protein denaturation and uniform charge masking [59].
For native PAGE, maintaining the correct ionic strength is critical for preserving non-covalent protein interactions. A study on Native SDS-PAGE (NSDS-PAGE)—a modified approach that aims to retain some native properties—found that reducing the SDS concentration in the running buffer from the standard 0.1% to 0.0375% was crucial for retaining Zn²⁺ in metalloproteins and preserving enzymatic activity [7]. BN-PAGE uses specific buffers like BisTris-ACA (100 mM BisTris-HCl, 500 mM 6-aminocaproic acid) which provide appropriate ionic strength while stabilizing protein complexes [23].
Temperature Control During Electrophoresis
Temperature management is essential for preventing protein denaturation and ensuring consistent results.
SDS-PAGE is typically performed at room temperature [6]. Since proteins are already denatured, moderate heat generation during electrophoresis does not adversely affect separation and may even help maintain proteins in a denatured state.
Native PAGE, however, is often run at 4°C to maintain protein stability and prevent denaturation or aggregation during separation [6] [3]. The lower temperature helps preserve weak non-covalent interactions that maintain protein complexes and enzymatic function. BN-PAGE protocols specifically recommend running the electrophoresis with chilled buffers at 4°C to protect labile protein interactions [23].
Table 2: Quantitative Comparison of Optimal Buffer Conditions
| Parameter | SDS-PAGE | Native PAGE | Experimental Impact |
|---|---|---|---|
| Typical Running Buffer pH | 7.7 (MOPS/Tris) [7] | 6.8 (BN-PAGE Cathode) [7] | Affects protein charge, migration direction & stability |
| Sample Buffer pH | 8.5 (LDS buffer) [7] | 7.2 (BN-PAGE) [7] | Influences protein denaturation (SDS-PAGE) or stability (Native) |
| Critical Additive Concentration | 0.1% SDS (standard) [7] | 0.02% Coomassie G-250 (BN-PAGE) [7] | SDS denatures; Coomassie provides charge without denaturation |
| Optimal Running Temperature | Room Temperature [6] | 4°C [6] [3] | Prevents heat-induced denaturation of native structures |
| Metal Ion Retention | 26% Zn²⁺ retention [7] | 98% Zn²⁺ retention (NSDS-PAGE) [7] | Critical for studying functional metalloproteins |
Experimental Protocols and Data
Standard SDS-PAGE Protocol
Sample Preparation:
- Mix protein sample with 4X LDS sample buffer (106 mM Tris HCl, 141 mM Tris Base, 0.51 mM EDTA, 2% LDS, 10% Glycerol, pH 8.5) [7].
- Add reducing agent (e.g., 50 mM DTT or 5% β-mercaptoethanol) to break disulfide bonds [59].
- Heat samples at 70°C for 10 minutes or 95°C for 5 minutes to denature proteins [7] [59].
- Centrifuge briefly to collect condensed sample.
Gel Electrophoresis:
- Use pre-cast or freshly prepared polyacrylamide gel with appropriate percentage (e.g., 10-12% for most proteins).
- Load samples and molecular weight markers into wells.
- Fill electrophoresis chamber with running buffer (e.g., 50 mM MOPS, 50 mM Tris Base, 0.1% SDS, 1 mM EDTA, pH 7.7) [7].
- Run at constant voltage (e.g., 200V for 45 minutes) until dye front reaches bottom [7].
Blue Native PAGE (BN-PAGE) Protocol
Sample Preparation:
- Mix protein sample with 4X BN-PAGE sample buffer (50 mM BisTris, 50 mM NaCl, 10% Glycerol, pH 7.2) [7].
- Do not heat samples to preserve native structure.
- Add Coomassie G-250 to a final concentration of 0.25-0.5% to provide charge for migration [23].
Gel Electrophoresis:
- Use pre-cast NativePAGE Novex 4-16% Bis-Tris gels or equivalent.
- Load samples and native protein standards.
- Use anode buffer (50 mM BisTris, pH 6.8) and cathode buffer (50 mM BisTris, 50 mM Tricine, 0.02% Coomassie G-250, pH 6.8) [7].
- Run at constant voltage (e.g., 150V for 90-95 minutes) at 4°C until dye front reaches bottom [7].
Performance Comparison Data
Experimental studies directly comparing these techniques reveal significant functional differences:
- Metal Retention: When analyzing the Zn²⁺ proteome, standard SDS-PAGE retained only 26% of bound Zn²⁺, while modified native conditions (NSDS-PAGE) retained 98% of metal ions, crucial for studying functional metalloproteins [7].
- Enzymatic Activity: In a study with nine model enzymes, including four Zn²⁺ proteins, seven retained activity after NSDS-PAGE separation, while all nine were denatured and inactivated during standard SDS-PAGE [7].
- Proteomic Analysis: A comparative analysis of human bronchial smooth muscle cells found SDS-PAGE-MS assigned 2,552 proteins from the supernatant fraction, while nondenaturing 2DE-MS assigned 4,323 proteins with higher sensitivity for protein interaction studies [27].
The Scientist's Toolkit: Essential Research Reagents
Successful execution of either electrophoretic method requires specific reagent systems optimized for each technique's requirements.
Table 3: Essential Reagents for SDS-PAGE and Native PAGE
| Reagent Category | Specific Examples | Function & Importance |
|---|---|---|
| Denaturing Detergents | Sodium Dodecyl Sulfate (SDS), LDS [7] [59] | Unfolds proteins, masks intrinsic charge; enables MW-based separation in SDS-PAGE |
| Reducing Agents | Dithiothreitol (DTT), β-Mercaptoethanol (BME) [59] | Breaks disulfide bonds; ensures complete denaturation and subunit separation |
| Native Charge Agents | Coomassie G-250 (BN-PAGE) [7] [23] | Imparts negative charge to native proteins; enables migration without denaturation |
| Buffering Systems | Tris-Glycine, Bis-Tris, MOPS, Tricine [7] [59] | Maintains optimal pH; critical for protein stability and stacking effects |
| Protease Inhibitors | PMSF, Complete Protease Inhibitor Cocktails [7] | Prevents protein degradation during sample preparation; especially crucial for native PAGE |
| Molecular Weight Markers | Pre-stained Protein Ladders, Unstained Standards [7] [3] | Provides size references for molecular weight estimation and run monitoring |
The choice between SDS-PAGE and native PAGE represents a fundamental strategic decision in protein analysis, dictated primarily by research objectives. SDS-PAGE, with its controlled denaturing conditions including SDS, reducing agents, and heating, provides reliable separation by molecular weight ideal for quantitative analysis, purity assessment, and western blotting. Conversely, native PAGE, maintained at lower temperatures with non-denaturing buffers, preserves protein complexes, enzymatic activity, and metal cofactors, enabling functional studies and interaction analysis. Mastery of the distinct buffer conditions—particularly the precise management of pH, ionic strength through specific detergent concentrations, and temperature control—is essential for obtaining reliable, reproducible results with either technique. Understanding these methodological differences allows researchers to select the most appropriate approach for their specific protein characterization needs.
Native polyacrylamide gel electrophoresis (Native PAGE) serves as an indispensable tool for researchers who need to analyze proteins in their biologically active states. Unlike its denaturing counterpart SDS-PAGE, which separates proteins primarily by molecular weight, Native PAGE preserves higher-order structures, protein complexes, and enzymatic activity by omitting denaturing agents [60] [6]. This technique enables the separation of proteins based on their intrinsic charge, size, and shape, allowing researchers to study functional protein complexes, oligomeric states, and native interactions [60]. However, these advantages come with significant challenges, including protein aggregation during electrophoresis and difficulties in maintaining full biological activity throughout the separation process. This guide examines these challenges in the context of SDS-PAGE alternatives and presents both established and emerging methodological solutions.
Core Challenges in Native PAGE
Protein Aggregation During Electrophoresis
Protein aggregation represents a fundamental obstacle in Native PAGE, particularly when studying complex protein mixtures or proteins with inherent instability. During electrophoresis, proteins can undergo undesirable associations leading to the formation of both soluble and insoluble aggregates [61]. These aggregates can manifest as either amorphous structures lacking defined organization or highly ordered fibrillar assemblies rich in β-sheet architecture [61].
The aggregation process typically follows several pathways relevant to Native PAGE conditions. Proteins may self-associate through reversible association of native monomers, where "sticky" patches on protein surfaces facilitate oligomerization through electrostatic interactions or complementary surface features [61]. More problematic is the aggregation of conformationally altered monomers, where transient changes in protein structure create non-native states with strong association propensity [61]. Additionally, chemical modifications during sample preparation or electrophoresis can introduce new "sticky" patches on protein surfaces or alter electrostatic profiles, further promoting aggregation [61].
The impact of aggregation on Native PAGE results is substantial, leading to smeared bands, poor resolution, loss of target proteins, and inaccurate representation of native protein complexes. These issues become particularly pronounced when working with therapeutic proteins or multimeric enzymes where maintaining quaternary structure is essential for function.
Maintaining Protein Activity and Native Structure
Preserving biological activity throughout the electrophoresis process constitutes the second major challenge in Native PAGE. While the fundamental premise of Native PAGE is maintaining proteins in their functional state, numerous factors during separation can compromise activity, including:
- Dissociation of protein complexes due to weak interactions with the gel matrix or buffer components
- Loss of essential cofactors and metal ions that maintain structural integrity
- Oxidation or chemical modification of sensitive active site residues
- pH-induced conformational changes affecting catalytic efficiency
The retention of metal cofactors presents a particular challenge for metalloproteins. Traditional SDS-PAGE results in near-complete loss of bound metal ions, with one study showing only 26% Zn²⁺ retention in standard SDS-PAGE conditions [7]. This metal stripping directly abolishes enzymatic activity in metalloenzymes and disrupts structural stability in metal-stabilized proteins.
Comparative Analysis: Native PAGE vs. Alternative Techniques
Table 1: Technical Comparison of PAGE Methodologies for Addressing Aggregation and Activity Challenges
| Parameter | Native PAGE | SDS-PAGE | BN-PAGE | NSDS-PAGE |
|---|---|---|---|---|
| Protein State | Native, folded structure [6] | Denatured, linearized [60] [6] | Native, folded structure [7] | Native, folded structure [7] |
| Separation Basis | Size, charge, and shape [6] | Molecular weight only [60] [6] | Size and native charge [7] | Molecular weight with native features [7] |
| Aggregation Risk | High | Low | Moderate | Low |
| Activity Retention | High (when optimized) [6] | None [7] [6] | High [7] | High (7 of 9 model enzymes active) [7] |
| Metal Retention | Protocol-dependent | 26% Zn²⁺ retention [7] | High | 98% Zn²⁺ retention [7] |
| Resolution | Moderate [7] | High [7] [60] | Lower [7] | High [7] |
| Typical Applications | Functional studies, complexes [6] [21] | Molecular weight determination, purity analysis [60] [6] | Protein-protein interactions, membrane complexes [7] | Metalloprotein analysis, functional proteomics [7] |
Table 2: Performance Metrics for Enzyme Activity Retention Across PAGE Methods
| Method | Enzymes Retaining Activity | Metal Retention | Key Limitations |
|---|---|---|---|
| SDS-PAGE | 0 of 9 model enzymes [7] | 26% (Zn²⁺) [7] | Complete denaturation, no functional analysis [7] |
| BN-PAGE | 9 of 9 model enzymes [7] | High | Lower resolution, complex protocol [7] |
| NSDS-PAGE | 7 of 9 model enzymes [7] | 98% (Zn²⁺) [7] | Requires protocol optimization [7] |
| Native PAGE | Variable (method-dependent) | Variable (method-dependent) | Aggregation issues, inconsistent results |
Modified Native Methods: Experimental Solutions
Blue Native PAGE (BN-PAGE)
BN-PAGE represents a specialized Native PAGE variant that incorporates Coomassie Brilliant Blue G-250 dye, which confers additional negative charge to protein complexes, improving separation while largely maintaining native structure and function [7] [6].
