SDS-PAGE vs Native PAGE: A Comprehensive Guide to Protein Separation Techniques
This article provides a detailed comparison of SDS-PAGE and Native PAGE, two fundamental protein separation techniques in biochemistry and molecular biology.
SDS-PAGE vs Native PAGE: A Comprehensive Guide to Protein Separation Techniques
Abstract
This article provides a detailed comparison of SDS-PAGE and Native PAGE, two fundamental protein separation techniques in biochemistry and molecular biology. Tailored for researchers, scientists, and drug development professionals, it covers the core principles, methodological applications, troubleshooting strategies, and comparative analysis of these techniques. Readers will gain practical insights into selecting the appropriate method based on their research goals, whether for determining molecular weight, studying native protein complexes, or preparing samples for downstream analysis like western blotting or mass spectrometry.
Core Principles: How SDS-PAGE and Native PAGE Work
Fundamental Principle of Polyacrylamide Gel Electrophoresis (PAGE)
Polyacrylamide Gel Electrophoresis (PAGE) is a foundational technique in biochemistry and molecular biology for separating biological macromolecules, primarily proteins, based on their physicochemical properties. Within this field, two primary methodologies—SDS-PAGE and Native PAGE—serve distinct and complementary purposes. The fundamental principle underpinning all PAGE techniques is the movement of charged molecules through an inert polyacrylamide gel matrix under the influence of an electric field, which results in their separation [1]. The specific nature of this separation, however, is dictated by whether the protein's native structure is preserved, framing a critical dichotomy in protein research.
Core Principles of PAGE and the SDS-PAGE/Native PAGE Dichotomy
The core mechanism of PAGE relies on the fact that charged protein molecules will migrate through a porous gel towards an electrode of opposite charge when an electric field is applied [1]. The polyacrylamide gel, formed through the polymerization of acrylamide and bisacrylamide, acts as a molecular sieve [1]. The pore size of this sieve can be precisely controlled by varying the concentrations of these components, allowing for the separation of molecules across a wide size range [2] [3].
This general principle branches into two main approaches:
- SDS-PAGE (Sodium Dodecyl Sulfate-PAGE) is a denaturing technique. It employs the anionic detergent sodium dodecyl sulfate (SDS) and often a reducing agent (like DTT or β-mercaptoethanol) to unfold and linearize proteins. SDS binds to the protein backbone in a constant ratio, masking the protein's intrinsic charge and imparting a uniform negative charge proportional to its mass [2] [3] [4]. This process eliminates the influence of a protein's native charge and shape, ensuring separation occurs almost exclusively based on polypeptide chain length or molecular weight [2] [5].
- Native PAGE is a non-denaturing technique. It is performed without SDS or reducing agents, and the sample is not heated prior to loading [5] [6]. This preserves the protein's higher-order structure—its secondary, tertiary, and quaternary conformations—and its inherent biological activity [5] [1]. Consequently, separation in Native PAGE is based on a combination of the protein's intrinsic charge, size, and shape [5] [6].
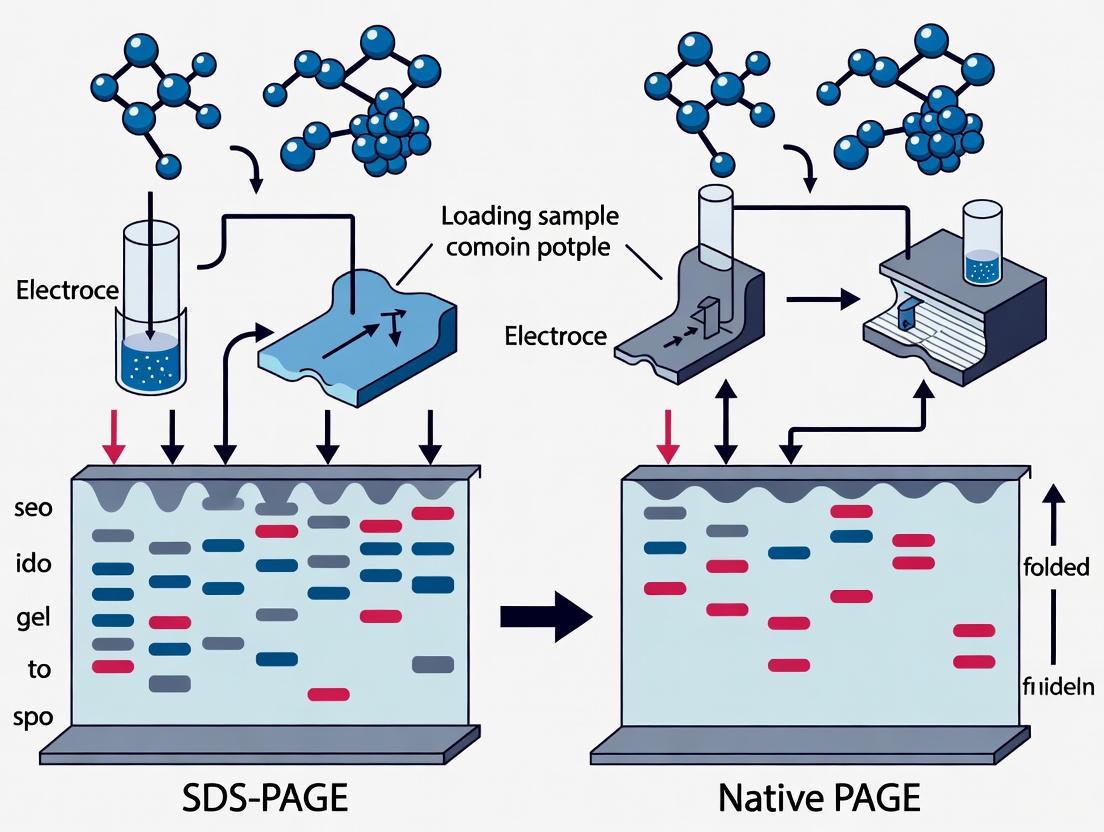
The following workflow diagram illustrates the key procedural divergences between these two methods:
Comparative Analysis: SDS-PAGE vs. Native PAGE
The choice between SDS-PAGE and Native PAGE has profound implications for the outcome of an experiment and the type of information that can be obtained. The table below provides a direct comparison of these two techniques across critical parameters.
Table 1: A direct comparison of SDS-PAGE and Native PAGE methodologies.
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| Core Principle | Separation based solely on molecular weight [2] [5]. | Separation based on size, intrinsic charge, and shape [5] [6]. |
| Protein State | Denatured and linearized [5] [3]. | Native, folded conformation preserved [5] [6]. |
| Key Reagents | SDS, reducing agents (DTT/β-ME) [6] [4]. | No denaturing or reducing agents [6]. |
| Sample Preparation | Heating step required (e.g., 95°C for 5 mins) [2] [4]. | No heating step [6]. |
| Protein Function | Biological activity is destroyed [5] [7]. | Biological activity is retained [5] [7]. |
| Charge of Proteins | Uniform negative charge from SDS [3] [4]. | Native charge (positive, negative, or neutral) is maintained [6]. |
| Primary Applications | Molecular weight determination, purity assessment, western blotting [3]. | Study of protein complexes, oligomerization, enzymatic activity [5] [7]. |
| Protein Recovery | Typically not recoverable in functional form [6]. | Can be recovered for functional assays [5]. |
Hybrid Approaches: Native SDS-PAGE (NSDS-PAGE)
To address the limitation of SDS-PAGE in destroying native protein function while striving to maintain its high resolution, hybrid methods have been developed. Native SDS-PAGE (NSDS-PAGE) is one such innovation. This technique modifies standard SDS-PAGE conditions by removing SDS and EDTA from the sample buffer, omitting the heating step, and significantly reducing the SDS concentration in the running buffer (e.g., to 0.0375%) [7]. Research has demonstrated that this approach can achieve high-resolution separation similar to traditional SDS-PAGE while allowing a majority of model enzymes to retain their activity and preserving bound metal ions in metalloproteins [7]. This makes NSDS-PAGE a powerful tool for applications in fields like metallomics.
Detailed Experimental Protocols
1. Gel Preparation:
- Assemble Casting Module: Thoroughly clean glass plates and spacers, then assemble the gel cassette using a casting frame or binder clips [2].
- Prepare and Pour Resolving Gel: Mix components for the separating gel (e.g., acrylamide/bis-acrylamide, Tris-HCl buffer pH ~8.8, SDS, APS, TEMED). Pipette the solution into the cassette, leaving space for the stacking gel. Overlay with water-saturated butanol or isopropanol to ensure a flat, oxygen-free surface and allow to polymerize for 20-30 minutes [2] [4].
- Prepare and Pour Stacking Gel: After polymerization, discard the overlay and rinse. Mix the stacking gel solution (lower acrylamide concentration, Tris-HCl buffer pH ~6.8, SDS, APS, TEMED). Pour onto the resolving gel and immediately insert a clean comb without introducing air bubbles. Allow to polymerize [2] [4].
2. Sample Preparation:
- Mix the protein sample with an SDS-PAGE sample loading buffer (containing SDS, a reducing agent, glycerol, and a tracking dye like bromophenol blue) [2] [3].
- Heat the mixture at 95-100°C for 3-5 minutes to denature the proteins [4].
- Centrifuge at high speed (e.g., 15,000 rpm) for 1-2 minutes to pellet insoluble debris [2].
3. Electrophoresis:
- Mount the polymerized gel in the electrophoresis apparatus and fill the upper and lower chambers with running buffer (e.g., Tris-Glycine-SDS buffer) [2] [4].
- Carefully load the prepared samples and a molecular weight marker into the wells.
- Connect the power supply and run the gel at a constant voltage (e.g., 100-200 V) until the dye front reaches the bottom of the gel [2] [3].
4. Post-Electrophoresis Analysis:
- Disassemble the apparatus and carefully pry the glass plates apart to remove the gel.
- The gel is typically stained (e.g., with Coomassie Blue or silver stain) to visualize the protein bands or used for downstream applications like western blotting [3] [4].
1. Gel Preparation:
- The process is similar to SDS-PAGE gel casting. However, the resolving and stacking gel solutions are prepared without SDS or other denaturing agents [6]. The buffer systems may also differ.
2. Sample Preparation:
- The protein sample is mixed with a non-denaturing sample buffer (containing glycerol and a tracking dye, but no SDS, reducing agents, or heat treatment) [7] [6].
3. Electrophoresis:
- The gel is run in a running buffer that lacks SDS [7] [6]. To minimize heat-induced denaturation, the electrophoresis is often performed at 4°C [6]. The applied voltage may also be lower than in SDS-PAGE.
4. Post-Electrophoresis Analysis:
- Proteins can be visualized by staining. Crucially, because native function is preserved, proteins can be extracted from the gel for activity assays or other functional studies [5].
The Scientist's Toolkit: Essential Reagents and Materials
Successful execution of PAGE requires a set of specific reagents and materials. The following table catalogs the key components and their functions in the process.
Table 2: Essential research reagents and materials for Polyacrylamide Gel Electrophoresis.
| Item | Function / Purpose |
|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked polyacrylamide gel matrix that acts as a molecular sieve [1]. |
| Ammonium Persulfate (APS) & TEMED | Catalyze the free-radical polymerization of acrylamide to form the gel [1] [4]. |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers a uniform negative charge [2] [3]. |
| Reducing Agents (DTT, β-ME) | Cleave disulfide bonds to fully unfold proteins [3] [1]. |
| Tris-based Buffers | Provide the conductive medium and maintain stable pH during electrophoresis [1]. |
| Coomassie Brilliant Blue | A dye used to stain and visualize proteins in the gel after electrophoresis [3]. |
| Molecular Weight Markers | A mixture of proteins of known sizes, allowing estimation of the molecular weight of unknown proteins [4]. |
| Glycine | In discontinuous SDS-PAGE, acts as a trailing ion in the stacking gel to focus proteins into sharp bands [4]. |
The separation mechanism in either technique can be visualized as a molecular race through a porous mesh, governed by different rules, as shown below:
Data Analysis and Interpretation
Quantitative and Qualitative Analysis
- Molecular Weight Determination: In SDS-PAGE, the distance migrated by a protein band is inversely proportional to the logarithm of its molecular mass (MW). A calibration curve is generated by plotting the log(MW) of standard marker proteins against their migration distance (Rf). The MW of an unknown protein can then be estimated from this curve [8].
- Activity Staining: After Native PAGE, gels can be incubated with specific substrates to detect enzymatic activity, revealing the position of active enzymes [5] [7].
- Software-Based Analysis: Tools like MatGel, a MATLAB-based program, can automate the detection and quantification of protein spots from 2D-PAGE gel images, enabling high-throughput quantitative proteomic studies [9].
Case Study: Inferring Protein Quaternary Structure
The combined use of SDS-PAGE and Native PAGE is powerful for studying protein complexes. Consider a protein that migrates as a 60 kDa band on non-reducing SDS-PAGE but as a 120 kDa band on Native PAGE [10].
- Interpretation: The 60 kDa band on SDS-PAGE indicates the polypeptide chain length. The 120 kDa band on Native PAGE indicates the mass of the native, folded complex. The difference reveals that the native protein is a dimer of two 60 kDa subunits. Since this occurs under non-reducing conditions (no agents to break disulfide bonds), the inference is that the two subunits are held together by non-covalent interactions (e.g., hydrophobic or electrostatic forces) and not by disulfide bridges [10].
SDS-PAGE and Native PAGE are not merely technical alternatives but are foundational tools that enable researchers to answer fundamentally different biological questions. SDS-PAGE simplifies protein identity to molecular weight, providing a high-resolution, denaturing separation ideal for analytical and preparative workflows like western blotting. In contrast, Native PAGE embraces the complexity of protein native state, allowing for the study of function, interaction, and structure in a context that mimics the physiological environment. The choice between them is dictated by the research objective: determining weight and purity demands SDS-PAGE, while probing activity and complexes necessitates Native PAGE. Understanding this core principle and the explicit differences between these techniques is indispensable for designing robust experiments and generating meaningful data in protein science and drug development.
Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) is a foundational technique in biochemistry and molecular biology that separates proteins based solely on their molecular weight under denaturing conditions. Its development, primarily by Laemmli in 1970, provided a simple, reliable method for analyzing protein mixtures, determining molecular mass, and assessing purity, making it indispensable for modern research and drug development [11] [12]. This guide details the core principles, protocols, and applications of SDS-PAGE, framing it within the broader context of protein separation research by contrasting it with its complementary technique, Native PAGE.
Core Principles and Mechanism of Action
The primary objective of SDS-PAGE is to negate the influence of a protein's inherent charge and three-dimensional structure, ensuring separation is dependent only on polypeptide chain length.
- Protein Denaturation and Linearization: The detergent Sodium Dodecyl Sulfate (SDS) plays a dual role. First, it binds extensively to hydrophobic regions of proteins, disrupting hydrogen bonds and van der Waals forces. This interaction unfolds and linearizes the proteins, destroying their secondary and tertiary structures [13].
- Charge Masking and Uniformity: SDS is an anionic detergent. When bound to proteins, it coats them in a uniform layer of negative charge. This masks the proteins' intrinsic charges, which arise from their variable amino acid compositions. Consequently, all SDS-bound proteins carry a similar negative charge density per unit mass [13] [12].
- Separation by Molecular Sieving: The polyacrylamide gel acts as a molecular sieve. When an electric field is applied, the negatively charged protein-SDS complexes migrate toward the positive anode. Smaller proteins navigate the porous gel matrix more easily and migrate farther, while larger proteins are more hindered and travel a shorter distance. This results in a separation based strictly on molecular weight [13] [12].
Step-by-Step Experimental Protocol
A robust SDS-PAGE protocol ensures clear, reproducible results. The following methodology is standard for most vertical mini-gel systems.
Sample Preparation
Proper sample preparation is critical for successful denaturation.
Combine Sample and Buffer: Mix the protein sample with an equal volume of 2X Laemmli Sample Buffer. A typical 2X buffer contains [6] [13]:
- SDS: Denatures proteins and provides uniform negative charge.
- Glycerol: Adds density for easy gel loading.
- Tris-HCl (pH 6.8): Buffers the sample.
- Bromophenol Blue: A tracking dye to monitor electrophoresis progress.
- Reducing Agent (DTT or β-mercaptoethanol): Added to "reducing" SDS-PAGE to break disulfide bonds, ensuring complete unfolding of polypeptide subunits [11] [14].
Heat Denaturation: Heat the mixture at 95°C for 5 minutes [14]. This step is crucial for彻底破坏hydrogen bonds and ensuring complete linearization of the proteins. After heating, briefly centrifuge the samples to collect condensation.
Non-Reducing Conditions: For studying protein complexes or disulfide bonds, omit the reducing agent (DTT/β-ME). This is "non-reducing SDS-PAGE," where separation is based on the size of the disulfide-linked complex rather than individual subunits [11].
Gel Preparation and Casting
Polyacrylamide gels are formed through a chemical polymerization process.
- Gel Composition: The gel is a polymer network created by reacting acrylamide with the cross-linking agent bis-acrylamide. The polymerization is initiated by ammonium persulfate (APS) and catalyzed by TEMED, which generates free radicals to drive the reaction [13] [12].
- Resolving Gel: This lower portion of the gel has a higher acrylamide concentration (e.g., 7-15%) and a higher pH (∼8.8). It is responsible for the size-based separation of proteins. The percentage should be chosen based on the target protein's size: lower percentages for large proteins, higher percentages for small proteins [13] [12] [14].
- Stacking Gel: This upper portion has a lower acrylamide concentration (∼4%) and a different pH (∼6.8). Its function is to concentrate all protein samples into a sharp, unified band before they enter the resolving gel, greatly improving resolution [13] [12].
Electrophoresis Run
The prepared gel cassette is placed in a running chamber filled with an appropriate buffer (e.g., Tris-Glycine-SDS).
- Load Samples and Markers: Load prepared samples into the wells. Always include a protein ladder (molecular weight marker) in at least one well. This standard contains proteins of known sizes, enabling estimation of the molecular weights of unknown sample proteins [12].
- Apply Electric Field: Connect the apparatus to a power supply. The choice of running mode is a key consideration [15]:
- Constant Voltage: Safer as heat production decreases over time, but run times can be longer, potentially leading to diffuse bands. Suitable for running multiple chambers from one power pack.
- Constant Current: Provides a constant migration rate and sharper bands, but requires monitoring as heat generation can increase, risking overheating. A typical setting is 100-150 V for 40-60 minutes, or until the dye front reaches the bottom of the gel [15] [14].
- Temperature Control: Maintain a constant temperature between 10°C-20°C to prevent "smiling" effects (bands curving upwards) caused by uneven heat distribution across the gel [14].
Protein Visualization
After separation, proteins are invisible and must be stained for detection.
- Coomassie Staining: The most common method. It is quantitative and compatible with downstream applications like mass spectrometry.
- Stain: Incubate the gel in Coomassie Brilliant Blue R-250 solution (0.05% dye, 40% ethanol, 10% acetic acid) for 30 minutes to 2 hours [16].
- Destain: Agitate the gel in a destaining solution (40% ethanol, 10% acetic acid) until the background is clear and protein bands are sharply visible. Typically, bands with ≥50 ng of protein can be detected [16].
- Silver Staining: A more sensitive alternative, capable of detecting 2-5 ng of protein per band. However, it is less quantitative and often incompatible with further protein analysis [16].
The workflow below summarizes the key steps of the SDS-PAGE protocol.
Key Technical Differences: SDS-PAGE vs. Native PAGE
SDS-PAGE and Native PAGE serve distinct purposes in protein analysis. Their fundamental differences are critical for selecting the appropriate technique for a given research question. The table below provides a structured comparison.
Table 1: Comparative Analysis of SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight only [6] [17] | Size, overall charge, and shape [6] [5] |
| Protein State | Denatured and linearized [6] [12] | Native, folded conformation [6] [5] |
| SDS Presence | Yes, required for denaturation and charge masking [6] | No [6] |
| Reducing Agents | Often used (DTT/BME) to break disulfide bonds [6] [11] | Not used [6] |
| Protein Function Post-Run | Lost due to denaturation [6] | Retained (proteins remain active) [6] [12] |
| Protein Recovery | Typically not recoverable in functional form [6] | Can be recovered for functional studies [6] [5] |
| Primary Applications | Molecular weight determination, purity checks, protein expression analysis [6] [18] | Studying protein complexes, oligomerization, and enzymatic activity [6] [5] |
The following diagram illustrates the conceptual differences in how proteins are treated and separated in each method.
Essential Reagents and Materials
Successful SDS-PAGE requires specific reagents, each with a defined role in the process.
Table 2: The SDS-PAGE Researcher's Toolkit: Key Reagents and Their Functions
| Reagent/Material | Function |
|---|---|
| Sodium Dodecyl Sulfate (SDS) | Anionic detergent that denatures proteins and confers a uniform negative charge [13]. |
| Acrylamide/Bis-acrylamide | Monomer and cross-linker that polymerize to form the porous gel matrix for molecular sieving [13] [12]. |
| Ammonium Persulfate (APS) & TEMED | Initiator and catalyst for the polymerization of the polyacrylamide gel [13] [12]. |
| DTT or β-Mercaptoethanol | Reducing agents that break disulfide bonds to fully unfold proteins into individual subunits [13] [14]. |
| Tris-based Buffers | Provide the appropriate ionic strength and pH environment for gel polymerization and electrophoresis [13] [12]. |
| Coomassie Brilliant Blue | Dye used for staining and visualizing separated protein bands on the gel [16]. |
| Protein Molecular Weight Marker | Standard containing proteins of known sizes for calibrating and estimating unknown protein weights [12]. |
Applications in Research and Drug Development
SDS-PAGE is a versatile workhorse in life science laboratories with wide-ranging applications.
- Protein Purity and Identity Analysis: It is routinely used to assess the purity of protein preparations during purification and to confirm the identity of a protein, such as verifying the expression of a recombinant therapeutic protein in biotech and pharmaceutical settings [18] [12].
- Molecular Weight Determination: By comparing the migration distance of an unknown protein to a standard curve generated by the protein ladder, researchers can estimate its molecular mass [12].
- Western Blotting: SDS-PAGE is the essential first separation step before proteins are transferred to a membrane for detection with specific antibodies via western blotting [11] [14].
- Food Science and Allergen Detection: The technique is pivotal in food science for protein profiling, detecting adulteration, and identifying allergens across various food categories like cereals, dairy, and meat products [11].
- Clinical Diagnostics: Clinical laboratories use SDS-PAGE to analyze serum or other bodily fluid proteins for diagnosing certain diseases, such as identifying immunoglobulin abnormalities [18].
Troubleshooting for Optimal Results
Several factors are critical for achieving sharp, high-resolution bands.
- Gel Percentage Selection: The acrylamide concentration must be appropriate for the target protein size. Gradient gels (e.g., 4-20%) are excellent for separating a wide range of molecular weights simultaneously. Proteins ≥ 200 kDa resolve best in 4-8% gels, while smaller proteins require higher percentages (e.g., 12-15%) [14].
- Optimal Protein Loading: Overloading wells causes smearing, while underloading results in faint bands. A general guideline is to load ≤2 µg of a purified protein or ≤20 µg of a complex mixture like cell lysate for Coomassie staining [14].
- Managing Joule Heating: Excessive heat causes band distortion ("smiling"). Using constant voltage, running the gel in a cold room, or employing a magnetic stirrer in the buffer tank can help dissipate heat and ensure even migration [15] [14].
- Complete Sample Denaturation: Incomplete heating or insufficient reducing agent can lead to aberrant banding patterns due to residual protein structure. Ensuring samples are heated at 95°C for 5 minutes with adequate reducing agent is crucial [14].
Within the landscape of protein analysis techniques, Native Polyacrylamide Gel Electrophoresis (Native PAGE) serves as a critical methodology for studying proteins in their biologically active state. This technique exists in a complementary relationship with its denaturing counterpart, SDS-PAGE, forming a comprehensive framework for protein separation research. While SDS-PAGE revolutionized molecular weight determination by unraveling proteins into uniform linear chains, it does so at the cost of destroying native structure and function [5]. Native PAGE addresses this fundamental limitation by preserving the intricate three-dimensional architecture of proteins throughout the separation process.
The core distinction lies in what property governs separation: in SDS-PAGE, migration depends almost exclusively on molecular mass, whereas in Native PAGE, separation depends on the protein's intrinsic charge, molecular size, and three-dimensional shape [6] [12]. This preservation of native conformation allows researchers to investigate functional properties that are inaccessible through denaturing methods, including enzymatic activity, protein-protein interactions, and the composition of multi-subunit complexes [5]. For drug development professionals, this capability is invaluable, as it enables the study of therapeutic protein complexes and their interactions under conditions that mimic the physiological environment.
Fundamental Principles of Native PAGE
The Mechanism of Non-Denaturing Separation
The operational principle of Native PAGE hinges on maintaining proteins in their native, folded conformation during electrophoretic separation. Without denaturing agents like SDS, a protein's migration through the polyacrylamide gel matrix is governed by a combination of factors: its inherent electrostatic charge, hydrodynamic size, and molecular shape [12] [17]. When an electric field is applied, the gel acts as a molecular sieve, creating a frictional force that regulates protein movement. Smaller, more compact proteins navigate the porous matrix more easily than larger complexes, while the protein's net charge at the running buffer pH determines its electrophoretic mobility and direction [12].
This multi-parameter separation mechanism stands in stark contrast to SDS-PAGE, where the binding of sodium dodecyl sulfate confers a uniform negative charge and unravels the protein structure, effectively making separation dependent solely on polypeptide chain length [6] [4]. In Native PAGE, proteins with high negative charge density migrate faster toward the anode, while larger proteins experience greater frictional resistance [12]. The resulting separation reflects the protein's native charge-to-mass ratio, preserving not just the primary structure but also the secondary, tertiary, and quaternary structures that define its biological function [19] [17].
Key Variants: BN-PAGE and CN-PAGE
Native PAGE encompasses several specialized techniques tailored to different research needs, with Blue Native PAGE (BN-PAGE) and Clear Native PAGE (CN-PAGE) being the most prominent.
Blue Native PAGE (BN-PAGE): This method incorporates Coomassie Brilliant Blue dye into the cathode buffer, which confers negative charges to protein complexes without causing significant denaturation [6] [7]. The dye binding allows for the separation of even basic proteins and provides excellent resolution for hydrophobic membrane protein complexes [7]. The blue dye also enables visual tracking of electrophoresis progress and does not interfere with subsequent western blotting or mass spectrometry analysis.
Clear Native PAGE (CN-PAGE): This technique separates proteins based solely on their intrinsic charge in a gradient gel without using Coomassie dye [6]. CN-PAGE offers milder conditions that are suitable for fragile protein complexes that might be disrupted by dye binding, though it may provide lower resolution for some protein mixtures [7]. The absence of dye allows for direct spectroscopic analysis and is preferable when studying metal-containing proteins or conducting certain enzymatic assays immediately after separation.
Comparative Analysis: Native PAGE versus SDS-PAGE
Fundamental Differences in Separation Mechanisms
The choice between Native PAGE and SDS-PAGE represents a fundamental decision in experimental design, with each technique providing distinct information about protein characteristics. The table below summarizes the core differences between these two electrophoretic methods.
Table 1: Key Differences Between Native PAGE and SDS-PAGE
| Criteria | Native PAGE | SDS-PAGE |
|---|---|---|
| Separation Basis | Size, charge, and shape [6] [12] | Molecular weight only [6] [17] |
| Gel Conditions | Non-denaturing [6] [19] | Denaturing [6] [19] |
| SDS Presence | Absent [6] [19] | Present [6] [4] |
| Sample Preparation | Not heated [6] | Heated (70-100°C) [6] [4] |
| Reducing Agents | Absent [6] | Often present (e.g., DTT, β-mercaptoethanol) [6] [11] |
| Protein State | Native, folded conformation [6] [5] | Denatured, linearized [6] [5] |
| Protein Function | Retained [6] [5] | Lost [6] [5] |
| Protein Recovery | Possible with retained function [6] [19] | Not functional if recovered [6] |
| Primary Applications | Study structure, function, interactions [6] [5] | Determine molecular weight, check purity [6] [5] |
Practical Implications for Protein Characterization
The methodological differences between Native PAGE and SDS-PAGE lead to distinct practical outcomes in protein analysis. A compelling example involves characterizing a multi-subunit protein: on SDS-PAGE, it might migrate as 60 kDa subunits, while on Native PAGE, the intact complex migrates as a 120 kDa species, revealing its dimeric quaternary structure stabilized by non-covalent interactions [10]. This interpretive power makes Native PAGE indispensable for studying oligomerization states and protein-protein interactions [5].