Protocol Overview:
- Sample Buffer: 50 mM BisTris, 50 mM NaCl, 16 mM HCl, 10% glycerol, 0.001% Ponceau S, pH 7.2 [7]
- Cathode Buffer: 50 mM BisTris, 50 mM Tricine, 0.02% Coomassie G-250, pH 6.8 [7]
- Anode Buffer: 50 mM BisTris, 50 mM Tricine, pH 6.8 [7]
- Gel System: Native-PAGE Novex 4-16% Bis-Tris gradient gels [7]
- Electrophoresis Conditions: Constant voltage (150V), room temperature, 90-95 minutes [7]
Applications and Limitations: BN-PAGE has proven particularly valuable for studying membrane protein complexes and protein-protein interactions [7]. However, it suffers from lower resolution compared to denaturing methods and can introduce ambiguities in molecular weight determination [7]. The dye itself may occasionally interfere with subsequent analytical techniques or affect protein function in sensitive systems.
Native SDS-PAGE (NSDS-PAGE)
NSDS-PAGE represents a hybrid approach that modifies traditional SDS-PAGE conditions to preserve native characteristics while maintaining high resolution [7].
Key Protocol Modifications:
- SDS Reduction: Running buffer SDS concentration reduced from 0.1% to 0.0375% [7]
- EDTA Elimination: Removed from both sample and running buffers to preserve metal cofactors [7]
- Sample Preparation Omission: No heating step and elimination of SDS from sample buffer [7]
- Sample Buffer Composition: 100 mM Tris HCl, 150 mM Tris base, 10% glycerol, 0.0185% Coomassie G-250, 0.00625% Phenol Red, pH 8.5 [7]
- Running Buffer: 50 mM MOPS, 50 mM Tris Base, 0.0375% SDS, pH 7.7 [7]
Performance Advantages: This modified approach demonstrated remarkable success in preserving both metal ions and enzymatic activity. Experimental data showed Zn²⁺ retention increased from 26% in standard SDS-PAGE to 98% in NSDS-PAGE, with seven of nine model enzymes retaining activity after separation [7]. This positions NSDS-PAGE as a compelling alternative when both high resolution and functional preservation are required.
Advanced In-Gel Activity Assays
Recent methodological advances enable direct functional analysis following Native PAGE separation. These approaches are particularly valuable for studying enzymatic deficiencies and variant impacts, as demonstrated in research on medium-chain acyl-CoA dehydrogenase (MCAD) [21].
In-Gel Activity Assay Protocol:
- Electrophoresis: High-resolution clear native PAGE (hrCN-PAGE) with 4-16% gradient gels
- Staining Solution: Contains physiological substrate (octanoyl-CoA for MCAD) and nitro blue tetrazolium chloride (NBT) as electron acceptor
- Visualization: Formation of insoluble purple diformazan precipitate at active enzyme bands
- Incubation: 10-15 minutes at room temperature [21]
Applications: This methodology enables differentiation between active tetramers and inactive aggregated or fragmented forms of enzymes, providing insights impossible with standard solution-based assays [21]. The approach has been successfully applied to characterize pathogenic variants in metabolic disorders, revealing how specific mutations affect oligomeric state and function separately from catalytic impairment [21].
Research Reagent Solutions
Table 3: Essential Reagents for Native PAGE and Modified Protocols
| Reagent | Function | Protocol-Specific Considerations |
|---|---|---|
| Coomassie G-250 | Imparts charge in BN-PAGE [7] | Critical for protein complex separation; alternative to SDS |
| Low-concentration SDS (0.0375%) | Limited denaturation in NSDS-PAGE [7] | Balance between resolution and native state preservation |
| Cyclodextrin | SDS removal for in-gel refolding [62] | Enables fluorescence detection of refolded GFPs after SDS-PAGE |
| Nitro Blue Tetrazolium (NBT) | Electron acceptor in activity assays [21] | Forms insoluble purple precipitate for activity visualization |
| Glycerol | Density agent in sample buffers [7] | Prevents sample diffusion without significant denaturation |
| Alternative Detergents | Milder extraction and separation [63] | Sarkosyl and sodium N-lauroyl glutamate for sensitive proteins |
Methodological Workflow and Decision Pathway
The following diagram illustrates the strategic decision process for selecting appropriate electrophoresis methods based on research objectives and sample characteristics:
Addressing the dual challenges of protein aggregation and activity maintenance in Native PAGE requires thoughtful method selection and potential protocol modifications. While standard Native PAGE remains valuable for many applications, emerging hybrid approaches like NSDS-PAGE offer compelling alternatives by balancing the high resolution of denaturing methods with the functional preservation of native techniques. The optimal choice depends critically on specific research objectives, with BN-PAGE excelling for complex analysis, NSDS-PAGE providing superior resolution for metalloproteins, and specialized in-gel activity assays enabling direct functional characterization. As methodological innovations continue to evolve, researchers now possess an expanding toolkit to overcome the traditional limitations of native electrophoresis while preserving the biological relevance that makes these approaches indispensable.
Best Practices for Sample Preparation and Loading to Ensure Reproducibility
In protein analysis research, the choice between SDS-PAGE and Native PAGE fundamentally shapes experimental outcomes. SDS-PAGE denatures proteins into linear chains, separating them primarily by molecular weight, while Native PAGE preserves protein complexes in their native, functional state for analysis of charge, size, and interactions [9] [15]. Sample preparation is the most critical factor determining the success and reproducibility of either technique, as improper handling can compromise resolution, accuracy, and reliability. This guide details best practices and protocols for preparing and loading samples to ensure consistent, high-quality results in both methodologies.
Core Principles: SDS-PAGE vs. Native PAGE
The foundational difference between these techniques lies in their treatment of protein structure, which dictates every aspect of sample preparation.
Comparative Workflow: Sample Preparation
The diagram below contrasts the core sample preparation workflows for SDS-PAGE and Native PAGE, highlighting key divergences in denaturation and reduction steps.
Key Distinctions in Sample Treatment:
- Denaturation and Charge Manipulation: SDS-PAGE uses sodium dodecyl sulfate (SDS) to denature proteins and impart a uniform negative charge, ensuring separation is based almost solely on molecular weight [57] [15]. Native PAGE omits SDS, allowing proteins to retain their native conformation, intrinsic charge, and functional activity [9].
- Reduction of Disulfide Bonds: SDS-PAGE often includes a reducing agent like β-mercaptoethanol (BME) or dithiothreitol (DTT) to break disulfide bonds, further linearizing proteins [64] [18]. This step is excluded in Native PAGE to preserve protein complexes and quaternary structure [9].
Detailed Methodologies and Protocols
SDS-PAGE: Denaturing Sample Preparation
This protocol ensures complete protein denaturation for accurate molecular weight separation [64] [57] [65].
Sample Buffer Composition: The standard Laemmli buffer contains:
Step-by-Step Protocol:
- Mix Sample and Buffer: Combine protein sample with 2X or 5X SDS sample buffer. A typical ratio is 1:1 or 1:4 (buffer:sample) [65] [66].
- Denature and Reduce: Heat mixture at 95°C for 5-10 minutes [64] [57]. This step is crucial for complete unfolding.
- Brief Centrifugation: Spin samples briefly to collect condensation and pellet debris [64].
- Load Gel: Load clarified supernatant into gel wells. Recommended protein amounts are 1 µg for purified proteins and 10-50 µg for complex lysates [64] [66].
Native PAGE: Non-Denaturing Sample Preparation
This protocol maintains protein native state to study function and interactions [7] [9].
Sample Buffer Composition: Native buffer typically contains:
- No SDS or Denaturants: Preserves native structure.
- Glycerol: For easy loading.
- Tracking Dye.
- Mild Buffer: Such as Tris at neutral pH to maintain protein stability.
Step-by-Step Protocol:
Optimizing Reproducibility: Critical Factors
Several variables must be controlled to ensure consistent, reliable results across experiments.
Key Factors for Reproducibility
- Buffer Composition and pH: Precise buffer pH is critical for consistent protein migration, especially in Native PAGE where intrinsic charge drives separation [18].
- Sample Integrity: Avoid repeated freeze-thaw cycles and protease contamination. Use protease inhibitors and store aliquots at -80°C.
- Protein Concentration: Overloading causes smearing, underloading prevents detection. Optimize concentration for your detection method [64].
- Gel Percentage: Match gel pore size to protein molecular weight for optimal resolution [57] [65].
- Electrophoresis Conditions: Maintain constant voltage (150-200V for mini-gels) and ensure buffer levels cover electrodes [64] [66].
Experimental Data and Performance Comparison
Direct comparisons demonstrate how preparation methods affect experimental outcomes.
Quantitative Comparison of Metal Retention and Enzyme Activity
The table below summarizes experimental data comparing protein functionality after different PAGE methods, highlighting the dramatic impact of sample preparation.
| Experimental Metric | SDS-PAGE | Native SDS-PAGE | BN-PAGE |
|---|---|---|---|
| Zn²⁺ Retention in Proteomic Samples | 26% | 98% | Not Specified |
| Active Model Enzymes | 0 out of 9 | 7 out of 9 | 9 out of 9 |
| Separation Basis | Molecular Weight | Molecular Weight (High Resolution) | Native Charge & Size |
| Protein State | Denatured | Mixed (Mostly Native) | Fully Native |
Source: Experimental data from PMC (2014) [7]
Impact on Antibody Purity Analysis
Comparative studies between SDS-PAGE and Capillary Electrophoresis-SDS (CE-SDS) reveal limitations in traditional SDS-PAGE for detecting specific impurities like nonglycosylated IgG, which can co-migrate with main species [67]. While not a direct Native PAGE comparison, this highlights how separation limitations affect analytical precision.
Research Reagent Solutions
Essential reagents and their functions for reproducible sample preparation.
| Reagent/Category | Function in SDS-PAGE | Function in Native PAGE |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins; confers uniform negative charge [57] | Not Used |
| β-Mercaptoethanol (BME) or DTT | Reducing agent; breaks disulfide bonds [64] [18] | Not Used |
| Tris-Based Buffers | Maintains pH during denaturation and electrophoresis [65] | Maintains native-friendly pH environment |
| Glycerol | Increases sample density for gel loading [7] | Increases sample density for gel loading |
| Tracking Dye (Bromophenol Blue) | Visualizes migration front during electrophoresis [65] | Visualizes migration front during electrophoresis |
| Protease Inhibitors | Preserves sample integrity by preventing proteolysis | Preserves sample integrity by preventing proteolysis |
Advanced Applications: Native SDS-PAGE
A hybrid approach called Native SDS-PAGE (NSDS-PAGE) offers a compromise, modifying standard SDS-PAGE conditions by removing SDS and EDTA from sample buffer, omitting heating, and reducing SDS in running buffer [7]. This method maintains high-resolution separation while preserving metal ions and enzymatic activity in many proteins, demonstrating how preparation tweaks can expand analytical capabilities.
Reproducible sample preparation requires strict adherence to technique-specific protocols: complete denaturation and reduction for SDS-PAGE versus strict preservation of native conditions for Native PAGE. The choice between them should be driven by research goals—molecular weight determination and purity assessment favor SDS-PAGE, while functional studies and complex analysis require Native PAGE. By meticulously controlling buffer composition, handling conditions, and loading parameters, researchers can ensure reliable, reproducible results that advance understanding of protein structure and function.