Furthermore, the preservation of biological activity following Native PAGE enables direct functional analysis. Proteins separated via Native PAGE can be excised from the gel and subjected to activity assays, providing a direct link between protein bands and enzymatic function [5] [12]. This capability is particularly valuable in drug development for identifying active therapeutic protein complexes and studying their behavior under native conditions. In contrast, SDS-PAGE is ideal for determining subunit molecular weights, assessing sample purity, and preparing proteins for western blotting, but provides no information about native structure or function [5] [12].
Experimental Methodology for Native PAGE
Standard Protocol and Workflow
The following workflow outlines the key steps in performing Native PAGE, highlighting critical stages where protocol deviations from SDS-PAGE are essential for preserving protein function.
Diagram 1: Native PAGE experimental workflow.
Gel Preparation
Native PAGE typically uses polyacrylamide gels ranging from 4-16% acrylamide concentration, with gradient gels often employed to resolve complex protein mixtures [7]. The gel and running buffers lack SDS and other denaturing agents. Common buffer systems include Tris-glycine or Tris-borate at neutral to slightly alkaline pH (typically pH 7.2-8.8) to maintain protein stability [7] [12]. The gel polymerization process is similar to SDS-PAGE, using ammonium persulfate (APS) and TEMED as catalysts [12].
Sample Preparation
Critical to Native PAGE success is non-denaturing sample preparation. Protein samples are mixed with a mild, non-denaturing sample buffer containing sucrose or glycerol to increase density, and a tracking dye like bromophenol blue [6] [7]. Notably, samples are not heated before loading, as heat would denature proteins and defeat the purpose of native electrophoresis [6]. The buffer should maintain a pH that preserves protein function, typically near physiological pH (7.0-7.5) [12].
Electrophoresis Conditions
Native PAGE is typically performed at 4°C to minimize protein denaturation and proteolytic activity during separation [6]. The applied voltage is generally lower than in SDS-PAGE (e.g., 100-150V for mini-gels), with longer run times to complete separation [7]. The electrophoresis apparatus should be placed in a cold room or using a cooling unit to maintain temperature control throughout the run.
Post-Electrophoresis Analysis
Following separation, proteins can be visualized using standard staining methods like Coomassie Brilliant Blue or silver stain [6]. For functional studies, proteins can be recovered from the gel through passive diffusion, electro-elution, or by blotting to membranes under native conditions [12]. Enzymatic activity can be detected directly using specific activity stains or zymography [5].
Research Reagent Solutions
Successful Native PAGE requires specific reagents tailored to preserve protein native state. The following table outlines essential materials and their functions.
Table 2: Essential Reagents for Native PAGE
| Reagent/Category | Function in Native PAGE | Examples & Notes |
|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the porous gel matrix for size-based separation [12] | Concentrations typically 4-16%; ratio determines pore size [12] |
| Non-denaturing Buffers | Provides ionic environment without disrupting structure [7] | Tris-glycine, Tris-borate; pH 7.2-8.8 [7] [12] |
| Tracking Dye | Visualizes electrophoresis progress [7] | Bromophenol blue; no SDS present [7] |
| Coomassie Dye (BN-PAGE) | Imparts charge for membrane protein separation [6] [7] | Coomassie Brilliant Blue G-250; added to cathode buffer [7] |
| Glycerol/Sucrose | Increases sample density for gel loading [7] | Included in sample buffer; typically 10-20% concentration [7] |
| Molecular Weight Markers | Size estimation reference under native conditions [7] | NativeMark unstained standards; different migration vs. SDS-PAGE markers [7] |
Advanced Applications and Recent Developments
Research and Drug Development Applications
Native PAGE serves as a critical tool in both basic research and pharmaceutical development, enabling investigations that would be impossible with denaturing techniques.
Protein-Protein Interaction Studies: Native PAGE effectively characterizes stable protein complexes and oligomeric states, as the technique preserves non-covalent interactions between subunits [5] [10]. This application is invaluable for mapping interactomes and understanding cellular signaling complexes.
Enzymatic Characterization: The ability to recover active enzymes after separation allows researchers to link specific protein bands to catalytic function [5]. This is particularly useful in identifying isozymes and studying enzymatic regulation in complex biological samples.
Metalloprotein Analysis: Native PAGE preserves non-covalently bound metal ions essential for the function of many proteins [7]. Research demonstrates that modified Native PAGE conditions (NSDS-PAGE) can retain up to 98% of Zn²⁺ bound in proteomic samples, enabling analysis of metalloenzymes that would be disrupted by standard SDS-PAGE [7].
Therapeutic Protein Characterization: In drug development, Native PAGE analyzes higher-order structure of biologic therapeutics, confirming proper assembly of multi-subunit proteins and detecting aggregation that might impact efficacy or safety [20].
Integration with Modern Analytical Techniques
The field of native separation continues to evolve, with significant advances in coupling Native PAGE with high-resolution detection methods:
Native MS (Mass Spectrometry): Native PAGE-separated proteins can be subsequently analyzed by native mass spectrometry, providing information about intact mass, subunit composition, stoichiometry, and post-translational modifications of protein complexes [20]. This powerful combination offers unprecedented insights into protein higher-order structure.
Two-Dimensional Electrophoresis: Native PAGE is employed as the first dimension in 2D electrophoresis, followed by denaturing SDS-PAGE in the second dimension [12]. This approach separates protein complexes in the first dimension and their individual subunits in the second, providing a comprehensive view of complex composition.
Advanced Native Separation Methods: Recent developments include liquid chromatography and capillary electrophoresis under non-denaturing conditions, which complement Native PAGE for specific applications [20]. These methods address challenges in resolution and detection of low-abundance proteins in complex mixtures.
Native PAGE remains an indispensable technique in the protein separation research landscape, offering unique capabilities for studying proteins in their functional, native state. Its complementary relationship with SDS-PAGE provides researchers with a comprehensive toolkit for protein characterization—from primary structure determination to functional complex analysis. As drug development increasingly focuses on complex biologics and targeted therapies, the ability to study protein higher-order structure and interactions under non-denaturing conditions becomes ever more critical. The continued evolution of Native PAGE methodologies and their integration with advanced analytical techniques ensures this decades-old technique will remain relevant for addressing contemporary challenges in proteomics and therapeutic development.
In the field of protein separation research, the choice of electrophoresis technique fundamentally shapes experimental outcomes. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Native PAGE serve distinct purposes in biochemical analysis, with their differences primarily emanating from their specific chemical components [6]. These key chemicals—SDS, buffers, and reducing agents—dictate whether proteins maintain their native conformation or become denatured for molecular weight-based separation [5]. This technical guide examines the core chemical components that define these methodologies, providing researchers and drug development professionals with a detailed framework for selecting appropriate separation strategies based on their experimental objectives, whether for protein characterization, functional studies, or therapeutic development.
Core Chemical Components in PAGE Techniques
Sodium Dodecyl Sulfate (SDS): The Denaturing Agent
SDS serves as the foundational chemical that enables SDS-PAGE to separate proteins based primarily on molecular weight. This anionic detergent performs multiple critical functions simultaneously [21]:
- Protein Denaturation: SDS disrupts hydrogen bonds and van der Waals forces, effectively unraveling tertiary and secondary protein structures into linear polypeptides [22].
- Charge Masking: By binding uniformly to the hydrophobic regions of proteins at an approximate ratio of 1.4g SDS per 1g of protein, SDS confers a uniform negative charge density that overwhelms proteins' intrinsic charges [6] [21].
- Molecular Shape Standardization: The linearizing effect of SDS creates consistent rod-like shapes for all proteins, eliminating conformational differences that might otherwise affect migration [5].
For effective SDS binding, several conditions must be met: SDS monomer concentration must exceed 1mM, ionic strength must remain low (10-100mM), and disulfide bonds must be reduced to allow complete unfolding [21]. This comprehensive action of SDS ensures that electrophoretic mobility depends almost exclusively on molecular weight rather than native charge or structure [17].
Electrophoresis Buffers: pH Control and Ion Management
Buffer systems create the pH environment and ionic conditions necessary for controlled protein migration during electrophoresis. Different buffer formulations have been developed to optimize separation for various protein size ranges:
Table: Buffer Systems for Protein Electrophoresis
| Buffer System | Optimal Separation Range | Key Characteristics | Primary Applications |
|---|---|---|---|
| Tris-Glycine | 10-300 kDa | Classical discontinuous system; sharp bands but alkaline pH limits shelf life | General purpose protein separation |
| Tris-Acetate | Up to 500 kDa | Suitable for high molecular weight proteins | Large proteins and complexes |
| Tricine | 1-30 kDa | Improved resolution for low molecular weight separations | Small proteins and peptides |
| Bis-Tris | Wide range | Neutral pH extends gel shelf life; sharper bands | Pre-cast gels; long-term storage |
The Tris-Glycine system employs a discontinuous buffer approach where the stacking gel (pH ~6.8) and separating gel (pH ~8.8) create ion movement boundaries that concentrate proteins before separation [23]. This stacking effect produces sharper bands and improved resolution. More recent developments like Bis-Tris buffers (pH ~6.5-7.0) offer enhanced stability and reduced gel degradation for commercial pre-cast applications [21].
Reducing Agents: Disulfide Bond Cleavage
Reducing agents are essential components specifically in SDS-PAGE sample preparation that break covalent disulfide linkages between cysteine residues:
- β-Mercaptoethanol (2-5% in sample buffer): A potent reducing agent that converts disulfide bonds to free sulfhydryl groups, enabling complete protein unfolding [23].
- Dithiothreitol (DTT) (2-3% in sample buffer): A more stable alternative with less odor, similarly effective in reducing disulfide bridges [21].
These agents are typically included in the SDS-PAGE sample buffer and become activated during the 95-100°C heating step that precedes electrophoresis [23]. The combination of reducing agents with SDS ensures complete denaturation and linearization of proteins. In contrast, Native PAGE protocols deliberately omit reducing agents to preserve native protein structure and maintain subunit interactions within protein complexes [6] [10].
Diagram: Chemical Workflow in PAGE Techniques
Comparative Analysis of Chemical Components
The distinct chemical compositions of SDS-PAGE and Native PAGE directly determine their separation mechanisms and subsequent applications in research.
Table: Comprehensive Comparison of Chemical Components
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| SDS Presence | Present (1-2% in gel and buffer) [21] | Absent [6] |
| Reducing Agents | β-mercaptoethanol (4-5%) or DTT (2-3%) in sample buffer [21] | None present [6] |
| Sample Preparation | Heating at 95-100°C for 5 minutes [23] | No heating, samples kept at 4°C [6] |
| Buffer System | Varies (Tris-Glycine, Bis-Tris, etc.) with SDS [21] | Similar systems but without SDS [6] |
| Separation Basis | Molecular weight only [17] | Size, charge, and shape [5] |
| Protein State | Denatured and linearized [22] | Native, folded conformation [6] |
| Protein Function Post-Separation | Lost [5] | Preserved [5] |
| Protein Recovery | Not typically functional [6] | Possible with retained activity [6] |
The presence or absence of SDS creates fundamentally different separation environments. In SDS-PAGE, the uniform negative charge from SDS creates a consistent charge-to-mass ratio across all proteins, making molecular weight the sole determinant of migration distance through the gel matrix [17]. In Native PAGE, proteins retain their intrinsic charges, which vary based on amino acid composition, resulting in separation influenced by both molecular size and net charge at the running pH [6] [19].
The temperature differential during electrophoresis further distinguishes these techniques. SDS-PAGE typically runs at room temperature, while Native PAGE often requires 4°C to maintain protein stability and prevent denaturation during separation [6]. This temperature control is essential for preserving labile protein complexes in their functional state throughout the Native PAGE process.
Experimental Protocols and Methodologies
SDS-PAGE Sample Preparation Protocol
The denaturing nature of SDS-PAGE requires specific sample treatment to ensure complete protein denaturation and reduction:
Sample Dilution: Mix protein sample with SDS-PAGE sample buffer (typically 1:1 to 1:4 ratio) containing:
- 2% SDS (w/v)
- 5% β-mercaptoethanol (v/v) or 100mM DTT
- 10% glycerol (v/v)
- 0.002% bromophenol blue (w/v)
- 62.5mM Tris-HCl, pH 6.8 [23]
Denaturation: Heat samples at 95-100°C for 5 minutes in a dry bath or water bath to ensure complete protein denaturation and SDS binding [23].
Cooling and Centrifugation: Briefly centrifuge heated samples (10-15 seconds at 10,000×g) to collect condensation and ensure uniform sample distribution [23].
Gel Loading: Load 10-40μL of prepared sample into gel wells, including molecular weight markers for calibration [23].
Critical considerations include using fresh reducing agents (particularly β-mercaptoethanol, which oxidizes over time) and ensuring sufficient SDS concentration (at least 3-4× the protein mass by weight) for complete coating [21].
Native PAGE Sample Preparation Protocol
Native PAGE maintains protein structure through gentle processing:
Sample Preparation: Mix protein sample with native sample buffer containing:
- No SDS or other denaturants
- 10-20% glycerol (for density)
- Tracking dye (bromophenol blue or similar)
- Appropriate cofactors if needed for stability [6]
Non-denaturing Conditions: Omit heating step entirely. Keep samples at 4°C throughout preparation to maintain protein stability [6].
Buffer Compatibility: Dialyze samples into low-ionic strength running buffer if necessary to prevent distortion during electrophoresis.
Gel Loading: Load samples directly into gel wells without prior heating [6].
The key to successful Native PAGE lies in maintaining the protein's native state throughout the process, which may require optimization of pH, buffer composition, and temperature for different protein systems.
Impact on Protein Separation and Research Applications
Protein Migration Patterns and Data Interpretation
The different chemical environments of SDS-PAGE and Native PAGE produce distinct migration patterns that inform protein characterization:
In SDS-PAGE, migration distance correlates with molecular weight, allowing size estimation by comparison to protein standards [24]. The denatured, linear proteins migrate through the polyacrylamide matrix with smaller proteins moving faster than larger ones [23].
In Native PAGE, migration depends on both size and charge, creating more complex migration patterns where the same protein may appear at different positions depending on the gel pH and its intrinsic charge [6]. This property makes Native PAGE particularly valuable for studying charge variants of proteins.
A compelling case study demonstrates how combining these techniques reveals protein quaternary structure: a protein migrated at 120 kDa in Native PAGE but at 60 kDa in non-reducing SDS-PAGE, indicating a non-covalent dimeric structure composed of two 60 kDa subunits [10]. This information would be inaccessible using either technique alone.
Application-Specific Methodology Selection
The choice between SDS-PAGE and Native PAGE depends fundamentally on research objectives:
SDS-PAGE is optimal for:
- Molecular weight determination [6]
- Assessing protein purity and homogeneity [5]
- Western blot analysis, as denaturation exposes linear epitopes [22]
- Protein expression profiling [6]
Native PAGE is preferred for:
- Studying native protein complexes and quaternary structure [10]
- Enzyme activity assays after separation [5]
- Analyzing protein-protein interactions [22]
- Purification of functional proteins [6]
Diagram: Technique Selection Based on Research Goals
The Scientist's Toolkit: Essential Research Reagents
Successful protein electrophoresis requires specific chemical reagents optimized for each technique:
Table: Essential Reagents for Protein Electrophoresis
| Reagent Category | Specific Examples | Function | Technical Notes |
|---|---|---|---|
| Denaturing Agents | SDS (Sodium dodecyl sulfate) | Denatures proteins; confers uniform negative charge | Use electrophoresis grade; critical concentration >1mM [21] |
| Reducing Agents | β-mercaptoethanol, DTT (Dithiothreitol) | Breaks disulfide bonds | Fresh preparation recommended; DTT more stable [21] |
| Gel Matrix Components | Acrylamide, Bis-acrylamide (29:1 ratio) | Forms polyacrylamide gel matrix | Neurotoxic until polymerized; handle with gloves [23] |
| Polymerization Catalysts | APS (Ammonium persulfate), TEMED | Initiates and accelerates gel polymerization | TEMED has strong odor; prepare APS fresh or store frozen [21] |
| Buffer Systems | Tris-glycine, Tris-acetate, Tricine, Bis-Tris | Maintains pH and provides ions for conduction | Choice depends on protein size range [21] |
| Tracking Dyes | Bromophenol blue, Xylene cyanol | Visualizes migration progress | Does not bind proteins; migrates ahead of small proteins |
| Staining Reagents | Coomassie Brilliant Blue, Silver stain, SYPRO Ruby | Visualizes separated proteins | Silver stain most sensitive (~1 ng); Coomassie ~50 ng [24] |
Additional specialized reagents for Native PAGE include Coomassie G-250 for Blue Native PAGE (which adds negative charge to proteins without complete denaturation) and various cofactors that may be necessary to maintain protein stability during separation [6].
The strategic application of specific chemical components—SDS, buffers, and reducing agents—defines the fundamental capabilities and limitations of both SDS-PAGE and Native PAGE. SDS-PAGE employs a denaturing chemistry that standardizes protein charge and structure, making it ideal for molecular weight determination and analytical applications where protein denaturation is acceptable or desirable. In contrast, Native PAGE utilizes non-denaturing conditions that preserve native protein structure and function, enabling the study of protein complexes and functional attributes. The informed selection between these techniques, and potential use of both in complementary approaches, provides researchers with powerful tools for comprehensive protein characterization in both basic research and drug development applications.
Within the context of protein separation research, the fundamental choice between SDS-PAGE and Native PAGE dictates whether proteins are analyzed in a denatured or natively preserved state. This distinction is not merely technical but fundamentally shapes the type of biological information that can be extracted, thereby influencing subsequent conclusions in biochemical research and drug development. Polyacrylamide gel electrophoresis (PAGE) serves as a cornerstone technique, but its various forms—particularly denaturing SDS-PAGE and non-denaturing Native PAGE—diverge dramatically in their impact on protein structure [12]. These methodologies offer researchers complementary tools: one for deconstructing proteins into their constituent polypeptides, and the other for probing functional complexes in their biologically active forms. This guide examines the core principles, methodological specifics, and practical applications of these techniques, providing a framework for selecting the appropriate approach based on research objectives in protein analysis.
Fundamental Principles and Structural Impact
The separation mechanisms of SDS-PAGE and Native PAGE rest on fundamentally different interactions with protein structures, leading to distinct outcomes in structural preservation.
SDS-PAGE: Complete Denaturation and Charge Manipulation
In SDS-PAGE, the anionic detergent sodium dodecyl sulfate (SDS) plays a destructive yet systematic role in protein denaturation. When protein samples are heated to 70–100°C in the presence of SDS and a reducing agent like β-mercaptoethanol or dithiothreitol (DTT), several transformative events occur [4] [3]. The SDS molecules bind to the hydrophobic regions of the polypeptide backbone in a constant weight ratio of approximately 1.4 g SDS per 1 g of protein [4]. This binding confers a uniform negative charge to the polypeptides, effectively masking their intrinsic electrical charges [4] [12]. Concurrently, the reducing agent cleaves disulfide bonds, while the heat disrupts hydrogen bonds and van der Waals forces, collectively destroying tertiary and quaternary structures [4]. The result is the complete unfolding of proteins into linear, rod-like SDS-polypeptide complexes that migrate through the polyacrylamide gel matrix based almost exclusively on molecular mass [25] [12]. This process renders proteins biologically inactive but allows for precise molecular weight determination.
Native PAGE: Preservation of Native Architecture
In stark contrast, Native PAGE employs non-denaturing conditions without SDS or reducing agents [6] [25]. Proteins remain in their folded, native conformations throughout the separation process, preserving their secondary, tertiary, and quaternary structures [12]. Consequently, multimeric proteins maintain their subunit interactions, and enzymes often retain their catalytic activity after separation [12]. The migration of proteins through the gel depends on a combination of factors including the protein's intrinsic net charge at the running buffer pH, its molecular size, and its three-dimensional shape [26] [12]. Proteins with higher negative charge density migrate faster, as do smaller proteins that experience less frictional resistance from the gel matrix [12]. This preservation of native structure enables the study of protein complexes and functional properties, though it complicates molecular weight determination due to the influence of charge and conformation on migration.
Comparative Analysis: Separation Characteristics
The table below summarizes the key differences in separation characteristics and outcomes between SDS-PAGE and Native PAGE.
Table 1: Comparative Analysis of SDS-PAGE and Native PAGE Separation Characteristics
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| Gel Nature | Denaturing [19] [6] | Non-denaturing [19] [6] |
| Sample Preparation | Heating with SDS and reducing agents [6] [4] | No heating; no denaturants [6] [25] |
| Protein State | Denatured, linearized polypeptides [25] [3] | Native, folded conformation [25] [5] |
| Separation Basis | Molecular mass [19] [12] | Net charge, size, and shape [26] [12] |
| Charge Manipulation | SDS imposes uniform negative charge [4] [3] | Relies on intrinsic protein charge [25] [12] |
| Quaternary Structure | Disrupted; subunits separate [4] [5] | Preserved; complexes remain intact [25] [12] |
| Biological Activity | Lost [25] [5] | Often retained [25] [12] |
| Protein Recovery | Non-functional polypeptides [19] [6] | Potentially functional, active proteins [19] [12] |
| Primary Applications | Molecular weight determination, purity assessment, western blotting [6] [3] | Study of protein complexes, oligomerization, native function [25] [5] |
Detailed Methodological Protocols
Robust, reproducible results depend on strict adherence to standardized protocols for each technique. The workflows for SDS-PAGE and Native PAGE are illustrated below.
SDS-PAGE Experimental Protocol
Sample Preparation:
- Lysis: Solubilize cells or tissue in a buffer containing 1% SDS to immediately denature proteins and inhibit proteases [4].
- Reduction and Denaturation: Mix the protein sample with a Laemmli-style sample buffer containing final concentrations of 1x SDS (2-4%), 50-100 mM DTT or 5% β-mercaptoethanol, 10% glycerol, and 62.5 mM Tris-HCl at pH 6.8 [4] [3]. Heat the mixture at 95°C for 5 minutes (or 70°C for 10 minutes) to ensure complete denaturation and reduction of disulfide bonds [4].
- Cooling: Briefly centrifuge the heated samples to bring down condensation before loading onto the gel.
Gel Electrophoresis:
- Gel Selection: Choose an appropriate acrylamide concentration based on target protein size: 8% for large proteins (25-200 kDa), 10% for standard separation (15-100 kDa), or 12-15% for small proteins (<50 kDa) [3]. Precast gradient gels (e.g., 4-12% or 4-20%) provide a broader separation range [4] [3].
- Electrophoresis Conditions: Load equal amounts of protein (5-50 μg) per well alongside a molecular weight marker. Run the gel in an SDS-containing Tris-glycine running buffer (25 mM Tris, 192 mM glycine, 0.1% SDS, pH 8.3) at constant voltage (100-150 V) for approximately 40-60 minutes, or until the dye front reaches the bottom of the gel [3].
Native PAGE Experimental Protocol
Sample Preparation:
- Gentle Lysis: Use mild, non-ionic detergents (e.g., digitonin) or osmotic shock in a cold isotonic buffer to preserve protein complexes without denaturation [27]. Maintain samples at 4°C throughout preparation.
- Native Buffer: Prepare samples in a non-denaturing buffer containing 50 mM Bis-Tris, 50 mM NaCl, 10% glycerol, and pH 7.2 [7]. Crucially, omit SDS, reducing agents, and heating steps [6] [25].
Gel Electrophoresis:
- Gel Selection: Use lower acrylamide percentages (e.g., 4-16% gradient gels) to accommodate folded proteins and protein complexes [27] [7].
- Electrophoresis Conditions: Load samples into wells and run in a Tris-based running buffer without SDS. For Blue Native (BN)-PAGE, the cathode buffer contains Coomassie G-250 dye, which confers a negative charge shift to membrane proteins [27]. For Clear Native (CN)-PAGE, mixed micelles of anionic and neutral detergents replace the dye to avoid interference with downstream activity assays [27]. Run electrophoresis at constant voltage (e.g., 150 V) at 4°C to maintain protein stability and function [6].
The Scientist's Toolkit: Essential Reagents and Materials
Successful execution of electrophoretic separations requires specific reagents, each serving a distinct function in the process.
Table 2: Key Research Reagent Solutions for PAGE Techniques
| Reagent/Material | Function | SDS-PAGE | Native PAGE |
|---|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins; imparts uniform negative charge [4] [3] | Essential | Not Used |
| DTT/β-mercaptoethanol | Reduces disulfide bonds [4] | Essential | Not Used |
| Coomassie G-250 Dye | Provides charge shift for membrane proteins [27] | Not Used | Used in BN-PAGE |
| Digitonin | Mild detergent for solubilizing membrane complexes [27] | Not Used | Optional |
| Acrylamide/Bis-acrylamide | Forms the porous gel matrix [4] [12] | Essential | Essential |
| APS and TEMED | Catalyzes gel polymerization [4] [12] | Essential | Essential |
| Molecular Weight Markers | Calibrates gel for size estimation [4] [12] | Essential (Denatured) | Used (Native) |
| Tris-based Buffers | Maintains pH during electrophoresis [4] [12] | Essential | Essential |
| Glycerol | Increases sample density for gel loading [4] | Essential | Essential |
Downstream Applications and Data Interpretation
The choice of electrophoresis method directly enables specific downstream analytical techniques and dictates how results must be interpreted.
Applications Enabled by SDS-PAGE
- Molecular Weight Determination: The primary application, achieved by comparing protein migration distances to a standard curve generated by known molecular weight markers run on the same gel [3] [12]. The linear relationship between log molecular weight and relative migration distance (Rf) allows for size estimation.
- Western Blotting: Following separation, proteins are transferred to a membrane for immunodetection with specific antibodies [26] [3]. The denatured, linearized epitopes are often more accessible for antibody binding.
- Purity Assessment and Quantification: A single, sharp band suggests a pure protein, while multiple bands indicate contaminants [3]. Staining intensity with Coomassie, silver, or fluorescent dyes can be used for semi-quantitative analysis via densitometry [3].
Applications Enabled by Native PAGE
- In-Gel Enzyme Activity Staining: Specific substrates and colorimetric detection methods can visualize active enzymes directly in the gel, confirming functional preservation after separation [27]. This is particularly valuable for complexes like those in the mitochondrial oxidative phosphorylation system [27].
- Analysis of Protein Complexes and Oligomeric States: BN-PAGE and CN-PAGE are indispensable for studying native protein-protein interactions, supercomplex formation (e.g., respiratory chain complexes I, III, and IV), and determining stoichiometry [27].
- Functional Protein Purification: As proteins retain their native state, they can be electro-eluted from native gels for use in functional assays, antibody production, or structural studies [12].
Technical Considerations and Troubleshooting
Optimizing electrophoretic separations requires attention to critical parameters and awareness of common pitfalls.
Critical Parameters for Success
- pH Control: In SDS-PAGE, a discontinuous buffer system with a stacking gel at pH ~6.8 and a resolving gel at pH ~8.8 creates a stacking effect that sharpens bands [4]. For Native PAGE, the buffer pH must be carefully selected to maintain protein solubility and native charge [12].
- Acrylamide Concentration: The appropriate pore size is critical for resolution. High-percentage gels better resolve small proteins, while low-percentage gels are optimal for large proteins and complexes [3] [12].
- Temperature: SDS-PAGE is typically run at room temperature. Native PAGE is best performed at 4°C to minimize protein denaturation and proteolytic activity [6].
Troubleshooting Common Issues
- Smiling or Frowning Bands: Often caused by uneven heating during electrophoresis. Ensure the gel apparatus is properly connected and that the buffer level is even across the gel [26] [3].
- Poor Resolution or Smearing: In SDS-PAGE, this can result from insufficient denaturation (re-heat samples with fresh DTT) or overloading. In Native PAGE, aggregation can cause smearing [26] [3].
- Atypical Banding Patterns: In SDS-PAGE, unexpected bands may indicate protein degradation, proteolysis, or post-translational modifications. Protease inhibitors in the lysis buffer can help [26]. In Native PAGE, multiple bands may represent genuine oligomeric states of the same protein.
Advanced Techniques and Future Directions
Electrophoresis technology continues to evolve with hybrid methodologies and refined applications.
- Native SDS-PAGE (NSDS-PAGE): A modified technique that reduces SDS concentration and omits heating and reducing agents, allowing some proteins to retain bound metal ions and enzymatic activity while maintaining high resolution [7].