Head-to-Head Comparison: Validating Your Choice of Technique
Polyacrylamide Gel Electrophoresis (PAGE) is a foundational technique in biochemistry for separating protein mixtures. The choice between its two primary forms, SDS-PAGE and Native PAGE, profoundly impacts the experimental outcome, influencing the type of data obtained and the downstream applications possible. [9] SDS-PAGE denatures proteins to separate them by molecular weight, while Native PAGE preserves proteins in their native, functional state, separating them based on a combination of size, charge, and shape. [6] [3] This guide provides a direct, data-driven comparison of these two techniques to help researchers select the optimal method for their protein analysis research.
Core Principles and Direct Technical Comparison
In SDS-PAGE, the anionic detergent Sodium Dodecyl Sulfate (SDS) denatures proteins and binds to them in a constant ratio, imparting a uniform negative charge. [59] [3] This masks the proteins' intrinsic charge, and separation occurs primarily based on polypeptide chain length and molecular weight as they migrate through a polyacrylamide gel matrix. [3] In contrast, Native PAGE is performed in the absence of denaturing agents. Proteins remain in their folded, native conformation, and their migration through the gel depends on their intrinsic charge, size, and three-dimensional shape. [6] [9] This allows the study of functional protein complexes and their interactions.
The table below summarizes the critical differences between the two techniques.
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight (mass) of polypeptide chains [6] [3] | Native size, intrinsic charge, and 3D shape of the protein [6] [9] |
| Gel Conditions | Denaturing gel containing SDS [6] | Non-denaturing gel; no SDS present [6] |
| Sample Preparation | Heated (70-100°C) in sample buffer with SDS and often a reducing agent (e.g., DTT, β-mercaptoethanol) [6] [59] [3] | Not heated; prepared in non-denaturing buffer without reducing agents [6] |
| Protein State | Denatured and linearized [6] | Native, folded conformation [6] [3] |
| Protein Function Post-Separation | Lost [6] | Retained [6] [3] |
| Protein Recovery & Downstream Use | Typically cannot be recovered in a functional state; ideal for western blotting and mass spectrometry [6] [9] | Can be recovered in active form for functional assays (e.g., activity studies, interaction analyses) [6] [9] [3] |
| Net Charge on Proteins | Uniformly negative due to SDS binding [59] [3] | Based on intrinsic charge (can be positive or negative) [6] |
| Primary Applications | Determine molecular weight, check purity/expression, protein profiling [6] [18] | Study protein complexes, oligomerization, native function, and enzymatic activity [6] [9] |
Experimental Protocols and Workflows
SDS-PAGE Standard Protocol
The following workflow, developed by Laemmli, is a standard discontinuous SDS-PAGE protocol. [59]
1. Gel Preparation: A polyacrylamide gel is cast in two layers: a resolving gel (typically pH 8.8, 10-12% acrylamide) where protein separation occurs, and a stacking gel (pH 6.8, 4-6% acrylamide) that concentrates the protein samples into a sharp band before they enter the resolving gel. [59] [3] The polymerization reaction is catalyzed by TEMED and initiated by Ammonium Persulfate (APS). [59] [3]
2. Sample Preparation: Protein samples are mixed with an SDS-containing sample buffer, which includes a reducing agent like Dithiothreitol (DTT) or β-mercaptoethanol to break disulfide bonds. [59] [18] The mixture is then heated to 95°C for 5 minutes to fully denature the proteins. [59]
3. Electrophoresis: The prepared samples and a molecular weight marker are loaded into wells. A voltage (e.g., 100V for a mini-gel) is applied, causing proteins to migrate toward the anode. The run is often tracked with a dye like bromophenol blue. [59]
4. Post-Electrophoresis Analysis: After separation, proteins are visualized using stains like Coomassie Brilliant Blue or more sensitive fluorescent stains. Gels can also be used for western blotting or, with advanced techniques like PEPPI-MS, intact proteins can be recovered from the gel for top-down mass spectrometry. [59] [68]
SDS-PAGE Workflow: A denaturing process where samples are heated with SDS and DTT before separation by molecular weight.
Native PAGE Standard Protocol
The Native PAGE protocol differs critically in its sample and gel conditions to preserve protein structure. [6] [3]
1. Gel Preparation: A polyacrylamide gel is cast without SDS or other denaturants. The buffer system is chosen to maintain a pH that preserves protein native state and activity (often cooler temperatures, like 4°C, are used during the run to minimize denaturation). [6] [3]
2. Sample Preparation: The protein sample is mixed with a non-denaturing sample buffer that contains no SDS, detergents, or reducing agents. The sample is not heated prior to loading. [6]
3. Electrophoresis: Samples are loaded and a voltage is applied. Because proteins retain their native charge, the buffer system and pH are critical for ensuring correct migration direction (toward the anode for negatively charged proteins). The gel apparatus is often kept cool. [6] [3]
4. Post-Electrophoresis Analysis: Proteins can be visualized with specific activity stains if enzymatic, or with gentle staining methods. A key advantage is that proteins can be recovered from the gel via passive diffusion or electro-elution for downstream functional studies. [6] [3]
Native PAGE Workflow: A non-denaturing process that maintains protein structure and function.
Research Reagent Solutions
The table below details essential reagents and materials required for executing SDS-PAGE and Native PAGE experiments.
| Reagent/Material | Function/Purpose | Key Considerations |
|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked polyacrylamide gel matrix that acts as a molecular sieve. [3] | The concentration (%T) determines pore size, affecting the separation range of protein sizes. [3] |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers a uniform negative charge. [59] | Essential for SDS-PAGE; must be absent in Native PAGE. Binds ~1.4g per 1g of protein. [59] |
| TEMED & APS | Catalytic system (TEMED) and radical initiator (APS) for polymerizing acrylamide gel. [59] [3] | Amounts and temperature affect polymerization speed and gel properties. [59] |
| DTT or β-Mercaptoethanol | Reducing agents that break disulfide bonds in proteins. [59] [18] | Used in reducing SDS-PAGE to fully dissociate subunits. Omitted in non-reducing SDS-PAGE and Native PAGE. [6] [18] |
| Tris-based Buffers | Provides the ionic environment and pH for electrophoresis. [59] | Discontinuous systems use different pH in stacking (pH 6.8) and resolving (pH 8.8) gels for SDS-PAGE. [59] |
| Coomassie Brilliant Blue | Reversible dye for staining proteins post-electrophoresis. [68] | Also acts as an extraction enhancer for recovering intact proteins from gels in PEPPI-MS. [68] |
| Molecular Weight Markers | A set of pre-defined proteins of known size run alongside samples. [59] [3] | Allows estimation of the molecular weight of unknown proteins in SDS-PAGE. [59] |
Key Experimental Data and Interpretation
The distinct separation principles of SDS-PAGE and Native PAGE lead to different but complementary data, crucial for inferring protein properties.
A classic example involves determining a protein's quaternary structure. Consider a scenario where a protein migrates as a 60 kDa band on non-reducing SDS-PAGE but migrates as a 120 kDa species on Native PAGE. [10]
- Inference: The protein is a dimer of 60 kDa subunits that are not linked by disulfide bonds. [10]
- Rationale: SDS-PAGE disrupts non-covalent interactions, dissociating the native dimer into its 60 kDa monomers. The use of "non-reducing" conditions means disulfide bonds remain intact; since it still runs as a monomer, the subunits are held together by non-covalent forces. In Native PAGE, the protein remains a folded 120 kDa dimer, and its migration is influenced by its larger native size and intrinsic charge. [10]
This contrast highlights how the techniques are used together to understand protein composition beyond what either can reveal alone. Furthermore, while SDS-PAGE is renowned for its denaturing conditions, certain robust proteins like Fluorescent Proteins (FPs) can retain their fluorescence even during SDS-PAGE with minor protocol adaptations, enabling direct in-gel fluorescence detection without the need for western blotting. [69]
SDS-PAGE and Native PAGE are complementary pillars of protein analysis. The choice is dictated by the research question: SDS-PAGE is the definitive tool for determining molecular weight, assessing purity, and analyzing denatured proteins, while Native PAGE is indispensable for probing native structure, protein-protein interactions, and enzymatic function. [6] [9] By understanding their distinct separation bases, gel conditions, and protein recovery outcomes, researchers can strategically apply these techniques to advance their work in biochemistry, proteomics, and drug development.
In the fields of biochemistry and molecular biology, polyacrylamide gel electrophoresis (PAGE) serves as a fundamental technique for protein separation, yet researchers must often choose between two divergent methodologies: SDS-PAGE and Native PAGE. These techniques represent fundamentally different approaches to protein analysis, each with distinct advantages and limitations that dictate their appropriateness for specific research scenarios. SDS-PAGE, or sodium dodecyl sulfate-polyacrylamide gel electrophoresis, employs denaturing conditions to separate proteins primarily by molecular weight, while Native PAGE maintains proteins in their natural, folded state, enabling separation based on combined factors of size, charge, and shape [9] [6]. This guide provides a comprehensive comparison framework, supported by experimental data and procedural details, to assist researchers in selecting the optimal electrophoretic method for their specific protein analysis requirements.
The critical distinction between these techniques lies in their treatment of protein structure. SDS-PAGE utilizes the anionic detergent SDS to denature proteins, mask their intrinsic charge, and unfold them into linear chains, ensuring separation occurs almost exclusively based on molecular mass [9] [59]. In contrast, Native PAGE preserves the tertiary and quaternary structures of proteins, maintaining their biological activity and enabling the study of functional complexes and interactions [9] [3]. This fundamental difference dictates their applications in research, with SDS-PAGE excelling in molecular weight determination and purity assessment, while Native PAGE proves invaluable for studying native protein complexes, conformational states, and enzymatic activities.
Fundamental Principles: How SDS-PAGE and Native PAGE Work
The Mechanism of SDS-PAGE
SDS-PAGE operates on the principle of complete protein denaturation to achieve separation by molecular weight. The technique employs sodium dodecyl sulfate (SDS), an anionic detergent that binds uniformly to polypeptide chains in a constant weight ratio of approximately 1.4 grams of SDS per 1 gram of protein [59] [3]. This binding confers a uniform negative charge density to all proteins, effectively masking their intrinsic electrical charges. The proteins are typically heated in the presence of SDS and reducing agents like β-mercaptoethanol or dithiothreitol (DTT) to disrupt hydrogen bonds and cleave disulfide linkages, resulting in fully denatured, linear polypeptides [59] [6].
During electrophoresis, these SDS-polypeptide complexes migrate through a polyacrylamide gel matrix toward the anode when an electric field is applied. The gel acts as a molecular sieve, with smaller polypeptides moving more rapidly through the pores while larger ones are retarded [59] [3]. The discontinuous buffer system, typically utilizing Tris-glycine-chloride with differing pH in stacking and separating gels, creates a stacking effect that concentrates proteins into sharp bands before they enter the separating gel, thereby enhancing resolution [59]. The resulting separation enables accurate molecular weight estimation when compared with standardized protein ladders, making SDS-PAGE exceptionally valuable for assessing protein purity, subunit composition, and expression levels.
The Mechanism of Native PAGE
Native PAGE employs a fundamentally different approach by maintaining proteins in their native, folded conformation throughout the separation process. Without denaturing agents, proteins retain their higher-order structure, including secondary, tertiary, and quaternary arrangements [9] [3]. Consequently, separation depends on both the intrinsic charge of the protein at the running buffer pH and the protein's size and three-dimensional shape [6] [3]. In this technique, proteins migrate according to their charge-to-mass ratio, with more negatively charged proteins moving faster toward the anode, while the gel matrix provides sieving based on hydrodynamic size [9].