- Two-Dimensional (2D) PAGE: This powerful method combines isoelectric focusing (IEF) with SDS-PAGE, separating proteins first by their isoelectric point (pI) and then by molecular weight. This provides the highest resolution for analyzing complex protein mixtures [12].
- Mass Spectrometry Compatibility: Gel bands excised from both SDS-PAGE and Native PAGE can be analyzed by mass spectrometry for protein identification. However, the Coomassie dye from BN-PAGE must be removed prior to analysis [27].
Practical Guide: When and How to Use Each Technique
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) is a foundational analytical technique in biochemistry and molecular biology laboratories worldwide. Developed by Laemmli in 1970, this method enables researchers to separate complex protein mixtures based primarily on molecular weight [6] [11]. The technique's robustness and relative simplicity have made it indispensable for diverse applications ranging from protein purity assessment and molecular weight determination to western blotting and quality control in food and pharmaceutical sciences [26] [11].
This technical guide provides a comprehensive SDS-PAGE protocol framed within the broader context of protein separation research, specifically contrasting SDS-PAGE with native PAGE methodologies. Understanding these complementary techniques empowers researchers to select the optimal approach for their specific experimental questions, whether studying protein size under denaturing conditions or investigating native structure-function relationships.
Principles of SDS-PAGE
SDS-PAGE separates proteins through the combined effects of a polyacrylamide gel matrix and the anionic detergent sodium dodecyl sulfate (SDS). The fundamental separation mechanism relies on two key principles:
Protein Denaturation and Uniform Charge Conferral: SDS binds to hydrophobic regions of proteins at a consistent ratio of approximately 1.4 g SDS per 1 g of protein [12]. This binding denatures proteins into linear polypeptide chains while masking their intrinsic charges. The result is that all SDS-bound proteins carry a strong negative charge roughly proportional to their polypeptide length [6] [17].
Molecular Sieving: The polyacrylamide gel matrix creates a porous network through which proteins migrate under an electric field. Smaller proteins navigate these pores more easily and migrate faster, while larger proteins encounter greater resistance and migrate more slowly [26] [12]. This molecular sieving effect separates proteins primarily by molecular weight with minimal influence from their original charge or structure [17].
Materials and Reagents
Research Reagent Solutions
The following table details essential reagents required for SDS-PAGE:
Table 1: Essential Reagents for SDS-PAGE
| Reagent | Function | Typical Composition/Notes |
|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked polymer gel matrix that acts as a molecular sieve [12]. | Concentration determines pore size (e.g., 12% for 40-100 kDa proteins) [26]. |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers uniform negative charge [6]. | Added to sample buffer and running buffer [12]. |
| Tris-HCl Buffer | Maintains stable pH during electrophoresis to prevent protein damage [26]. | Different pH for stacking (∼6.8) and resolving (∼8.8) gels [26]. |
| Ammonium Persulfate (APS) & TEMED | Catalyzes acrylamide polymerization [12]. | TEMED stabilizes the free radical polymerization initiated by APS [12]. |
| Reducing Agents (DTT or β-mercaptoethanol) | Breaks disulfide bonds to fully denature proteins [6] [11]. | Added to sample buffer for "reducing SDS-PAGE" [11]. |
| Glycerol | Increases sample density for facile well loading [7]. | Component of sample loading buffer. |
| Tracking Dye (Bromophenol Blue) | Visualizes migration progress during electrophoresis [28]. | Added to sample buffer. |
| Coomassie Brilliant Blue | Stains separated proteins for visualization post-electrophoresis [28]. |
Step-by-Step Protocol
Gel Preparation
Polyacrylamide gels consist of two distinct layers: a resolving (separating) gel and a stacking gel.
1. Prepare the Resolving Gel:
- Choose an appropriate acrylamide concentration based on your target protein sizes (see Table 2) [26].
- Combine components in the following order: water, acrylamide/bis-acrylamide solution, Tris buffer (pH ∼8.8), SDS, ammonium persulfate, and finally TEMED. Mix gently to avoid introducing bubbles.
- Immediately pipette the solution into gel cassettes, leaving space for the stacking gel. Carefully overlay with isopropanol or water to create a flat interface.
- Allow complete polymerization (typically 20-30 minutes).
Table 2: Acrylamide Concentration Guidelines
| Acrylamide Percentage | Optimal Protein Separation Range |
|---|---|
| 15% | 10 - 50 kDa [26] |
| 12% | 40 - 100 kDa [26] |
| 10% | >70 kDa [26] |
| 4-20% (Gradient) | Broad range (e.g., 10-200 kDa) [26] |
2. Prepare and Cast the Stacking Gel:
- Once the resolving gel has polymerized, remove the overlay liquid.
- Prepare a lower percentage acrylamide solution (typically 4-5%) with Tris buffer at pH ∼6.8 [12].
- Add SDS, APS, and TEMED, then pipette onto the polymerized resolving gel.
- Immediately insert a clean comb, avoiding air bubbles.
- Allow to polymerize completely before carefully removing the comb.
Sample Preparation
Proper sample preparation is critical for successful separation:
- Dilute Protein Sample: Mix protein extract with an appropriate volume of buffer. Keep salt concentrations below 500 mM to prevent smearing [26].
- Add SDS Loading Buffer: Combine the diluted sample with SDS-PAGE sample buffer containing SDS, glycerol, tracking dye (e.g., bromophenol blue), and Tris buffer. For reducing SDS-PAGE, include a reducing agent like DTT (dithiothreitol) or β-mercaptoethanol [11].
- Denature Proteins: Heat the sample at 70-100°C for 5-10 minutes [26] [12]. This step ensures complete protein denaturation and SDS binding.
- Centrifuge: Briefly spin the heated samples to collect condensation.
Electrophoresis Setup and Execution
The workflow below outlines the complete SDS-PAGE procedure:
Diagram 1: SDS-PAGE Workflow from Gel Casting to Visualization.
Assemble Electrophoresis Apparatus:
- Place the polymerized gel cassette into the electrophoresis chamber.
- Fill inner and outer chambers with running buffer (e.g., Tris-glycine buffer containing 0.1% SDS) [12].
Load Samples and Molecular Weight Markers:
- Using gel-loading tips, carefully pipette prepared protein samples into individual wells.
- Load one well with a molecular weight marker (protein ladder) for size calibration [26].
Execute Electrophoretic Run:
- Connect the apparatus to a power supply with the cathode (black) at the top and anode (red) at the bottom.
- Apply constant voltage: 120-200V for mini-gels [28].
- Run until the tracking dye front reaches the bottom of the gel (typically 1-1.5 hours).
- Terminate the run before the dye front runs off the gel.
Protein Visualization
Following electrophoresis, separate proteins are invisible within the gel and must be stained for detection:
- Coomassie Staining:
- Carefully remove the gel from its cassette.
- Immerse the gel in Bio-Safe Coomassie Stain for 1 hour with gentle agitation [28].
- For higher sensitivity, stain overnight.
- Destain with distilled water until protein bands are clear against a light background.
Troubleshooting Common Issues
Table 3: Common SDS-PAGE Issues and Solutions
| Issue | Potential Cause | Solution |
|---|---|---|
| Smeared Bands | Incomplete denaturation; high salt concentration [26]. | Add fresh reducing agent; boil samples for 5 min; reduce salt concentration. |
| "Smiling" Bands | Buffer/gel overheating during run [26]. | Check running buffer composition; run at correct voltage. |
| Weak/Faint Bands | Protein concentration too high or too low [26]. | Determine protein concentration before loading (Bradford/BCA assay). |
| Unexpected Bands | Protein degradation, modification, or aggregation [26]. | Use protease/phosphatease inhibitors; include fresh reducing agents. |
SDS-PAGE vs. Native PAGE: A Critical Comparison
SDS-PAGE and Native PAGE represent two complementary approaches for protein separation, each with distinct advantages and applications. The table below summarizes their key differences:
Table 4: Comprehensive Comparison of SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight only [6] [17] | Size, overall charge, and 3D shape [6] [5] |
| Protein State | Denatured and linearized [6] [12] | Native, folded conformation [6] [5] |
| SDS Presence | Present in gel and buffers [6] | Absent [6] |
| Reducing Agents | Typically present (DTT/BME) [6] | Absent [6] |
| Sample Preparation | Heating required (70-100°C) [6] | No heating [6] |
| Protein Function Post-Run | Lost [6] | Retained [6] [5] |
| Protein Recovery | Not recoverable in functional form [6] | Recoverable for activity studies [6] |
| Primary Applications | Molecular weight determination, purity check, western blotting [6] | Studying protein complexes, oligomerization, enzymatic activity [6] [5] |
| Typical Run Temperature | Room temperature [6] | 4°C [6] |
Choosing the Appropriate Technique: The experimental objective dictates the choice of electrophoresis method. SDS-PAGE is ideal for determining molecular weight, assessing sample purity, and preparing for western blotting [6] [12]. In contrast, Native PAGE is the method of choice for investigating protein-protein interactions, oligomeric state, conformational changes, and enzymatic activity in the native state [5] [10]. For example, if a protein runs as a 60 kDa band on non-reducing SDS-PAGE but as a 120 kDa band on Native PAGE, this suggests the native protein is a non-covalent dimer of two 60 kDa subunits [10].
SDS-PAGE remains a cornerstone technique in life sciences due to its reliability, robustness, and straightforward interpretation. This protocol provides a comprehensive guide from sample preparation through electrophoresis, enabling researchers to effectively separate and analyze proteins by molecular weight. The critical comparison with Native PAGE underscores the importance of methodological selection based on research questions. While SDS-PAGE excels in denaturing applications, Native PAGE offers unique advantages for functional and structural studies under non-denaturing conditions. Mastery of both techniques, along with emerging variations like NSDS-PAGE that aim to preserve some native functions [7], provides researchers with a powerful toolkit for comprehensive protein analysis in basic research, drug development, and diagnostic applications.
Within the landscape of protein electrophoresis, polyacrylamide gel electrophoresis (PAGE) serves as a foundational technique for separating and analyzing protein mixtures. The choice between its two primary forms—SDS-PAGE and Native PAGE—is dictated by the research objectives and fundamentally shapes the type of information that can be obtained [5]. This guide details the Native PAGE method, a technique designed to separate proteins based on their intrinsic charge, size, and shape while preserving their native conformation and biological activity [12].
The core distinction lies in the treatment of the protein sample. SDS-PAGE employs the denaturing detergent sodium dodecyl sulfate (SDS) and heat to unfold proteins, coat them with a uniform negative charge, and separate them primarily by molecular weight [12] [5]. In contrast, Native PAGE is performed in the absence of denaturing agents, allowing proteins to remain in their folded, functional state [12]. This preservation is critical for experiments aimed at studying protein-protein interactions, oligomeric states, enzymatic activity, and the presence of non-covalently bound cofactors, such as metal ions [7] [5]. While SDS-PAGE excels in determining molecular weight and assessing purity, Native PAGE provides a window into the functional proteome, making it an indispensable tool for researchers and drug development professionals investigating protein function and complex biology.
Core Principles and Key Differences from SDS-PAGE
Understanding the mechanistic differences between Native PAGE and SDS-PAGE is crucial for selecting the appropriate technique and correctly interpreting results. The following table summarizes the fundamental distinctions.
Table 1: Core Differences Between Native PAGE and SDS-PAGE
| Feature | Native PAGE | SDS-PAGE |
|---|---|---|
| Protein State | Native, folded structure retained [5] | Denatured, linearized subunits [12] |
| Separation Basis | Combined factors: intrinsic charge, size, and 3D shape [12] | Primarily by molecular mass (weight) [12] |
| Biological Activity | Retained post-separation; enzymes can remain active [7] [12] | Destroyed during denaturation [7] |
| Detergent (SDS) | Absent | Present in sample and running buffers [12] |
| Sample Preparation | No heating or reducing agents; often kept at 4°C [12] | Heated (typically 70-100°C) with SDS and reducing agents [12] |
| Molecular Weight Determination | Not straightforward due to charge/shape influence [5] | Highly effective; migration rate correlates with log(MW) [12] |
| Primary Applications | Studying protein complexes, oligomerization, enzymatic function, and protein-protein interactions [5] | Determining protein purity, subunit molecular weight, and expression analysis [12] [5] |
A key technical consideration is the buffer system. While standard Native PAGE uses no SDS, an advanced variant known as Native SDS-PAGE (NSDS-PAGE) uses drastically reduced SDS concentrations (e.g., 0.0375% in the running buffer) and omits denaturing steps (heating, EDTA) from the sample preparation. This modification can allow for high-resolution separation while still retaining metal ions and enzymatic activity in many proteins [7]. Another related technique, semi-native PAGE, also uses SDS in the gel but loads non-denatured protein samples, leading to separation based on differences in protein stability and allowing for the study of metal complex-protein interactions [29].
Detailed Native PAGE Protocol
Reagent and Buffer Formulations
The following "Scientist's Toolkit" lists essential materials and their specific functions for a standard Native PAGE experiment. Precise buffer composition is critical for maintaining native conditions.
Table 2: Research Reagent Solutions for Native PAGE
| Reagent/Material | Function & Key Characteristics |
|---|---|
| Acrylamide/Bis-acrylamide | Forms the porous gel matrix; pore size is determined by the total percentage of acrylamide [12]. |
| Ammonium Persulfate (APS) | Initiates the polymerization reaction of acrylamide to form the polyacrylamide gel [12]. |
| TEMED | Catalyst that accelerates the gel polymerization reaction by generating free radicals from APS [12]. |
| Native PAGE Sample Buffer | A non-denaturing buffer (e.g., containing Tris, glycerol, and a visible dye like Phenol Red) to prepare the sample for loading without disrupting protein structure [7]. |
| Native PAGE Running Buffer | A non-denaturing buffer (e.g., Bis-Tris/Tricine-based) that conducts current while maintaining a pH that preserves protein native state [7]. |
| Coomassie G-250 | A dye included in some native running buffers (e.g., cathode buffer) that can impart a slight negative charge to proteins for consistent migration [7]. |
Specific Buffer Compositions (Examples) [7]:
- Sample Buffer (4X): 50 mM BisTris, 50 mM NaCl, 16 mM HCl, 10% Glycerol, 0.001% Ponceau S, pH 7.2.
- Running Buffer - Cathode: 50 mM BisTris, 50 mM Tricine, 0.02% Coomassie G-250, pH 6.8.
- Running Buffer - Anode: 50 mM BisTris, 50 mM Tricine, pH 6.8.
Step-by-Step Experimental Procedure
Step 1: Gel Preparation Cast a polyacrylamide gel (typically 4-16% gradient or a single percentage) without adding SDS. A stacking gel with a lower percentage of acrylamide and lower pH is often used to concentrate the protein samples into sharp bands before they enter the resolving gel [12]. The gel is polymerized using APS and TEMED.
Step 2: Sample Preparation Keep all reagents and protein samples on ice or at 4°C to minimize proteolysis and denaturation. Mix the protein sample with a non-denaturing Native PAGE sample buffer. Crucially, do not heat the sample. Simply mix by pipetting or gentle vortexing. The glycerol in the buffer adds density for easy gel loading [7] [12].
Step 3: Electrophoresis Setup and Execution
- Assemble the gel cassette in the electrophoresis tank.
- Fill the inner (upper) and outer (lower) chambers with the appropriate anode and cathode running buffers [7].
- Load the prepared protein samples and native molecular weight standards into the wells.
- Connect the power supply and run the electrophoresis at a constant voltage (e.g., 150V).* To maintain protein integrity, it is advisable to run the gel in a cold room or with a cooling apparatus [12]. Continue the run until the dye front migrates to the bottom of the gel.
Step 4: Post-Electrophoresis Analysis Once separation is complete, proteins can be detected using various methods:
- Activity Staining: If the protein is an enzyme, the gel can be incubated with a specific substrate to detect a colored or fluorescent product, directly localizing active enzyme bands [12].
- Protein Staining: Standard stains like Coomassie Brilliant Blue or silver stain can be used to visualize all protein bands.
- Electro-elution: Functional, native proteins can be recovered from the gel for downstream assays using passive diffusion or electro-elution techniques [12].
Diagram 1: Native PAGE Experimental Workflow
Troubleshooting and Best Practices
Optimizing Separation and Maintaining Activity
Successful Native PAGE requires careful attention to conditions that preserve the native state of proteins. The following table outlines common challenges and recommended solutions.
Table 3: Native PAGE Troubleshooting Guide
| Problem | Potential Cause | Solution |
|---|---|---|
| Poor Resolution/Smearing | Protein aggregation or precipitation; proteolysis. | Keep samples cold; use fresh protease inhibitors; optimize gel percentage for protein size range [12]. |
| No Bands Detected | Protein did not enter gel or migrated off gel. | Verify protein concentration; check buffer polarity (proteins migrate toward anode); use a colored native marker to track migration [7]. |
| Loss of Enzymatic Activity | Protein denatured during procedure. | Ensure no SDS is present; avoid heating samples; run gel at 4°C; use appropriate pH buffers to avoid extremes [12]. |
| Artifactual Bands | Non-specific protein interactions or breakdown. | Include mild detergents (e.g., digitonin) in buffers if compatible with activity; ensure sample purity and freshness [12]. |
Key Considerations for Experimental Design
- pH Management: The running buffer pH is critical as it influences the protein's net charge and thus its migration. Avoid pH extremes that can irreversibly denature proteins [12].
- Gel Percentage: Low-percentage gels (e.g., 4-8%) are better for resolving large proteins and complexes, while high-percentage gels (e.g., 12-16%) are for smaller proteins. Gradient gels (e.g., 4-16%) provide the broadest resolution range [12].
- Advanced Techniques: For specific applications like analyzing metalloproteins, consider the NSDS-PAGE method, which offers a compromise between the high resolution of SDS-PAGE and the metal-retention capabilities of native conditions [7].
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) represents a foundational methodology in biochemical research for analyzing complex protein mixtures. This technique revolutionized protein science by enabling separation based primarily on molecular weight, providing critical insights into protein composition, expression, and identity. Within the broader context of protein separation research, SDS-PAGE stands in contrast to Native PAGE, which separates proteins based on both size and charge while maintaining their native conformation and biological activity [6] [5]. Where Native PAGE preserves protein structure and function for studying enzymatic activity, protein complexes, and protein-protein interactions, SDS-PAGE deliberately denatures proteins to focus exclusively on molecular weight characterization and analytical applications that do not require functional proteins [5] [12]. This technical guide explores the core applications of SDS-PAGE in molecular weight determination and western blotting, providing researchers with detailed methodologies, practical considerations, and comparative insights relevant to drug development and biomedical research.
Principles of SDS-PAGE
Fundamental Mechanisms
SDS-PAGE separates proteins through a combination of molecular sieving and uniform charge application. The key mechanistic principle involves the detergent sodium dodecyl sulfate (SDS), which binds to protein hydrophobic regions in a constant weight ratio (approximately 1.4 g SDS per 1 g of polypeptide) and confers a uniform negative charge that overwhelms proteins' intrinsic charge differences [30] [12]. This SDS binding occurs alongside sample treatment with reducing agents like dithiothreitol (DTT) or β-mercaptoethanol, which break disulfide bonds, and heating at 95-100°C, which disrupts hydrogen bonds [30]. These treatments collectively linearize proteins into rods of similar charge-to-mass ratios, ensuring separation occurs almost exclusively by molecular weight rather than charge or conformation [2] [26].
When an electric field is applied, the negatively charged SDS-protein complexes migrate toward the anode through the polyacrylamide gel matrix, which acts as a molecular sieve. Smaller proteins navigate the porous network more easily and migrate faster, while larger proteins encounter greater resistance and migrate more slowly [2] [12]. This results in distinct protein bands positioned according to molecular weight, enabling accurate size determination when compared to standards of known molecular weight [26] [12].
Discontinuous Gel System
SDS-PAGE employs a discontinuous buffer system with two distinct gel regions that enhance separation resolution. The stacking gel, with lower acrylamide concentration (typically 4-5%) and pH ~6.8, serves to concentrate heterogeneous protein samples into sharp bands before they enter the resolving gel [30] [12]. The resolving gel contains higher acrylamide concentration (variable based on target protein sizes) and higher pH (~8.8), where actual molecular weight-based separation occurs [12]. This two-phase system ensures proteins enter the resolving gel simultaneously as thin, concentrated bands, significantly improving resolution compared to a single-phase gel system [30].
Table 1: Key Reagents in SDS-PAGE and Their Functions
| Reagent | Function | Technical Considerations |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins; confers uniform negative charge | Critical for masking intrinsic protein charge; binds in constant ratio |
| DTT or β-mercaptoethanol | Reduces disulfide bonds | Fresh preparation needed as oxidizing agents lose efficacy |
| Acrylamide/bis-acrylamide | Forms porous gel matrix | Concentration determines pore size; neurotoxic in monomer form |
| APS and TEMED | Polymerization initiators and catalyst | TEMED stabilizes free radicals; APS generates persulfate radicals |
| Tris-glycine buffer | Conducting medium for electrophoresis | Maintains pH; glycine serves as trailing ion in stacking phase |
| Coomassie Blue/Silver stain | Protein visualization | Coomassie detects ~100 ng; silver detects ~1 ng protein |
Molecular Weight Determination
Methodology and Protocol
The determination of protein molecular weight using SDS-PAGE relies on comparing protein migration distances against standards of known molecular weights. The step-by-step methodology encompasses:
Sample Preparation: Protein samples are diluted in loading buffer containing SDS, reducing agents (DTT or β-mercaptoethanol), glycerol for density, and tracking dye (bromophenol blue) [30]. Samples are heated at 95-100°C for 3-5 minutes to ensure complete denaturation [2] [26]. For cell or tissue lysates, protease and phosphatase inhibitors are typically added to prevent degradation [31].
Gel Selection and Preparation: The appropriate acrylamide concentration is selected based on the target protein size range (Table 2). While hand-casting gels remains common, commercial pre-cast gels provide consistency and convenience [26] [32]. The gel cassette is assembled in the electrophoresis apparatus filled with running buffer (typically Tris-glycine with 0.1% SDS) [32].
Electrophoresis: Samples and molecular weight markers are loaded into wells. A constant voltage (100-200V) is applied until the dye front approaches the gel bottom (typically 1-2 hours for mini-gels) [2] [32]. The process is performed at room temperature, though some protocols recommend cooling to prevent heat-induced artifacts [6].
Visualization and Analysis: Following electrophoresis, proteins are fixed and stained with Coomassie Brilliant Blue, silver stain, or fluorescent dyes [12]. The migration distance of each protein band is measured from the well to band center. A standard curve is generated by plotting the logarithm of molecular weights of standard proteins against their migration distances (Rf values), enabling molecular weight interpolation for unknown proteins [26] [12].
Table 2: Gel Concentration Guidelines for Optimal Protein Separation
| Protein Size Range | Recommended Gel Percentage | Separation Characteristics |
|---|---|---|
| 4-40 kDa | 15-20% | High-density gel for small proteins |
| 12-45 kDa | 15% | Optimal for lower MW proteins |
| 10-70 kDa | 12.5% | Standard range for many proteins |
| 15-100 kDa | 10% | Broad intermediate range |
| 50-200 kDa | 8% | For larger proteins |
| >200 kDa | 4-6% | Low percentage for very large proteins |
Technical Considerations and Limitations
Several factors influence the accuracy of molecular weight determination by SDS-PAGE. Glycosylated or membrane proteins may exhibit anomalous migration due to incomplete SDS binding or unusual amino acid compositions, potentially leading to molecular weight inaccuracies of 10-20% [26]. The linear range of the standard curve typically spans 10-200 kDa, with reduced accuracy outside this range [32]. For multi-subunit proteins, SDS-PAGE under reducing conditions separates individual subunits, providing information about subunit composition rather than intact complex size [12]. This contrasts with Native PAGE, which preserves oligomeric structure and provides information about native molecular weight and quaternary structure [6] [5].
Western Blotting
Workflow and Technical Execution
Western blotting (immunoblotting) combines SDS-PAGE separation with specific antibody detection to identify target proteins within complex mixtures. The multi-stage workflow includes:
Protein Separation: Proteins are first separated by SDS-PAGE as described in Section 3.1, ensuring optimal resolution for the protein of interest through appropriate gel percentage selection [26] [33].
Electroblotting: Separated proteins are transferred from the gel to a solid membrane support (typically nitrocellulose or PVDF) using electrophoretic transfer [33]. The transfer apparatus is assembled in a "sandwich" configuration with the gel and membrane positioned between filter papers and porous pads, all submerged in transfer buffer. When current is applied, proteins migrate out of the gel and bind to the membrane, creating a replica of the separation pattern [33]. Transfer efficiency depends on protein size, gel concentration, transfer time, and buffer composition [33].
Blocking: The membrane is incubated with a blocking solution (non-fat dry milk, BSA, or commercial blocking reagents) to cover remaining protein-binding sites and prevent nonspecific antibody binding [33]. This critical step reduces background signal and improves the signal-to-noise ratio.
Antibody Incubation: The blocked membrane is sequentially probed with primary antibody (specific to the target protein) and enzyme- or fluorophore-conjugated secondary antibody (specific to the primary antibody host species) [33]. The indirect method using conjugated secondary antibodies provides signal amplification and flexibility [33]. Washes with buffer containing mild detergent (e.g., Tween-20) between incubations remove unbound antibodies [33].
Detection: For enzyme-conjugated antibodies (typically HRP or AP), incubation with appropriate substrates produces detectable signals. Chemiluminescent substrates generate light that can be captured on film or with digital imaging systems, while chromogenic substrates produce visible precipitate on the membrane [33]. Fluorescently labeled antibodies enable direct detection using appropriate imaging systems and offer multiplexing capabilities [33].
Applications and Methodological Variations
Western blotting serves multiple research applications beyond simple protein detection. It provides semi-quantitative data on protein abundance when proper controls and normalization are employed [33]. Loading controls (e.g., housekeeping proteins like β-actin or GAPDH) account for sample loading variations, while negative controls verify antibody specificity [26]. Western blotting can detect post-translational modifications using modification-specific antibodies, often combined with enzymatic treatments (e.g., phosphatases) to verify modification-dependent signals [31]. The technique also identifies protein isoforms and proteolytic fragments through characteristic banding patterns and size shifts [26].
Several methodological variations address specific research needs. For low-abundance proteins, wheat germ agglutinin (WGA) enrichment or immunoprecipitation prior to western blotting enhances detection sensitivity [31]. Fluorescent western blotting enables multiplex detection of multiple proteins simultaneously using antibodies from different host species or directly conjugated primary antibodies [33]. Quantitative western blotting employs careful standard curves and validated normalization strategies for more accurate quantification [33].
Table 3: Western Blot Troubleshooting Guide
| Problem | Potential Causes | Solutions |
|---|---|---|
| Weak/faint bands | Insufficient protein; low antibody affinity | Optimize protein loading; titrate antibodies; extend exposure |
| High background | Incomplete blocking; insufficient washing | Optimize blocking buffer; increase wash stringency; titrate antibodies |
| Multiple non-specific bands | Antibody cross-reactivity; protein degradation | Include negative controls; use fresh protease inhibitors |
| Smiling bands | Buffer/gel heating during electrophoresis | Run at lower voltage; ensure adequate cooling |
| No bands | Transfer issues; antibody incompatibility | Verify transfer with reversible stain; check antibody specifications |
Comparative Analysis: SDS-PAGE vs. Native PAGE
The strategic selection between SDS-PAGE and Native PAGE depends fundamentally on research objectives, as these techniques provide complementary protein information. SDS-PAGE excels in molecular weight determination, purity assessment, and protein expression analysis, offering high resolution and reproducibility for denatured proteins [6] [12]. Its denaturing nature, however, destroys native protein structure, enzymatic activity, and non-covalent interactions, limiting functional studies [6] [7]. In contrast, Native PAGE preserves protein folding, biological activity, and protein complexes, enabling investigation of oligomerization states, protein-protein interactions, and enzymatic function [6] [5]. The trade-off includes more complex migration patterns dependent on both size and charge, potentially lower resolution for complex mixtures, and challenges in molecular weight interpretation due to tertiary structure effects [5] [12].
Recent methodological advances have bridged these traditional distinctions. Native SDS-PAGE (NSDS-PAGE) modifies standard conditions by eliminating heating steps, reducing SDS concentration, and removing EDTA, resulting in improved retention of metal cofactors and enzymatic activity while maintaining reasonable separation resolution [7]. Blue Native PAGE (BN-PAGE) utilizes Coomassie dye to confer charge while preserving native structure, particularly useful for studying membrane protein complexes and oligomeric states [6] [7]. Two-dimensional electrophoresis combining Native PAGE or isoelectric focusing with subsequent SDS-PAGE provides comprehensive protein characterization, resolving complex mixtures by native charge or isoelectric point in the first dimension and molecular weight in the second [12].