The preservation of native structure allows Native PAGE to maintain protein functionality, enabling researchers to recover active proteins after separation [6]. This characteristic makes the technique particularly valuable for studying functional attributes, including enzymatic activity, protein-protein interactions, and oligomerization states [9]. Variants of Native PAGE include Blue Native PAGE (BN-PAGE), which utilizes Coomassie brilliant blue dye to impart charge for separation, and Clear Native PAGE (CN-PAGE), which relies on the proteins' intrinsic charge in a gradient gel without dye [6]. The maintenance of native conformations comes at the cost of molecular weight determination accuracy, as migration depends on multiple factors beyond mass alone.
Figure 1: Decision Framework for PAGE Technique Selection
Technical Comparison: Methodologies and Buffer Systems
The practical implementation of SDS-PAGE and Native PAGE differs significantly in buffer composition, sample preparation, and electrophoretic conditions. These technical differences directly impact the type of information obtainable from each method and must be carefully considered during experimental design.
Table 1: Comparative Methodologies of SDS-PAGE versus Native PAGE
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| Gel Type | Denaturing gel [6] | Non-denaturing gel [6] |
| SDS Presence | Present in gel and buffers [59] [6] | Absent [6] |
| Reducing Agents | DTT or β-mercaptoethanol in buffer [6] | No reducing agents [6] |
| Sample Preparation | Heating at 70-100°C [59] [6] [3] | No heating [6] |
| Separation Basis | Molecular weight [9] [6] [3] | Size, charge, and shape [9] [6] [3] |
| Protein Net Charge | Always negative (from SDS) [59] [6] | Positive or negative (intrinsic) [6] |
| Typical Running Temperature | Room temperature [6] | 4°C [6] |
| Protein State | Denatured [9] [6] | Native conformation/folded [9] [6] |
| Protein Recovery & Function | Cannot be recovered functionally [6] | Can be recovered with function intact [6] |
| Primary Applications | Molecular weight determination, purity assessment, expression analysis [6] | Structure, subunit composition, functional studies [9] [6] |
The buffer systems for these techniques reflect their divergent purposes. SDS-PAGE employs buffers containing SDS (typically 0.1-0.2%) and may include EDTA, while Native PAGE buffers lack detergents and may utilize different buffer systems like Bis-Tris at neutral pH [7]. Sample preparation fundamentally differs, with SDS-PAGE requiring heating samples to 70-100°C in sample buffer containing SDS and reducing agents, while Native PAGE uses mild buffers without heating to preserve native structure [6] [3]. These methodological differences extend to running conditions, with Native PAGE often performed at 4°C to maintain protein stability, while SDS-PAGE is typically run at room temperature [6].
Experimental Protocols: Step-by-Step Methodologies
SDS-PAGE Experimental Protocol
Gel Preparation: The standard SDS-PAGE protocol begins with preparation of a discontinuous gel system consisting of a stacking gel (4-6% acrylamide, pH ~6.8) and a separating gel (8-15% acrylamide, pH ~8.8) [59]. The gel solution is formed by mixing acrylamide and bisacrylamide cross-linker in appropriate ratios, with polymerization catalyzed by ammonium persulfate (APS) and TEMED [59] [3]. The separating gel is poured first and overlayered with a water-immiscible alcohol (e.g., isopropanol) to ensure a flat surface, followed after polymerization by the stacking gel into which a sample comb is inserted to form wells [59].
Sample Preparation: Protein samples are mixed with SDS-PAGE sample buffer (typically containing Tris-HCl, SDS, glycerol, bromophenol blue, and reducing agents like DTT or β-mercaptoethanol) [59]. The mixture is heated to 95°C for 5 minutes or 70°C for 10 minutes to denature proteins [59]. For molecular weight estimation, standardized protein markers are prepared similarly.
Electrophoresis: The prepared gel is placed in an electrophoresis chamber filled with running buffer (typically Tris-glycine with 0.1% SDS) [59]. Denatured samples are loaded into wells, and electrophoresis is performed at constant voltage (100-200V) until the dye front approaches the gel bottom [59]. The process typically requires 30-90 minutes depending on gel size and voltage [59].
Detection: Following electrophoresis, proteins are visualized using stains such as Coomassie Brilliant Blue, silver stain, or fluorescent dyes [59]. For downstream analysis like western blotting, proteins are transferred to membranes electrophoretically [9].
Native PAGE Experimental Protocol
Gel Preparation: Native PAGE utilizes similar polyacrylamide gel matrices but without SDS or other denaturants [6]. Both continuous and discontinuous buffer systems may be employed, with the latter using different pH in stacking and separating gels similar to SDS-PAGE but without SDS [3]. The polymerization process is identical, using acrylamide, bisacrylamide, APS, and TEMED [3].
Sample Preparation: Protein samples are mixed with native sample buffer (typically containing Tris, glycerol, and a tracking dye like bromophenol blue) but without SDS or reducing agents [6] [7]. Critically, samples are not heated, and conditions are maintained to preserve protein structure [6]. Buffer composition may be adjusted to maintain protein stability, with particular attention to pH and ionic strength.
Electrophoresis: The gel is placed in an electrophoresis apparatus filled with appropriate running buffer (e.g., Tris-glycine without SDS) [6] [3]. Samples are loaded, and electrophoresis is typically performed at 4°C to maintain protein stability, with constant voltage applied until adequate separation is achieved [6]. Run times may be longer than SDS-PAGE due to the maintained protein structure creating more resistance during migration.
Detection and Recovery: Proteins are detected using standard staining methods, though milder staining conditions may be employed if functional recovery is desired [6]. Following electrophoresis, native proteins can be recovered from gels through passive diffusion, electroelution, or extraction buffers designed to maintain activity [3].
Research Applications and Performance Data
The choice between SDS-PAGE and Native PAGE significantly impacts the type and quality of information obtained from proteomic studies. Experimental data demonstrates that these techniques offer complementary strengths that can be strategically leveraged based on research objectives.
Table 2: Comparative Performance Data from Proteomic Studies
| Performance Metric | SDS-PAGE | Native PAGE | Experimental Context |
|---|---|---|---|
| Proteins Identified | 2552 proteins | 4323 proteins | Analysis of supernatant fraction from human bronchial smooth muscle cells [27] |
| Protein Abundance Range | 3.5% to 2×10^-4% | 3.6% to 1×10^-5% | Same study as above [27] |
| Advantage in Quantitative Comparison | Advantageous | Less suitable | SDS-PAGE better for visualizing quantity differences between samples [27] |
| Advantage in Interaction Studies | Limited | Advantageous | Native PAGE better for visualizing protein interactions [27] |
| Metal Ion Retention | 26% Zn²⁺ retention | 98% Zn²⁺ retention | Analysis of Zn-metalloproteins using modified NSDS-PAGE [7] |
| Enzymatic Activity Retention | None of 9 model enzymes | 7 of 9 model enzymes | Activity test after NSDS-PAGE separation [7] |
A 2019 study comparing 1D SDS-PAGE with nondenaturing 2DE for analysis of proteins from human bronchial smooth muscle cells provided compelling quantitative data on their relative performances [27]. When combined with quantitative LC-MS/MS analysis, nondenaturing methods identified substantially more proteins (4323 versus 2552) from the supernatant fraction and detected proteins across a wider abundance range [27]. The study concluded that SDS-PAGE offered advantages in comparative quantitation of specific proteins across differently treated samples, while native methods excelled in visualizing protein interactions [27].
Research on metalloprotein analysis demonstrated dramatic differences in metal retention between techniques. A study developing Native SDS-PAGE (NSDS-PAGE) - a hybrid approach - found that zinc retention increased from 26% with standard SDS-PAGE to 98% with the modified conditions [7]. Similarly, enzymatic activity preservation differed significantly, with all nine model enzymes losing activity after standard SDS-PAGE, while seven retained function after NSDS-PAGE separation [7]. These findings highlight the critical importance of technique selection for functional studies.
Research Reagent Solutions: Essential Materials for PAGE Experiments
Successful implementation of either SDS-PAGE or Native PAGE requires specific reagent systems tailored to each method's requirements. The following table summarizes key reagents and their functions in electrophoretic separations.
Table 3: Essential Research Reagents for PAGE Experiments
| Reagent | Function | SDS-PAGE | Native PAGE |
|---|---|---|---|
| Acrylamide-Bis | Forms porous gel matrix for separation | Required | Required |
| SDS | Denatures proteins and confers uniform charge | Required [59] [6] | Not used [6] |
| DTT/β-Mercaptoethanol | Reduces disulfide bonds | Required [59] [6] | Not used [6] |
| TEMED/APS | Catalyzes gel polymerization | Required [59] [3] | Required |
| Tris-Based Buffers | Maintains pH during electrophoresis | pH 6.8 (stacking) and 8.8 (separating) [59] | Varies, often neutral pH [7] |
| Coomassie Blue | Protein staining | Used [59] | Used (BN-PAGE employs in buffer) [6] [7] |
| Molecular Weight Markers | Size reference standards | Required [59] [3] | Optional (different migration principles) |
| Glycerol | Increases sample density for loading | Used in sample buffer [59] | Used in sample buffer [7] |
| Tracking Dye | Visualizes migration progress | Bromophenol blue [59] | Bromophenol blue or other [7] |
The reagent requirements reflect the different objectives of each technique. SDS-PAGE necessitates strong denaturants and reducing agents to achieve complete unfolding and charge uniformity, while Native PAGE employs milder conditions that maintain structural integrity [6]. Specialized variants like Blue Native PAGE incorporate Coomassie dye directly into the running buffer to impart charge to proteins while maintaining native structure [6] [7]. Understanding these reagent functions enables researchers to optimize protocols for specific protein systems and research goals.
Advanced Applications and Hybrid Approaches
The divergence between denaturing and native electrophoretic techniques has led to the development of specialized applications and hybrid approaches that leverage the strengths of both methods. Understanding these advanced applications expands the researcher's toolkit for addressing complex protein analysis challenges.
Two-dimensional electrophoresis (2D-PAGE) represents a powerful approach that combines the principles of both native and denaturing separation. In one application, nondenaturing 2DE in the first dimension separates protein complexes based on their intrinsic charge and size, followed by denaturing SDS-PAGE in the second dimension to resolve individual subunits [27]. This approach enables researchers to study protein interaction networks while still obtaining information about component polypeptides [27]. The technique has proven valuable for analyzing complex cellular systems, with one study identifying over 4300 proteins from human bronchial smooth muscle cells while simultaneously providing information about their interaction states [27].
The development of Native SDS-PAGE (NSDS-PAGE) demonstrates how hybrid approaches can bridge the gap between technique extremes. This method reduces SDS concentration in running buffers from 0.1% to 0.0375%, eliminates EDTA and heating steps, and modifies sample buffer composition [7]. The resulting protocol maintains high resolution separation while dramatically improving retention of metal ions (98% versus 26% for Zn²⁺ in standard SDS-PAGE) and preserving enzymatic activity in most tested enzymes [7]. Such hybrid approaches offer promising avenues for applications requiring both good resolution and maintenance of certain functional properties.
Figure 2: Advanced Electrophoresis Workflows and Applications
The choice between SDS-PAGE and Native PAGE represents a critical decision point in experimental design that directly determines the type and quality of information obtainable from protein studies. SDS-PAGE remains the gold standard for molecular weight determination, purity assessment, and comparative quantification, offering robust, reproducible separation based primarily on polypeptide size [9] [27]. Its denaturing nature, while destroying functional properties, provides exceptional resolution and compatibility with downstream techniques like western blotting and mass spectrometry [9]. Conversely, Native PAGE preserves protein structure and function, enabling studies of protein complexes, interactions, and enzymatic activities that are impossible with denaturing methods [9] [27].