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of SDS-PAGE and western blotting requires carefully selected reagents and materials. Key components include:
Separation Matrix: Polyacrylamide gels, typically in gradient or fixed percentages, provide the sieving matrix for protein separation. Hand-casting allows customization, while commercial pre-cast gels offer convenience and reproducibility [26] [32]. Agarose gels (0.5-1%) serve as alternatives for very large protein complexes (>700 kDa) [26].
Electrophoresis Equipment: Vertical gel electrophoresis systems with buffer chambers and compatible power supplies (capable of constant voltage/current) form the core instrumentation [2] [12]. Mini-gel systems (8×8 cm) enable rapid analysis, while larger formats enhance resolution for complex samples [12].
Transfer Systems: Wet/tank transfer apparatus provides high efficiency for diverse protein sizes; semi-dry systems offer convenience and speed; dry transfer systems eliminate buffer requirements [33]. Selection depends on protein characteristics and throughput needs.
Detection Platforms: Chemiluminescent imaging systems (CCD-based or film) provide high sensitivity; fluorescent scanners enable multiplexing; colorimetric detection requires minimal equipment but offers lower sensitivity [33].
Specialized Reagents: Protease and phosphatase inhibitors preserve sample integrity during preparation [31]. Enhanced chemiluminescent substrates increase detection sensitivity; reversible protein stains (Ponceau S) verify transfer efficiency; phospho-specific antibodies detect post-translational modifications [26] [33].
Validation Tools: Reference standards including pre-stained and unstained molecular weight markers enable size determination and transfer monitoring [26] [12]. Control lysates with known expression patterns verify antibody specificity; reference antibodies for housekeeping proteins facilitate normalization [26].
SDS-PAGE remains an indispensable tool in protein research, providing robust methodologies for molecular weight determination and western blot analysis. Its capacity to separate proteins based on size with high resolution and reproducibility has established it as a cornerstone technique in biochemistry, molecular biology, and drug development. When applied with understanding of its principles and limitations, and with appropriate consideration of alternative approaches like Native PAGE for functional studies, SDS-PAGE delivers critical insights into protein expression, identity, and modification. As protein analysis continues to evolve with advances in sensitivity, quantification, and multiplexing, SDS-PAGE maintains its fundamental role in the researcher's toolkit, bridging traditional protein chemistry with contemporary proteomic applications.
Within the realm of protein separation research, gel electrophoresis stands as a fundamental methodology. The choice between its two primary forms—Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Native PAGE—is dictated by the research question. While SDS-PAGE is the go-to technique for determining molecular weight and analyzing protein purity by denaturing proteins into uniform, negatively charged rods, Native PAGE serves a different, complementary purpose [6] [5]. It preserves proteins in their native, folded conformation, allowing for separation based on a combination of size, intrinsic charge, and shape [6]. This whitepaper details the applications of Native PAGE, focusing on its critical role in analyzing protein complexes and conducting functional studies, framed within the broader context of protein separation research. For researchers and drug development professionals, understanding this distinction is paramount for designing experiments aimed at probing protein function, interactions, and quaternary structure, rather than merely subunit composition.
The core difference lies in what is measured. SDS-PAGE provides information on the molecular weight of polypeptide chains, but Native PAGE provides insights into the functional, oligomeric state of a protein in solution [10]. This makes it an indispensable tool for characterizing biologically active proteins and their complexes in a state that closely mimics the physiological environment.
Core Principles and Comparative Advantages
The fundamental difference between these techniques stems from their sample treatment. In SDS-PAGE, the anionic detergent SDS denatures proteins and confers a uniform negative charge, while a reducing agent like DTT breaks disulfide bonds [6]. This means migration depends almost entirely on molecular mass. In contrast, Native PAGE uses no denaturing detergents or reducing agents, and samples are not heated [6]. Consequently, a protein's migration is determined by its size, overall charge, and three-dimensional shape [6].
The following table summarizes the key operational and outcome differences between the two techniques:
Table 1: Comparative Analysis of SDS-PAGE and Native PAGE
| Criteria | SDS PAGE | Native PAGE |
|---|---|---|
| Gel Type | Denaturing | Non-denaturing |
| SDS Presence | Present | Absent |
| Buffer Composition | Includes reducing agents (e.g., DTT, BME) | No denaturing or reducing agents |
| Sample Preparation | Heated | Not heated |
| Separation Basis | Molecular weight | Molecular size, overall charge, and shape |
| Protein Net Charge | Always negative | Can be positive or negative |
| Protein State | Denatured and linearized | Native, folded conformation |
| Protein Function Post-Separation | Lost | Retained |
| Protein Recovery | Not recoverable in functional form | Recoverable for downstream assays |
| Primary Applications | Determine molecular weight, check purity, study expression | Study protein structure, subunit composition, and function [6] |
This preservation of native structure is Native PAGE's greatest advantage. Because proteins retain their biological activity, they can be recovered from the gel for functional assays, activity stains, or further interaction studies [6] [5]. This is impossible with SDS-PAGE, where proteins are irreversibly denatured.
Key Applications in Protein Complex and Functional Analysis
Determining Oligomeric State and Quaternary Structure
A powerful application of Native PAGE is elucidating a protein's quaternary structure. A classic example involves comparing a protein's migration on non-reducing SDS-PAGE versus Native PAGE. On non-reducing SDS-PAGE, a protein may migrate as a band corresponding to 60 kDa, indicating its subunit mass. However, if the same protein migrates as a 120 kDa species on Native PAGE, it strongly infers that the native protein is a dimer of two 60 kDa subunits [10]. Furthermore, if this dimer is observed under non-reducing SDS conditions, it indicates the subunits are held together by non-covalent interactions (e.g., hydrophobic or electrostatic), not disulfide bonds, as SDS disrupts non-covalent interactions but leaves disulfides intact [10]. This simple comparative experiment provides immediate insight into protein architecture.
Studying Membrane Protein Complexes with GPCRs
Native PAGE is particularly valuable for studying challenging membrane proteins, such as G protein-coupled receptors (GPCRs), which are critical drug targets. A peer-reviewed protocol describes a Native PAGE assay to biochemically characterize agonist-dependent coupling between a solubilized GPCR and purified "mini-G" proteins [34]. This method uses high-resolution clear native electrophoresis (hrCNE) to visualize the formation of detergent-stable complexes between the receptor and the G protein [34]. The assay allows researchers to:
- Rapidly screen for successful complex formation, indicated by a mobility shift on the gel.
- Determine apparent affinities of agonists for the receptor-mini-G complex.
- Measure apparent affinities of mini-G proteins for the agonist-occupied receptor, providing a partial measure of agonist efficacy [34].
This method is relatively simple, cost-effective, and does not require purified receptors, making it accessible for many labs [34]. The workflow for this quantitative application is detailed in the diagram below.
Integration with Advanced Analytical Techniques
The utility of Native PAGE extends beyond the gel itself. Its compatibility with other native-state analytical methods creates powerful, multi-faceted workflows. For instance, membrane proteins reconstituted into peptidiscs (a detergent-free, native-like membrane mimetic) can be analyzed by Native PAGE and then subsequently ejected for analysis by native mass spectrometry (nMS) [35]. This combined approach allows researchers to study protein complexes, their oligomeric state, bound endogenous ligands (such as lipids or metabolites), and even drug binding directly from near-physiological environments [35] [36]. This synergy provides a more comprehensive picture of protein function and interaction networks.
Essential Reagents and Experimental Materials
Successful execution of a Native PAGE experiment, particularly for complex targets like membrane proteins, requires careful selection of reagents. The following table lists key materials and their functions based on cited protocols.
Table 2: Research Reagent Solutions for Native PAGE Experiments
| Reagent/Material | Function / Purpose | Example from Protocol |
|---|---|---|
| Membrane Mimetics | Solubilize and stabilize membrane proteins in a native-like state. | Peptidiscs [35], LMNG/CHS detergent [34] |
| Mini-G Proteins | Surrogate Gα subunits that stabilize GPCRs in an active state for coupling studies. | Used in GPCR-mini-G coupling assays [34] |
| Affinity Tags & Resins | For purifying the protein or complex of interest prior to electrophoresis. | Ni-NTA resin for His-tagged proteins [35] |
| Specialized Buffers | Provide the correct ionic environment and maintain protein stability without denaturation. | hrCNE buffers (e.g., with 6-amino hexanoic acid) [34] |
| Acrylamide/Bis-acrylamide | Forms the porous polyacrylamide gel matrix for separation. | Standard component of PAGE gels [34] |
| Protease Inhibitors | Prevent proteolytic degradation of the protein sample during preparation. | Essential for working with crude lysates or membrane preps [34] |
| Fluorescent Tags (e.g., EGFP) | Enable sensitive in-gel detection of proteins without denaturing staining. | EGFP-tagged receptor for direct visualization [34] |
Within the broader thesis of protein separation research, Native PAGE is not a competitor to SDS-PAGE but a specialized, powerful complement. While SDS-PAGE answers the fundamental question of "what is the size of the polypeptide chain?", Native PAGE addresses more complex biological questions: "What is the functional state of the protein?", "What complexes does it form?", and "How does it interact with its partners?" Its ability to preserve native structure and function makes it indispensable for studying oligomerization, protein-protein interactions, and the activity of membrane protein complexes like GPCRs. As drug discovery increasingly focuses on complex targets and functional mechanisms, Native PAGE, especially when integrated with other native analysis techniques, will remain a cornerstone of biochemical characterization for researchers and drug development professionals.
In the realm of protein separation research, the fundamental dichotomy between denaturing and native techniques frames a critical methodological choice. While SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis) has long been the workhorse for protein separation based primarily on molecular weight, it fundamentally dismantles protein complexes through denaturation, stripping proteins of their native structure, function, and interactions [19] [17] [5]. Native PAGE, in its various forms, addresses this limitation by preserving the native state of proteins during separation, enabling the study of intact multi-protein complexes, oligomeric states, and functional characteristics [6] [5]. Within this landscape, Blue Native PAGE (BN-PAGE) and Clear Native PAGE (CN-PAGE) have emerged as sophisticated advanced variants specifically designed for the high-resolution analysis of membrane protein complexes and metabolons, filling a crucial gap left by both standard native PAGE and SDS-PAGE [27] [37] [38].
These techniques are particularly indispensable for investigating the oxidative phosphorylation (OXPHOS) system in mitochondria, which comprises five multi-subunit complexes critical for cellular energy production [27]. First developed by Hermann Schägger in the 1990s, BN-PAGE has become an instrumental technique for gaining insights into the assembly pathways of these complexes, the composition of higher-order respiratory chain supercomplexes (respirasomes), and pathologic mechanisms in patients with monogenetic OXPHOS disorders [27]. The subsequent development of CN-PAGE provided a complementary approach that avoids potential interference from the Coomassie dye used in BN-PAGE, further expanding the toolbox for native proteomic analysis [27] [38]. This technical guide explores the principles, methodologies, and applications of these powerful techniques within the broader context of protein separation research.
Theoretical Foundations and Principles of Operation
Core Principles of BN-PAGE
The operational principle of BN-PAGE centers on the use of Coomassie Blue G-250 dye to impart a negative charge onto protein surfaces [27] [37] [39]. This anionic blue dye binds to hydrophobic protein surfaces through non-covalent interactions, imposing a negative charge shift that forces even basic proteins with hydrophobic domains to migrate towards the anode during electrophoresis at pH 7.0 [27]. This charge imposition serves a dual purpose: it provides the charge necessary for electrophoretic migration while simultaneously preventing aggregation of hydrophobic proteins by keeping them soluble in the absence of detergent during the electrophoresis process [27] [39]. The binding of Coomassie dye is proportional to the surface area of the complex, meaning that the charge-to-mass ratio remains relatively consistent, allowing for meaningful molecular weight estimations [39].
A critical aspect of BN-PAGE is the solubilization process, which typically employs mild nonionic detergents such as n-dodecyl-β-D-maltoside or digitonin to extract membrane proteins from biological membranes without dissociating the individual complexes [27] [37]. The choice of detergent is crucial; while n-dodecyl-β-D-maltoside effectively solubilizes individual OXPHOS complexes, the milder digitonin is preferred when preserving higher-order supercomplexes (respirasomes) is desired [27]. The extraction is supported by the addition of 6-aminocaproic acid, a zwitterionic salt with a zero net charge at pH 7.0 that does not interfere with electrophoresis but helps maintain complex integrity [27].
Core Principles of CN-PAGE
Clear Native PAGE represents a refinement of the original BN-PAGE technique, wherein the Coomassie blue G-250 dye is replaced by mixtures of anionic and neutral detergents in the cathode buffer [27]. Similar to Coomassie blue G-250, these mixed micelles induce a charge shift on membrane proteins to enhance their solubility and augment their electrophoretic migration towards the anode [27]. The most significant advantage of CN-PAGE is the absence of residual blue dye interference during downstream in-gel enzyme activity staining, as the Coomassie dye can sometimes inhibit enzymatic activity or interfere with spectroscopic detection methods [27]. However, this advantage may come at the cost of reduced resolution for some protein complexes compared to BN-PAGE, as the charge shift effect may be less uniform across different protein species.