The experimental data presented in this guide underscores their complementary nature. SDS-PAGE identified approximately 2552 proteins in comparative studies, while native methods detected over 4300 proteins from the same source, though with different quantitative capabilities [27]. The dramatic difference in metal retention (26% versus 98%) and enzymatic activity preservation further highlights the functional consequences of technique selection [7]. Researchers must therefore align their choice with specific research questions: SDS-PAGE for structural characterization and quantification, Native PAGE for functional studies and interaction mapping. Emerging hybrid approaches like NSDS-PAGE and sophisticated 2D systems that combine both principles offer promising avenues for comprehensive protein analysis that captures both structural and functional information [27] [7]. Through strategic selection and implementation of these electrophoretic tools, researchers can optimize their experimental outcomes and advance our understanding of protein systems.
In protein analysis research, the choice of electrophoresis method is fundamental to data interpretation and validation. Polyacrylamide Gel Electrophoresis (PAGE) serves as a cornerstone technique, with Sodium Dodecyl Sulfate-PAGE (SDS-PAGE) and Native PAGE representing two principal approaches with distinct capabilities and limitations [9] [3]. SDS-PAGE employs denaturing conditions to separate proteins based primarily on molecular weight, providing high-resolution separation that enables precise size determination and purity assessment [3] [47]. In contrast, Native PAGE maintains proteins in their folded, functional state, separating them based on a combination of molecular size, intrinsic charge, and three-dimensional structure [9] [3]. This fundamental difference in methodology directly impacts the type of biological information that can be extracted, the validation strategies required, and how results correlate with downstream analytical techniques.
The broader context for comparing these techniques lies in the growing need for orthogonal validation in protein characterization, particularly in pharmaceutical development and basic research where understanding both structural and functional properties is essential [7] [70]. This guide provides an objective comparison of SDS-PAGE versus Native PAGE performance, supported by experimental data and detailed methodologies, to enable researchers to select appropriate techniques and validate findings through correlation with complementary approaches.
Technical Comparison: Separation Mechanisms and Data Outputs
Fundamental Principles and Separation Mechanisms
The separation mechanisms of SDS-PAGE and Native PAGE dictate the types of biological information they can reveal. In SDS-PAGE, the anionic detergent SDS denatures proteins by disrupting non-covalent bonds and uniformly coats the polypeptide backbone with negative charges [3] [5]. This process masks proteins' intrinsic charge and eliminates structural influences, resulting in separation driven almost exclusively by molecular size as proteins migrate through the polyacrylamide gel matrix [9] [47]. The denaturation is typically enhanced with reducing agents like β-mercaptoethanol or dithiothreitol (DTT) to break disulfide bonds, ensuring complete unfolding of protein subunits [3] [5].
Conversely, Native PAGE employs non-denaturing conditions without SDS or reducing agents, preserving proteins' native conformations and biological activities [9] [3]. Separation occurs based on the protein's intrinsic charge, size, and three-dimensional structure, with the gel matrix providing a sieving effect that regulates movement according to these combined properties [3]. This preservation of native structure allows for the analysis of protein complexes, oligomeric states, and functional attributes that are destroyed under denaturing conditions [9].
Comparative Performance Data and Applications
Table 1: Quantitative Comparison of SDS-PAGE and Native PAGE Performance Characteristics
| Parameter | SDS-PAGE | Native PAGE | Experimental Basis |
|---|---|---|---|
| Molecular Weight Resolution | High resolution for proteins separated by size; can distinguish proteins with small molecular weight differences [47] | Lower resolution for molecular weight determination due to influence of charge and shape [7] | Comparative analysis of proteomic mixtures [7] |
| Functional Activity Retention | Enzymatic activity destroyed in all cases; 0% activity retention reported [7] | High activity retention; 100% of tested enzymes (9/9) remained active [7] | In-gel activity assays with model enzymes [7] |
| Metal Cofactor Retention | Minimal retention (26% for Zn²⁺) [7] | High metal retention (98% for Zn²⁺) demonstrated [7] | Metal analysis via LA-ICP-MS and TSQ staining [7] |
| Protein Complex Preservation | Disrupts non-covalent interactions; separates subunits individually [9] | Maintains oligomeric structures and protein-protein interactions [9] [71] | Analysis of OXPHOS complexes and supercomplexes [71] |
| Typical Separation Range | 10-250 kDa [22] | Variable based on native charge and structure | Standard protocol specifications [22] |
| Quantitative Capability | Limited; qualitative and semi-quantitative based on staining intensity [47] | Limited; qualitative assessment of native states [3] | Densitometry analysis with staining variability [47] |
Table 2: Application-Based Technique Selection Guide
| Research Application | Recommended Technique | Rationale | Validation Approaches |
|---|---|---|---|
| Molecular Weight Determination | SDS-PAGE | Provides accurate size estimation based on polypeptide length [3] [5] | Compare with protein standards/ladders [3] |
| Enzymatic Activity Studies | Native PAGE | Preserves catalytic function and cofactor binding [9] [7] | In-gel activity staining [7] [71] |
| Protein-Protein Interactions | Native PAGE or BN-PAGE | Maintains quaternary structure and complex formation [9] [71] | Follow with 2D SDS-PAGE or western blot [71] |
| Purity Assessment | SDS-PAGE | High resolution reveals contaminating proteins [9] [47] | Densitometry analysis of single versus multiple bands [5] |
| Metalloprotein Analysis | Native PAGE or NSDS-PAGE | Retains bound metal ions essential for function [7] | LA-ICP-MS or specific metal staining [7] |
| Membrane Protein Complexes | BN-PAGE | Specialized native method for hydrophobic complexes [71] | In-gel activity staining or 2D electrophoresis [71] |
| Therapeutic Antibody Characterization | SDS-PAGE or CE-SDS | Assess purity, fragmentation, and glycosylation variants [70] | Capillary electrophoresis correlation [70] |
Experimental Protocols and Methodologies
Standard SDS-PAGE Protocol
The following denaturing SDS-PAGE protocol represents the widely adopted methodology for protein separation by molecular weight, adapted from standard laboratory procedures [3] [5]:
Sample Preparation:
- Combine protein sample with SDS-PAGE sample buffer (typically containing 2% SDS, 50-100 mM Tris-HCl pH 6.8, 10% glycerol, 0.02% bromophenol blue) [3] [5].
- Add reducing agent (100 mM DTT or 5% β-mercaptoethanol) to break disulfide bonds [3].
- Heat samples at 70-100°C for 5-10 minutes to ensure complete denaturation [7] [3].
- Centrifuge briefly to collect condensed samples before loading.
Gel Preparation:
- Prepare resolving gel with appropriate acrylamide concentration (7-20% based on target protein size range) in Tris-HCl buffer pH 8.8, containing 0.1% SDS [3] [5].
- Polymerize with ammonium persulfate (APS) and tetramethylethylenediamine (TEMED) [3].
- Prepare stacking gel (4-5% acrylamide) in Tris-HCl buffer pH 6.8 and layer on top of polymerized resolving gel [3] [5].
- Insert well comb and allow to polymerize completely.
Electrophoresis:
- Assemble gel in electrophoresis chamber filled with running buffer (25 mM Tris, 192 mM glycine, 0.1% SDS, pH 8.3) [3].
- Load prepared samples and molecular weight markers into wells.
- Apply constant voltage (100-200V) until dye front reaches bottom of gel [7] [3].
- Proceed with protein detection by staining (Coomassie, silver stain) or western blotting [3] [5].
Standard Native PAGE Protocol
This non-denaturing Native PAGE protocol preserves protein structure and function, adapted from established methodologies [9] [3]:
Sample Preparation:
- Mix protein sample with native sample buffer (typically containing 50 mM Tris-HCl pH 6.8, 10% glycerol, 0.02% bromophenol blue) [3].
- Omit SDS, reducing agents, and heating steps to maintain native structure [9] [7].
- Keep samples on ice to prevent denaturation or proteolysis.
Gel Preparation:
- Prepare resolving gel with appropriate acrylamide concentration (6-10% typically used) in Tris-HCl buffer pH 8.8, without SDS [3].
- Polymerize with APS and TEMED as with SDS-PAGE.
- Prepare stacking gel (3-4% acrylamide) in Tris-HCl buffer pH 6.8 without SDS.
- Complete polymerization as with standard SDS-PAGE protocol.
Electrophoresis:
- Use native running buffer (25 mM Tris, 192 mM glycine, pH 8.3) without SDS [3].
- Load prepared samples and appropriate native molecular weight standards.
- Run electrophoresis at constant voltage (100-150V) at 4°C to maintain protein stability [3].
- For activity staining, proceed directly to appropriate detection methods without fixing [7] [71].
Advanced Methodologies: Native SDS-PAGE (NSDS-PAGE)
A hybrid approach called Native SDS-PAGE (NSDS-PAGE) has been developed to balance the benefits of both techniques, offering high resolution while preserving significant functional activity [7] [72]:
Modified Sample Preparation:
- Use sample buffer without SDS or EDTA [7].
- Eliminate the heating step to prevent denaturation [7].
- Optional addition of Coomassie G-250 (0.0185%) to enhance protein solubility and migration [7].
Modified Electrophoresis Conditions:
- Reduce SDS concentration in running buffer to 0.0375% (compared to standard 0.1%) [7].
- Remove EDTA from running buffer to preserve metal cofactors [7].
- Use standard precast Bis-Tris gels with modified buffer system [7].
Validation Data:
- This modified approach demonstrated retention of 98% of bound Zn²⁺ compared to 26% with standard SDS-PAGE [7].
- Seven of nine model enzymes tested retained activity after NSDS-PAGE separation, compared to complete denaturation with standard SDS-PAGE [7].
- Resolution comparable to standard SDS-PAGE was maintained for proteomic samples [7].
Data Validation and Correlation with Complementary Techniques
Orthogonal Validation Approaches
Validating electrophoretic data requires correlation with complementary analytical techniques to confirm findings and extract comprehensive biological insights:
Western Blot Analysis:
- Following both SDS-PAGE and Native PAGE, western blotting provides specific identification of target proteins using antibodies [9] [71].
- For Native PAGE, western blotting confirms identity while preserving information about native complexes [71].
- Standard protocol: Transfer proteins from gel to membrane, block with protein solution, incubate with primary antibody, detect with labeled secondary antibody [9].
Mass Spectrometry Identification:
- Excise protein bands from gels for identification by mass spectrometry [9].
- For SDS-PAGE: Bands typically represent individual polypeptides; identification is straightforward [9].
- For Native PAGE: Bands may represent complexes; additional separation by 2D electrophoresis may be needed before MS analysis [71].
In-Gel Enzyme Activity Staining:
- Exclusive to Native PAGE due to preservation of catalytic function [7] [71].
- Specific staining protocols exist for dehydrogenases, phosphatases, oxidases, and other enzyme classes [71].
- Directly confirms functional integrity after separation [7].
Two-Dimensional Electrophoresis:
- Combines Native PAGE or BN-PAGE in first dimension with SDS-PAGE in second dimension [71].
- Powerful approach for analyzing complex protein mixtures and subunit composition [71].
- Reveals both native complex organization and individual subunit identities [71].
Technique Correlation in Biopharmaceutical Analysis
In biopharmaceutical applications, correlation between electrophoresis platforms is essential for method validation and technology transfer:
SDS-PAGE and CE-SDS Correlation:
- Capillary electrophoresis SDS (CE-SDS) has emerged as an automated, quantitative alternative to traditional SDS-PAGE [70].
- Both techniques separate primarily by molecular size under denaturing conditions [70].
- Important differences exist: CE-SDS shows substantially reduced electrophoretic mobility for glycoproteins and reversed migration order between reduced and nonreduced proteins compared to SDS-PAGE [70].
- Method correlation studies are essential when implementing CE-SDS to replace SDS-PAGE in quality control workflows [70].
Native PAGE and BN-PAGE Specialized Applications:
- Blue Native PAGE (BN-PAGE) represents a specialized native technique particularly valuable for membrane protein complexes [71].
- BN-PAGE uses Coomassie G-250 to impart charge shift while maintaining native structure [71].
- Clear Native PAGE (CN-PAGE) eliminates the blue dye to reduce interference with downstream activity assays [71].
- These specialized native methods provide superior resolution for hydrophobic complexes like mitochondrial OXPHOS proteins [71].
Essential Research Reagents and Materials
Table 3: Research Reagent Solutions for Protein Electrophoresis
| Reagent/Category | Function/Purpose | Specific Examples | Technique Application |
|---|---|---|---|
| Detergents | Solubilize and denature proteins; impart charge | SDS (sodium dodecyl sulfate), Dodecyl-β-D-maltoside, Digitonin | SDS-PAGE (SDS), BN-PAGE (mild detergents) [7] [71] |
| Reducing Agents | Break disulfide bonds for complete denaturation | β-mercaptoethanol, Dithiothreitol (DTT), Tris(2-carboxyethyl)phosphine (TCEP) | SDS-PAGE (essential), Native PAGE (omitted) [3] [5] |
| Buffer Systems | Maintain pH and provide conducting medium | Tris-glycine, Bis-Tris, MOPS, HEPES | All PAGE variations [7] [3] |
| Staining Reagents | Visualize separated proteins | Coomassie Brilliant Blue, Silver stain, SYPRO Ruby, Fluorescent dyes | All PAGE variations (different sensitivities) [3] [5] |
| Molecular Standards | Reference for size estimation | Pre-stained protein ladders, Unstained protein standards, NativeMark standards | SDS-PAGE (denatured standards), Native PAGE (native standards) [3] |
| Activity Stain Reagents | Detect enzymatic function in gels | Tetrazolium salts, Naphthol derivatives, Specific chromogenic substrates | Native PAGE exclusively [7] [71] |
| Transfer Reagents | Move proteins to membranes for blotting | Methanol, Transfer buffers, Nitrocellulose/PVDF membranes | Western blotting after both techniques [9] |
The comparative analysis of SDS-PAGE and Native PAGE reveals complementary strengths that serve different research objectives in protein characterization. SDS-PAGE remains the gold standard for molecular weight determination, purity assessment, and subunit composition analysis due to its high resolution and predictable migration based primarily on polypeptide size [9] [47]. Conversely, Native PAGE provides unique insights into native protein structure, functional activity, protein-protein interactions, and metal cofactor retention that are destroyed under denaturing conditions [9] [7].
Strategic technique selection depends fundamentally on the research questions being addressed. For routine size-based separation and purity analysis, SDS-PAGE offers simplicity, reproducibility, and straightforward interpretation [47]. For functional studies, complex analysis, and metalloprotein characterization, Native PAGE preserves biological context at the cost of more complex migration patterns [9] [7]. The emerging Native SDS-PAGE (NSDS-PAGE) methodology demonstrates that hybrid approaches can balance resolution with functional preservation, retaining significant enzymatic activity and metal cofactors while maintaining high-resolution separation [7] [72].
Robust data validation requires correlation with orthogonal techniques, with western blotting, mass spectrometry, and activity assays providing complementary verification of electrophoretic results [9] [7] [71]. In pharmaceutical and industrial applications, understanding the correlation and limitations between traditional SDS-PAGE and emerging automated platforms like CE-SDS is essential for method validation and technology transfer [70]. Through strategic technique selection and comprehensive validation approaches, researchers can extract maximum biological insight from electrophoretic separations while ensuring data reliability and reproducibility.
Polyacrylamide gel electrophoresis (PAGE) is a foundational technique in biochemical research for separating and analyzing proteins. Among the various PAGE methods, Sodium Dodecyl Sulfate-PAGE (SDS-PAGE) and Native PAGE represent two fundamentally different approaches, each with distinct advantages and limitations. SDS-PAGE separates proteins primarily by molecular weight under denaturing conditions, while Native PAGE separates proteins based on their size, charge, and shape while preserving their native structure and function. For researchers and drug development professionals, selecting the appropriate method is critical for obtaining accurate, relevant data. This guide provides a balanced comparison of both techniques, supported by experimental data and protocols, to inform methodological choices in protein analysis research.
Principles of Separation: A Fundamental Distinction
The core difference between these techniques lies in their treatment of protein structure during separation, which directly dictates their applications and limitations.
SDS-PAGE Principle: This method uses the anionic detergent sodium dodecyl sulfate (SDS) to denature proteins. SDS binds uniformly to the polypeptide backbone in a constant weight ratio, masking the protein's intrinsic charge and unfolding it into a linear chain. Consequently, all proteins gain a uniform negative charge-to-mass ratio, and separation through the polyacrylamide gel matrix occurs almost exclusively based on molecular weight. Smaller proteins migrate faster than larger ones [9] [3] [15].
Native PAGE Principle: In contrast, Native PAGE is performed without denaturing agents. Proteins remain in their folded, native state, retaining their secondary, tertiary, and quaternary structures. Separation depends on the protein's intrinsic charge, size, and three-dimensional shape. The migration rate is determined by the protein's charge density and the frictional force it experiences from the gel matrix [6] [9] [15].
The diagram below illustrates the fundamental workflow differences between these two techniques.
Comparative Analysis: Advantages and Limitations
The different separation principles of SDS-PAGE and Native PAGE confer unique advantages and limitations, making each method suitable for distinct research objectives.
Table 1: Advantages and Limitations of SDS-PAGE and Native PAGE
| Aspect | SDS-PAGE | Native PAGE |
|---|---|---|
| Primary Application | Determining protein molecular weight, checking purity, analyzing subunit composition [6] [9] | Studying native protein structure, oligomerization, protein-protein interactions, and enzymatic activity [6] [9] |
| Key Advantages | - Simple, reproducible, and high-resolution for molecular weight determination [9] [3]- Applicable to almost any protein source [3]- Ideal for western blotting [9] | - Preserves protein function and native conformation [6] [3]- Enables post-separation activity assays and protein recovery [6] [9]- Identifies oligomeric states and complexes [21] |
| Major Limitations | - Denatures proteins, eliminating biological activity [6] [9]- Cannot be used to study function or native interactions [9]- Protein recovery is difficult [6] | - Complex data interpretation due to multiple separation factors (charge, size, shape) [6] [9]- Lower resolution for closely sized proteins [9]- Requires careful optimization of conditions (pH, temperature) [6] [3] |
| Impact on Protein Structure | Disrupts secondary, tertiary, and quaternary structures; proteins are linearized [9] [3] | Preserves secondary, tertiary, and quaternary structures; proteins remain folded [6] [3] |
| Typical Running Conditions | Room temperature [6] | 4°C to prevent denaturation and proteolysis [6] [3] |
Experimental Protocols and Data Interpretation
SDS-PAGE Protocol
A standard SDS-PAGE protocol involves several key steps [3] [18]:
- Sample Preparation: Protein samples are mixed with a loading buffer containing SDS and a reducing agent (e.g., DTT or β-mercaptoethanol). This mixture is heated at 70-100°C for 5-10 minutes. SDS denatures the protein, while the reducing agent cleaves disulfide bonds [6] [3].
- Gel Preparation: A discontinuous gel system is used, comprising a stacking gel (lower acrylamide %, pH ~6.8) and a resolving gel (higher acrylamide %, pH ~8.8). The stacking gel concentrates proteins into a sharp band before they enter the resolving gel, where separation occurs [3].
- Electrophoresis: The prepared samples and molecular weight markers are loaded into wells. The gel is run at a constant voltage (e.g., 100-200 V) until the dye front reaches the bottom [3].
- Detection: Proteins are visualized by staining with Coomassie Blue, silver stain, or other dyes [3].
Native PAGE Protocol
The Native PAGE protocol differs significantly in its treatment of the sample [6] [3]:
- Sample Preparation: Protein samples are prepared in a non-denaturing buffer without SDS or reducing agents. The sample is not heated to preserve the native structure [6].
- Gel and Buffer System: The polyacrylamide gel and running buffers are formulated without SDS or other denaturants. The pH of the buffer is critical as it determines the intrinsic charge of the proteins [3].
- Electrophoresis: The gel is typically run at 4°C to maintain protein stability and prevent unfolding during the run [6].
- Detection and Analysis: Proteins can be detected by standard staining. For functional studies, in-gel activity assays can be performed. For example, an oxidoreductase enzyme can be detected by incubating the gel with its substrate and a colorimetric electron acceptor like nitro blue tetrazolium (NBT), which forms a purple precipitate where the enzyme is active [21].
Interpreting Results: A Case Study
The different outputs of these methods can be used in tandem to gain deep insights into protein structure. Consider a protein that migrates as a 60 kDa band on non-reducing SDS-PAGE but as a 120 kDa band on Native PAGE [10].
This discrepancy reveals that the protein is a dimer of two 60 kDa subunits in its native state. The fact that it runs as a monomer on non-reducing SDS-PAGE indicates that the subunits are held together by non-covalent interactions (e.g., hydrophobic or electrostatic forces) rather than disulfide bonds, as SDS disrupts these non-covalent interactions [10].
Advanced Applications and Specialized Techniques
Specialized Forms of Native PAGE
- Blue Native PAGE (BN-PAGE): Uses the dye Coomassie G-250, which binds to proteins and confers a negative charge shift. This allows separation primarily by size and is excellent for analyzing membrane protein complexes and estimating native molecular weights [42].
- Clear Native PAGE (CN-PAGE): A milder technique that does not use Coomassie dye, separating proteins based on their intrinsic charge. It is advantageous for detecting catalytic activities and labile supramolecular assemblies that might be disrupted by the dye in BN-PAGE [42] [21].
Applications in Food Science and Drug Development
- SDS-PAGE is extensively used in food science for protein profiling, allergen detection, and assessing quality and adulteration in products like cereals, dairy, and meat [18].
- Native PAGE, particularly in-gel activity assays, is powerful in drug development and disease research for characterizing the functional impact of protein variants. For instance, it has been used to study how pathogenic variants in the enzyme MCAD affect its tetrameric structure and activity, providing insights into the molecular basis of metabolic disease [21].
Essential Research Reagent Solutions
The following table details key reagents required for performing SDS-PAGE and Native PAGE experiments.
Table 2: Essential Reagents for PAGE Experiments
| Reagent | Function | Application in SDS-PAGE | Application in Native PAGE |
|---|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked polyacrylamide gel matrix that acts as a molecular sieve [3] | Required | Required |
| Ammonium Persulfate (APS) & TEMED | Catalyzes the polymerization of acrylamide to form the gel [3] | Required | Required |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers uniform negative charge [6] [3] | Essential | Not Used |
| Reducing Agents (DTT, BME) | Cleaves disulfide bonds to fully denature proteins [6] [18] | Used in reducing SDS-PAGE | Not Used |
| Coomassie Brilliant Blue | Dye used to stain proteins for visualization [3] | Used for post-run staining | Used for post-run staining; also used in BN-PAGE for charge-shift during run [42] |
| Molecular Weight Markers | Set of proteins of known size for calibrating the gel and estimating sample protein sizes [3] | Essential | Useful, but migration is not based on mass alone |
SDS-PAGE and Native PAGE are complementary, not competing, techniques in the protein researcher's toolkit. SDS-PAGE is the undisputed method for routine analysis of protein molecular weight, purity, and subunit composition under denaturing conditions. Its simplicity, robustness, and high resolution make it a foundational technique. Conversely, Native PAGE is indispensable for functional proteomics, providing a window into the native world of protein complexes, oligomeric states, and biological activity. Its ability to separate proteins in their folded state allows for downstream analyses that are impossible with denaturing methods. The choice between them is not a matter of which is superior, but which is appropriate for the specific biological question at hand. A well-designed research strategy will often leverage the strengths of both to build a comprehensive understanding of protein structure and function.
The choice between SDS-PAGE and Native PAGE is a critical strategic decision that directly determines the compatibility and success of downstream analytical techniques. While both methods separate proteins through polyacrylamide gel electrophoresis, their fundamental differences in preserving protein native state create distinct pathways for subsequent analysis. This guide provides an objective comparison of how proteins separated by each method perform in mass spectrometry and functional activity assays, supported by experimental data and detailed protocols.
Fundamental Principles and Downstream Implications
SDS-PAGE and Native PAGE operate on different separation principles that inherently dictate their suitability for downstream applications. Understanding these core mechanisms is essential for selecting the appropriate method for specific research goals.
SDS-PAGE utilizes the anionic detergent sodium dodecyl sulfate (SDS) to denature proteins, linearize them, and impart a uniform negative charge. This results in separation based almost exclusively on molecular weight, as the SDS-protein complexes migrate through the gel matrix with smaller proteins moving faster [6] [3] [57]. The denaturing conditions disrupt higher-order structures, including quaternary arrangements and non-covalent interactions with cofactors [7].
Native PAGE maintains proteins in their native, folded conformation by omitting denaturing agents. Separation depends on the protein's intrinsic charge, size, and three-dimensional shape [6] [9]. This preservation of native structure enables the analysis of functional properties post-separation.
The diagram below illustrates the fundamental separation principles and their direct implications for downstream analysis:
Comparative Performance in Downstream Applications
Mass Spectrometry Compatibility
Mass spectrometry has become an indispensable tool for protein identification, characterization, and proteomic analysis. The compatibility of electrophoretically separated proteins with MS varies significantly between SDS-PAGE and Native PAGE.
SDS-PAGE to Mass Spectrometry provides excellent results for protein identification and covalent modification analysis. The denatured, reduced proteins are ideal for in-gel digestion with proteases like trypsin, which efficiently cleaves the linearized polypeptide chains into predictable peptides [57]. These peptides can be easily extracted from the gel for LC-MS/MS analysis, enabling protein identification through database searching. SDS-PAGE is particularly effective for identifying post-translational modifications that alter molecular weight, such as phosphorylation and glycosylation [57] [73]. The extensive history of combining SDS-PAGE with MS has resulted in well-optimized, robust protocols.
Native PAGE to Mass Spectrometry presents substantial challenges for traditional bottom-up proteomics approaches. The preserved tertiary and quaternary structures limit protease accessibility to cleavage sites, resulting in incomplete digestion and suboptimal peptide generation for LC-MS/MS [9]. However, Native PAGE shows promise for native mass spectrometry applications, where intact protein complexes can be analyzed to study stoichiometry and non-covalent interactions. This approach requires specialized instrumentation and data analysis techniques that are less established than conventional proteomics workflows.
Functional Activity Assays
The preservation of biological function after electrophoretic separation is where these two techniques diverge most dramatically, with Native PAGE offering distinct advantages for functional analysis.
SDS-PAGE completely eliminates protein function due to the denaturing conditions. The SDS detergent disrupts secondary and tertiary structures, while reducing agents like DTT or β-mercaptoethanol break disulfide bonds [6] [57]. This structural destruction irreversibly abolishes enzymatic activity, ligand binding capability, and protein-protein interactions [7]. Consequently, proteins separated by SDS-PAGE cannot be used for functional assays directly from the gel.
Native PAGE preserves protein function, enabling a wide range of activity assays post-separation. Numerous studies have demonstrated that enzymes separated via Native PAGE retain their catalytic activity [7] [3]. This functionality preservation allows researchers to excise protein bands from the gel and use them in activity assays, ligand binding studies, or protein-protein interaction analyses [9]. The technique is particularly valuable for studying metalloenzymes, as it maintains non-covalent interactions with essential metal cofactors that are often lost in SDS-PAGE [7].
Quantitative Experimental Data Comparison
The table below summarizes key experimental findings comparing the performance of SDS-PAGE and Native PAGE in supporting downstream applications:
Table 1: Comparative Performance in Downstream Applications
| Parameter | SDS-PAGE | Native PAGE | Experimental Basis |
|---|---|---|---|
| Enzyme Activity Retention | 0% (All enzymes denatured) | 77.8% (7 of 9 model enzymes active) | Study of nine model enzymes showed all denatured in SDS-PAGE versus majority active in Native PAGE [7] |
| Metal Cofactor Retention | 26% (Zn²⁺ retention in proteomic samples) | 98% (Zn²⁺ retention in proteomic samples) | Analysis of Zn²⁺ retention in LLC-PK1 cell proteome using LA-ICP-MS and TSQ staining [7] |
| Molecular Weight Determination | High accuracy for polypeptide chains | Limited accuracy for native complexes | Migration proportional to log(MW) in SDS-PAGE; influenced by charge/shape in Native PAGE [6] [3] |
| Protein Complex Stability | Disassembles subunits | Maintains quaternary structure | Native PAGE preserves oligomeric state; SDS-PAGE dissociates non-covalent complexes [10] |
| Post-Separation Protein Recovery | Not functional; possible for identification | Functional recovery for assays | Proteins can be electro-eluted or diffused from Native PAGE gels in active form [3] |
| Mass Spectrometry Compatibility | Excellent for identification via peptide mass fingerprinting | Challenging but possible for native MS | Denatured proteins ideal for proteolytic digestion; native structures require special approaches [57] |
Detailed Experimental Protocols
Protocol 1: Standard SDS-PAGE for Mass Spectrometry Analysis
This protocol details the standard procedure for SDS-PAGE separation optimized for subsequent protein identification by mass spectrometry [57] [74].
Sample Preparation:
- Mix protein sample with 4X LDS sample buffer (106 mM Tris HCl, 141 mM Tris Base, 2% LDS, 10% glycerol, 0.51 mM EDTA, pH 8.5) [7]
- Add reducing agent (50 mM DTT or 5% β-mercaptoethanol) to break disulfide bonds
- Heat samples at 70-100°C for 10 minutes to ensure complete denaturation [3] [74]
- Centrifuge at 15,000 rpm for 1 minute to pellet insoluble material
Gel Electrophoresis:
- Use precast or homemade polyacrylamide gels (8-12% acrylamide depending on protein size)
- Employ discontinuous buffer system with stacking gel (pH 6.8) and resolving gel (pH 8.8)
- Load molecular weight markers alongside samples
- Run at constant voltage (100-150V for mini-gels) until dye front reaches bottom [57]
Post-Electrophoresis Processing for MS:
- Stain with Coomassie Blue, SYPRO Ruby, or compatible fluorescent stains
- Excise protein bands of interest with clean scalpel
- Destain with 50% methanol/50 mM ammonium bicarbonate
- Digest in-gel with trypsin (12.5 ng/μL in 25 mM ammonium bicarbonate) overnight at 37°C
- Extract peptides with 50% acetonitrile/5% formic acid
- Analyze by LC-MS/MS
Protocol 2: Native PAGE for Functional Studies
This protocol describes the Native PAGE procedure optimized for preserving protein function, based on the method by [7] with modifications.
Sample Preparation (Non-denaturing):
- Mix protein sample with 4X Native PAGE sample buffer (50 mM BisTris, 50 mM NaCl, 10% glycerol, pH 7.2) [7]
- Do not heat the samples
- Omit reducing agents unless specifically studying reduced state
- Keep samples at 4°C to maintain stability
Gel Electrophoresis:
- Use pre-cast or homemade Native PAGE gels (4-16% gradient recommended)
- Employ non-denaturing, discontinuous buffer system
- Run at constant voltage (150V) at 4°C to prevent denaturation [6]
- Use native molecular weight standards
Activity Staining and Protein Recovery:
- For enzyme activity: Incubate gel in substrate solution specific to the enzyme
- For functional recovery: Electro-elute proteins in non-denaturing buffers
- For metal detection: Use specific stains like TSQ for zinc [7]
Protocol 3: Modified NSDS-PAGE for Balanced Resolution and Function
A hybrid approach called Native SDS-PAGE (NSDS-PAGE) offers a compromise between high resolution and function preservation [7].
Modified Sample Preparation:
- Prepare sample in NSDS buffer (100 mM Tris HCl, 150 mM Tris Base, 10% glycerol, 0.0185% Coomassie G-250, pH 8.5)
- Omit heating step
- Omit EDTA and reducing agents unless required
Modified Running Conditions:
- Reduce SDS concentration in running buffer to 0.0375% (vs. standard 0.1%)
- Run using standard SDS-PAGE equipment at 200V
- Maintain temperature at 4°C during separation
Downstream Applications:
- This method preserves activity for many enzymes while providing better resolution than BN-PAGE
- Enables metal retention (98% Zn²⁺ vs. 26% in standard SDS-PAGE) [7]
- Compatible with both activity assays and mass spectrometry with protocol adjustments
Decision Framework for Method Selection
The flowchart below provides a systematic approach for researchers to select the appropriate electrophoretic method based on their downstream analytical requirements:
Essential Research Reagents and Materials
Successful integration of electrophoresis with downstream analysis requires specific reagents optimized for each method. The table below details essential solutions and their functions:
Table 2: Essential Research Reagent Solutions for Downstream Analysis Integration
| Reagent/Solution | Composition | Function in Downstream Analysis |
|---|---|---|
| SDS-PAGE Sample Buffer | 106 mM Tris HCl, 141 mM Tris Base, 2% LDS, 10% glycerol, 0.51 mM EDTA, pH 8.5 [7] | Denatures proteins, imparts uniform charge for accurate MW determination prior to MS |
| Native PAGE Sample Buffer | 50 mM BisTris, 50 mM NaCl, 10% glycerol, 0.001% Ponceau S, pH 7.2 [7] | Maintains native structure and function for activity assays post-separation |
| SDS-PAGE Running Buffer | 50 mM MOPS, 50 mM Tris Base, 0.1% SDS, 1 mM EDTA, pH 7.7 [7] | Maintains denatured state during separation for optimal MS compatibility |
| Native PAGE Running Buffer | Cathode: 50 mM BisTris, 50 mM Tricine, 0.02% Coomassie G-250, pH 6.8; Anode: 50 mM BisTris, 50 mM Tricine, pH 6.8 [7] | Preserves protein function during electrophoresis while enabling separation |
| NSDS-PAGE Running Buffer | 50 mM MOPS, 50 mM Tris Base, 0.0375% SDS, pH 7.7 [7] | Balanced approach providing better resolution than BN-PAGE while retaining some function |
| Mass Spectrometry-Compatible Stains | Coomassie Blue, SYPRO Ruby, Flamingo Fluorescent | Visualize proteins without interfering with subsequent MS analysis |
| Activity Stain Substrates | Enzyme-specific chromogenic/fluorogenic substrates (e.g., NBT/BCIP for phosphatases) | Detect functional enzymes directly in Native PAGE gels |
| Metal Detection Stains | TSQ (6-methoxy-8-p-toluenesulfonamido-quinoline) for zinc [7] | Detect metalloproteins in gels while maintaining metal-protein interactions |
The integration of protein electrophoresis with downstream analytical techniques requires careful consideration of the fundamental differences between SDS-PAGE and Native PAGE. SDS-PAGE provides superior resolution and excellent compatibility with mass spectrometry for protein identification, but completely eliminates protein function. Native PAGE preserves biological activity and protein complexes, enabling functional studies but with lower resolution and challenges for traditional proteomics. The emerging NSDS-PAGE method offers a promising compromise, retaining significant function while improving resolution. Researchers should select electrophoretic methods based on their primary downstream objectives, using the decision framework and protocols provided to optimize their experimental outcomes.
Conclusion
SDS-PAGE and Native PAGE are not competing but complementary techniques, each offering unique advantages for protein analysis. SDS-PAGE is the undisputed method for determining molecular weight and analyzing denatured proteins, while Native PAGE is essential for investigating native structure, protein-protein interactions, and biological function. The choice between them must be guided by the specific research objective—whether it is analytical purity or functional characterization. Future directions in protein analysis point toward automation, miniaturization, and integration with AI-driven data analysis, yet these foundational electrophoretic methods will remain cornerstones of biomedical research, crucial for advancing drug discovery, diagnostics, and our fundamental understanding of biology.
References
- 1. Electrophoresis [https://www.genome.gov/genetics-glossary/Electrophoresis]
- 2. Electrophoresis [https://en.wikipedia.org/wiki/Electrophoresis]
- 3. Overview of Protein Electrophoresis [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-electrophoresis.html]
- 4. Electrophoresis Ppt | PPTX [https://www.slideshare.net/slideshow/electrophoresis-ppt-238623168/238623168]
- 5. Overview of SDS-PAGE [https://www.creative-proteomics.com/resource/overview-of-sds-page.htm]
- 6. Differences between SDS PAGE and Native PAGE [https://www.geeksforgeeks.org/biology/differences-between-sds-page-and-native-page/]
- 7. Native SDS-PAGE: High Resolution Electrophoretic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4517606/]
- 8. SDS PAGE vs Native PAGE [https://byjus.com/biology/difference-between-sds-page-and-native-page/]
- 9. How Is Native PAGE Different from SDS-PAGE? [https://synapse.patsnap.com/article/how-is-native-page-different-from-sds-page]
- 10. Interpret SDS-PAGE vs Native-PAGE Results [https://www.letstalkacademy.com/sds-page-vs-native-page-protein-dimer/]
- 11. With an anticipated CAGR of 6.2%, the SDS-PAGE ... [https://www.linkedin.com/pulse/anticipated-cagr-62-sds-page-electrophoresis-market-xpopf]
- 12. SDS-PAGE Electrophoresis Market - Global Forecast 2025- ... [https://www.researchandmarkets.com/reports/6140756/sds-page-electrophoresis-market-global?srsltid=AfmBOoq8CgjiRilBTGsEe4yazpXG9MSLrj_9ArAY7jZPhTz7B5SfvZJT]
- 13. Native PAGE Gels | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-gel-electrophoresis/protein-gels/specialized-protein-gels/nativepage-bis-tris-gels.html]
- 14. How SDS-PAGE Works: 7 Key Points Every Scientist ... [https://bitesizebio.com/580/how-sds-page-works/]
- 15. What is the difference between PAGE and SDS-PAGE? [https://www.aatbio.com/resources/faq-frequently-asked-questions/What-is-the-difference-between-PAGE-and-SDS-PAGE]
- 16. Comparison of in-gel protein separation techniques ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4234072/]
- 17. SDS-PAGE: A Cornerstone of Protein Analysis in Modern ... [https://www.metwarebio.com/sds-page-protein-analysis/]
- 18. Deciphering food proteins: The applications of SDS-PAGE ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429225003025]
- 19. Comparison of the performance of 1D SDS-PAGE with ... [https://pubmed.ncbi.nlm.nih.gov/30599360/]
- 20. Should I Use A Native Or Denaturing Gel? [https://info.gbiosciences.com/blog/should-i-use-a-native-or-denaturing-gel]
- 21. High-resolution native electrophoresis in-gel activity assay ... [https://www.nature.com/articles/s41598-025-24684-3]
- 22. SDS-PAGE in Food Science: Elevating Protein Insight and ... [https://www.eurofinsus.com/food-testing/resources/sds-page-in-food-science-elevating-protein-insight-and-product-integrity/]
- 23. Two-dimensional Blue Native/SDS-PAGE Analysis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2649812/]
- 24. SDS-PAGE Protocol [https://neutab.creative-biolabs.com/sds-page-analysis.htm]
- 25. Blue native electrophoresis protocol [https://www.abcam.com/en-us/technical-resources/protocols/blue-native-electrophoresis]
- 26. Validation of blue- and clear-native polyacrylamide gel ... [https://pubmed.ncbi.nlm.nih.gov/40966204/]
- 27. Comparison of the performance of 1D SDS-PAGE with ... [https://www.sciencedirect.com/science/article/abs/pii/S1570023218312546]
- 28. How to Prepare and Run an SDS-PAGE Gel Step by Step [https://synapse.patsnap.com/article/how-to-prepare-and-run-an-sds-page-gel-step-by-step]
- 29. Principle and Protocol of Native-PAGE [https://www.creativebiomart.net/resource/principle-protocol-principle-and-protocol-of-native-page-435.htm]
- 30. Overview of Protein Electrophoresis [https://www.thermofisher.com/tr/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-electrophoresis.html]
- 31. and clear-native polyacrylamide gel electrophoresis protocols ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0332065]
- 32. Deciphering metabolic networks by blue native ... [https://www.sciencedirect.com/science/article/pii/S221296851500015X]
- 33. A screening method for binding synthetic metallo ... [https://www.sciencedirect.com/science/article/pii/S0003269722002445]
- 34. SDS-PAGE Electrophoresis Buffer Insightful Analysis [https://www.archivemarketresearch.com/reports/sds-page-electrophoresis-buffer-639442]
- 35. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis ... [https://www.sciencedirect.com/science/article/pii/S000326972200207X]
- 36. Continuous monitoring of enzymatic activity within native ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3478437/]
- 37. How SDS-PAGE Separates Proteins [https://azurebiosystems.com/blog/how-sds-page-separates-proteins/]
- 38. Separation of Protein Oligomers by Blue Native Gel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2898288/]
- 39. 2-D native-PAGE/SDS-PAGE visualization of an oligomer's ... [https://pubmed.ncbi.nlm.nih.gov/16703630/]
- 40. Continuous monitoring of enzymatic activity within native ... [https://www.sciencedirect.com/science/article/abs/pii/S0003269712004393]
- 41. Mass Estimation of Native Proteins by Blue ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2953912/]
- 42. Advantages and limitations of clear-native PAGE [https://pubmed.ncbi.nlm.nih.gov/16220535/]
- 43. Diverse native-PAGE [http://www.assay-protocol.com/molecular-biology/electrophoresis/diverse-native-PAGE.html]
- 44. Protein Complex Identification and quantitative ... [https://pubmed.ncbi.nlm.nih.gov/31395906/]
- 45. SDS-PAGE Electrophoresis Market Size & Share 2025-2032 [https://www.360iresearch.com/library/intelligence/sds-page-electrophoresis]
- 46. ComplexFinder: A software package for the analysis of ... [https://www.sciencedirect.com/science/article/pii/S0005272821000773]
- 47. Advantages and Disadvantages of SDS-PAGE for Protein ... [https://www.mtoz-biolabs.com/advantages-and-disadvantages-of-sds-page-for-protein-separation.html]
- 48. Troubleshooting SDS-PAGE Gel Running Issues [https://www.goldbio.com/blogs/articles/troubleshooting-sds-page-gel-running-issues?srsltid=AfmBOoo9_roKYltxbd0F4eVUyBrKynqIEJYpof2YJD46gMA5N5a7QNJb]
- 49. Troubleshooting Sodium Dodecyl Sulfate- Polyacrylamide ... [https://www.hycultbiotech.com/sds-page/troubleshooting/]
- 50. Troubleshooting Common Electrophoresis Problems and ... [https://www.labx.com/resources/troubleshooting-common-electrophoresis-problems-and-artifacts/5539]
- 51. SDS-PAGE Troubleshooting: Why Are My Protein Bands ... [https://synapse.patsnap.com/article/sds-page-troubleshooting-why-are-my-protein-bands-fuzzy]
- 52. Protein Gel 1D Electrophoresis Support—Troubleshooting [https://www.thermofisher.com/us/en/home/technical-resources/technical-reference-library/protein-electrophoresis-western-blotting-support-center/protein-gel-1d-electrophoresis-support1/protein-gel-1d-electrophoresis-support-troubleshooting.html]
- 53. What Are the Different Types of Western Blot Gels? [https://www.licorbio.com/blog/types-of-western-blot-gels]
- 54. A Guide to Gradient Gels: The Why's and How's [https://bitesizebio.com/47184/gradient-gels/]
- 55. A Guide to Choosing the Right Gel for Protein Gel ... [https://biobasic-asia.com/choosing-guide-protein-gel/]
- 56. How to choose an acrylamide gel concentration for western ... [https://hellobio.com/how-to-choose-an-acrylamide-gel-concentration-for-western-blot/]
- 57. SDS-PAGE guide: Sample preparation to analysis [https://www.abcam.com/en-us/knowledge-center/western-blot/sds-page-guide]
- 58. Gradient Gels vs. Fixed % Gels: Which Should You Use? [https://nusep.us/blogs/precast-gel-education-center/4-20-gradient-gels-vs-fixed-gels-when-to-use-each?srsltid=AfmBOor4OjADSAPrVspDasC-JT4gE28BIdgles2TUQGbDZ4Ja6OrNv6_]
- 59. SDS-PAGE [https://en.wikipedia.org/wiki/SDS-PAGE]
- 60. Polyacrylamide Gel Electrophoresis, How It... | Technology Networks [https://www.technologynetworks.com/analysis/articles/polyacrylamide-gel-electrophoresis-how-it-works-technique-variants-and-its-applications-359100]
- 61. Exploring Protein Aggregation in Biological Products [https://link.springer.com/article/10.1208/s12249-025-03189-2]
- 62. In-gel refolding allows fluorescence detection of fully ... [https://www.sciencedirect.com/science/article/pii/S0003269725000995]
- 63. SDS protein interactions [https://www.sciencedirect.com/science/article/abs/pii/S0301462225001462]
- 64. SDS-PAGE Protocol [https://www.rockland.com/resources/sds-page-protocol/?srsltid=AfmBOoqkbnQCACooM-HzuSJ6U8SV1_q5wsslEsFC8Y7FbLqroE7BtIam]
- 65. Protein analysis SDS PAGE [https://www.qiagen.com/us/knowledge-and-support/knowledge-hub/bench-guide/protein/protein-analysis/protein-analysis-sds-page]
- 66. SDS-PAGE Experiment Protocol [https://microbiosci.creative-biogene.com/sds-page-experiment-protocol.html]
- 67. Comparing SDS-PAGE and CE-SDS for Antibody Purity ... [https://www.bioprocessintl.com/qa-qc/comparing-sds-page-and-ce-sds-for-antibody-purity-analysis]
- 68. In-depth structural proteomics integrating mass ... [https://www.frontiersin.org/journals/analytical-science/articles/10.3389/frans.2022.1107183/full]
- 69. Direct observation of fluorescent proteins in gels [https://pmc.ncbi.nlm.nih.gov/articles/PMC11808229/]
- 70. Comparison of glycoprotein separation reveals greater ... [https://www.sciencedirect.com/science/article/abs/pii/S0731708519319922]
- 71. Validation of blue- and clear-native polyacrylamide gel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12445495/]
- 72. OTT1313 [https://uwmrf.org/technology/ott1313/]
- 73. Application of SDS-PAGE in Protein Characterization [https://www.mtoz-biolabs.com/application-of-sds-page-in-protein-characterization.html]
- 74. The principle and method of polyacrylamide gel ... [https://ruo.mbl.co.jp/bio/e/support/method/sds-page.html]