Comparative Theoretical Framework
The following diagram illustrates the key decision points and experimental workflows for selecting and implementing BN-PAGE versus CN-PAGE:
Comparative Technical Specifications: BN-PAGE vs. CN-PAGE vs. SDS-PAGE
The selection of an appropriate electrophoretic technique requires a clear understanding of the capabilities and limitations of each method. The following table provides a comprehensive comparison of key technical parameters across BN-PAGE, CN-PAGE, and SDS-PAGE:
Table 1: Comparative Analysis of Electrophoretic Techniques for Protein Separation
| Parameter | BN-PAGE | CN-PAGE | SDS-PAGE |
|---|---|---|---|
| Separation Basis | Size, shape & dye binding [37] [39] | Native charge & size [27] [6] | Molecular weight only [19] [6] [17] |
| Gel Conditions | Non-denaturing [6] | Non-denaturing [6] | Denaturing [19] [6] |
| Charge Agent | Coomassie Blue G-250 [27] [37] | Mixed detergents [27] | SDS [19] [6] |
| Protein State | Native, folded complexes [37] [39] | Native, folded complexes [27] | Denatured, linearized [19] [6] |
| Complex Preservation | Yes (individual & supercomplexes) [27] | Yes [27] | No [19] [5] |
| Enzyme Activity | Retained (with possible dye interference) [27] | Retained (without dye interference) [27] | Lost [6] [5] |
| Molecular Weight Range | 100 kDa - 10 MDa [39] | Similar to BN-PAGE [27] | 5 - 250 kDa [19] |
| Typical Detergent | n-Dodecyl-β-D-maltoside / Digitonin [27] [37] | Various mild detergents [27] | SDS (strong denaturant) [19] [6] |
| Protein Recovery | Possible with native function [6] [5] | Possible with native function [27] [6] | Denatured, non-functional [19] [6] |
| Primary Applications | Respiratory complexes, supercomplex analysis, assembly studies [27] [37] | Enzyme activity assays, metal-binding studies [27] [7] | Molecular weight determination, purity checks [19] [6] |
Detailed Experimental Methodologies
Essential Research Reagent Solutions
The successful implementation of native electrophoresis requires specific reagent systems optimized for preserving protein complex integrity. The following table catalogues the essential materials and their functions in BN-PAGE and CN-PAGE protocols:
Table 2: Essential Research Reagent Solutions for Native PAGE
| Reagent/Chemical | Function/Purpose | Typical Concentration |
|---|---|---|
| Coomassie Blue G-250 | Imparts negative charge, prevents protein aggregation [27] [37] | 0.02% in cathode buffer [37] |
| n-Dodecyl-β-D-maltoside | Mild nonionic detergent for membrane protein solubilization [27] [37] | 2% for sample preparation [37] |
| Digitonin | Very mild detergent for supercomplex preservation [27] | Variable (optimized per sample) |
| 6-Aminocaproic Acid | Zwitterionic salt; stabilizes complexes, supports extraction [27] | 0.75 M in buffer A [37] |
| Bis-Tris | Buffering agent for maintaining neutral pH [27] [37] | 50 mM in anode buffer [37] |
| Protease Inhibitors | Prevent protein degradation during extraction [37] | 1 mM PMSF, 1 μg/mL leupeptin/pepstatin [37] |
| Tricine | Buffer component in cathode chamber [37] | 50 mM in cathode buffer [37] |
| Gradient Gel Acrylamide | Matrix for size-based separation [37] | 6-13% linear gradient [37] |
Sample Preparation Protocol
Proper sample preparation is critical for the success of native electrophoresis experiments. The following workflow outlines the key steps for mitochondrial sample preparation, adapted from validated protocols [27] [37]:
For BN-PAGE, the sample is supplemented with Coomassie Blue G-250 dye (0.02-0.05% final concentration) prior to loading [37]. For CN-PAGE, the dye addition step is omitted, and the samples are loaded directly onto the gel without dye [27]. It is noteworthy that sample preparation can be shortened compared to earlier protocols without compromising complex integrity, making the technique more accessible for small patient samples such as tissue biopsies or limited cell cultures [27].
Gel Casting and Electrophoresis Conditions
While pre-cast native gels are commercially available, manual casting provides greater flexibility and cost-effectiveness [27]. A linear acrylamide gradient gel (typically 6-13%) is recommended for optimal resolution of protein complexes across a broad molecular weight range [37]. The electrophoresis is typically performed at constant voltage (150 V) for approximately 2 hours or until the dye front approaches the bottom of the gel [37]. Maintaining the temperature at 4°C throughout the run helps preserve complex integrity, particularly for labile complexes [6].
Downstream Applications and Detection Methods
Following electrophoresis, several downstream applications can be employed to analyze the separated protein complexes:
In-gel Enzyme Activity Staining: Specific histochemical staining methods can detect intact, catalytically active enzymes directly in the native gel [27]. This has been successfully applied for Complexes I, II, IV, and V of the mitochondrial respiratory chain, with recent protocol improvements enhancing the sensitivity of Complex V activity staining [27]. A limitation is the comparative insensitivity of in-gel Complex IV activity staining and the lack of in-gel Complex III activity staining [27].
Two-dimensional BN/SDS-PAGE: This powerful combination involves excising a lane from the native gel, incubating it in SDS-containing denaturing buffer, and loading it onto a standard SDS-PAGE gel for separation in the second dimension [27] [37]. This approach reveals the subunit composition of the native complexes resolved in the first dimension [27] [40].
Western Blot Analysis: Specific antibodies can be used to detect individual protein components after transfer to PVDF membranes [37]. It is crucial to use antibodies that recognize the native conformation of the target proteins, as denatured epitopes may not be accessible [39].
Mass Spectrometry Identification: BN-PAGE separated complexes can be excised and subjected to mass spectrometry for comprehensive identification of protein components, providing a powerful tool for functional proteomics [40].
Applications in Research and Drug Development
The preservation of native protein structure and function makes BN-PAGE and CN-PAGE particularly valuable for both basic research and pharmaceutical applications. In basic research, these techniques have been instrumental in characterizing the subunit composition and assembly pathways of mitochondrial respiratory complexes [27] [37], identifying protein-protein interactions within supercomplexes [27] [39], and studying the impact of genetic mutations on complex assembly in mitochondrial disorders [27]. The ability to resolve complexomes - the entire set of protein complexes in a biological system - has opened new avenues for systematic studies of cellular machinery [38].
In drug development, native electrophoresis provides a means to study drug-target interactions in the context of intact protein complexes, screen for compounds that modulate complex assembly or stability, and investigate mechanisms of drug-induced toxicity related to mitochondrial dysfunction [27]. The technique's capacity to detect assembly intermediates makes it particularly useful for diagnosing and studying rare metabolic diseases [27], with potential applications in developing targeted therapies for these conditions.
Limitations and Troubleshooting Considerations
Despite their powerful capabilities, BN-PAGE and CN-PAGE have specific limitations that researchers must consider. The comparative insensitivity of some in-gel activity assays, particularly for Complex IV, and the complete lack of in-gel activity staining for Complex III represent methodological constraints [27]. The Coomassie dye used in BN-PAGE is not completely inert and may potentially disrupt some weak protein-protein interactions [39]. The presence of salts or other solutes in the sample can cause protein smearing, necessitating careful sample preparation and desalting steps [39].
Troubleshooting common issues often involves optimizing the detergent-to-protein ratio for solubilization, adjusting the acrylamide gradient to better resolve complexes of interest, and ensuring that protease inhibitors are fresh and effective to prevent degradation [37]. When resolution problems persist in BN-PAGE, switching to CN-PAGE may improve results, particularly for complexes sensitive to the Coomassie dye [27] [39]. For the analysis of metal-binding proteins, CN-PAGE or the related NSDS-PAGE (which retains 98% of bound Zn²⁺ compared to 26% in SDS-PAGE) may be preferable [7].
BN-PAGE and CN-PAGE represent sophisticated methodological advancements in the field of protein separation technology, offering unique capabilities for the analysis of native protein complexes that are simply unattainable through denaturing methods like SDS-PAGE. By preserving the structural and functional integrity of multi-protein assemblies, these techniques provide crucial insights into the molecular organization of cellular machinery, particularly in energy-transducing systems like mitochondria. The continuing refinement of these protocols, including shortened extraction procedures and enhanced activity staining methods [27], ensures their ongoing relevance in both basic research and drug development contexts. As our understanding of cellular processes increasingly emphasizes the importance of protein interactions within complex networks, the role of native electrophoresis techniques in providing a window into these functional assemblies remains indispensable.
In protein research, the selection of an appropriate separation technique is a critical determinant of success in downstream functional analyses. Polyacrylamide Gel Electrophoresis (PAGE) serves as a fundamental tool for protein separation, with Sodium Dodecyl Sulfate-PAGE (SDS-PAGE) and Native PAGE representing two principal methodologies with divergent applications and outcomes [5]. The core distinction lies in their treatment of protein structure: SDS-PAGE denatures proteins into uniform linear chains, facilitating separation by molecular mass, while Native PAGE preserves proteins in their native, folded conformation, maintaining higher-order structures and biological activity [6] [41]. This technical guide details the downstream processing strategies for recovering functional proteins, contextualized within the comparative framework of SDS-PAGE versus Native PAGE separation research. It provides detailed methodologies for protein separation, recovery, and subsequent functional validation, addressing the critical need for protocols that bridge separation science with functional proteomics.
Core Principles: SDS-PAGE vs. Native PAGE
The strategic choice between SDS-PAGE and Native PAGE is guided by the ultimate objective of the research—whether determining molecular weight and purity or investigating protein function, interactions, and native architecture [5]. Table 1 summarizes the fundamental differences between these two techniques.
Table 1: Fundamental Differences Between SDS-PAGE and Native PAGE
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight/mass [6] [17] | Protein size, overall charge, and shape [6] [41] |
| Protein State | Denatured and unfolded [5] | Native, folded conformation [5] |
| Detergent (SDS) | Present (denaturing agent) [6] | Absent [6] |
| Sample Preparation | Heating with SDS and reducing agents [6] | No heating, no denaturing agents [6] |
| Buffer Composition | Contains reducing agents (e.g., DTT, BME) [6] | No reducing or denaturing agents [6] |
| Net Protein Charge | Consistently negative (masked by SDS) [17] | Positive or negative (intrinsic charge) [6] |
| Functional Recovery | Not possible; proteins are inactivated [5] [6] | Proteins retain function and can be recovered [5] [6] |
| Primary Applications | Molecular weight determination, purity checks, subunit analysis [5] [11] | Study of protein complexes, oligomerization, enzymatic activity [5] [6] |
SDS-PAGE operates by coating proteins with the anionic detergent SDS, which confers a uniform negative charge and denatures the protein into a linear polypeptide chain [5] [17]. This process ensures that separation occurs almost exclusively based on polypeptide chain length, allowing for accurate molecular weight estimation [11]. Conversely, Native PAGE employs non-denaturing conditions, enabling proteins to migrate based on a combination of their intrinsic charge, size, and three-dimensional structure [6] [41]. This preservation of native state is the cornerstone for recovering functionally active proteins post-separation.
A hybrid approach, termed Native SDS-PAGE (NSDS-PAGE), has been developed to bridge the resolution of SDS-PAGE with the functional preservation of Native PAGE. This method utilizes minimal SDS concentrations and omits heating and chelating agents like EDTA, enabling the retention of enzymatic activity and bound metal ions in many proteins while maintaining high-resolution separation [7].
Experimental Protocols for Protein Separation and Recovery
Standard SDS-PAGE Protocol
This protocol is adapted from established methodologies [7] [11] and is suitable for determining protein molecular weight and sample purity.
- Sample Preparation: Mix the protein sample with an SDS-containing loading buffer (e.g., LDS sample buffer) including a reducing agent such as DTT or β-mercaptoethanol. Heat the mixture at 70-100°C for 5-10 minutes to ensure complete denaturation [6] [7].
- Gel Selection: Load samples into a polyacrylamide gel with an appropriate acrylamide concentration (e.g., 12% Bis-Tris gel). A stacking gel (typically 4-5% acrylamide) is often used to concentrate proteins before they enter the resolving gel [42] [41].
- Electrophoresis: Fill the tank with an SDS-running buffer (e.g., MOPS-SDS buffer). Apply a constant voltage (e.g., 200V) until the dye front migrates to the bottom of the gel. The process is typically performed at room temperature [6] [7].
- Visualization: Following electrophoresis, proteins are visualized using stains like Coomassie Brilliant Blue or more sensitive silver stain [41]. Note: Proteins separated by this method are denatured and generally unsuitable for functional recovery.
Native PAGE Protocol for Functional Recovery
This protocol is designed to preserve protein activity for downstream functional assays [6] [7].
- Sample Preparation: Mix the protein sample with a non-denaturing loading buffer that lacks SDS, reducing agents, and chelators like EDTA. The buffer typically contains Tris-HCl, glycerol (for density), and a tracking dye. Crucially, the sample must not be heated [6] [7].
- Gel Casting and Pre-run: Prepare a non-denaturing polyacrylamide gel without SDS. To pre-run the gel and establish equilibrium, run it for a short period (e.g., 30 minutes) in the chosen running buffer before loading samples. This step removes persulfate and other gel polymerization byproducts that could denature proteins [7].
- Electrophoresis Conditions: Load the prepared samples and run the gel in a native running buffer (e.g., Tris-Glycine or Bis-Tris, pH-specific for the proteins of interest). To prevent heat-induced denaturation, run the gel at 4°C [6].
- Protein Elution and Recovery:
- Electroelution: Following separation, the protein band of interest can be identified through mild staining or by comparing a small sliced guide strip. Excise the gel slice containing the target protein and place it in an electroelution device. An electric field drives the native protein out of the gel matrix into a small volume of suitable buffer, from which it can be recovered.
- Diffusion Elution: Crush the gel slice and incubate it in an appropriate assay or storage buffer. Gentle agitation facilitates the passive diffusion of the protein out of the gel.
- The recovered protein can now be used in downstream functional assays, such as activity measurements or interaction studies [5].
Workflow for Method Selection
The following diagram illustrates the decision-making process for selecting the appropriate electrophoresis technique based on research goals.
The Scientist's Toolkit: Essential Reagents and Materials
Successful protein separation and recovery depend on specific reagents and equipment. Table 2 catalogs key research reagent solutions and their functions in downstream processing.
Table 2: Key Research Reagent Solutions for Protein PAGE and Recovery
| Reagent/Material | Function/Purpose | Technical Notes |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and imparts uniform negative charge; core of SDS-PAGE [5] [11]. | Omit for Native PAGE. Concentration is critical in NSDS-PAGE [7]. |
| Acrylamide/Bis-acrylamide | Monomer and crosslinker forming the porous gel matrix for size-based separation [42]. | Pore size is determined by total acrylamide concentration (%T) and crosslinker ratio (%C) [42]. |
| Reducing Agents (DTT, BME) | Break disulfide bonds in proteins, ensuring complete unfolding in reducing SDS-PAGE [6] [11]. | Essential for subunit analysis. Omit for Native PAGE to preserve quaternary structure. |
| Coomassie Brilliant Blue | Anionic dye for staining proteins post-electrophoresis; allows visualization of protein bands [41]. | Can be compatible with downstream electroelution for Native PAGE if used in mild, non-fixing conditions. |
| Tris-based Buffers | Provide the necessary ionic strength and pH control in gels and running buffers [41]. | pH is critical for Native PAGE as it affects protein charge and migration direction [6]. |
| Protease Inhibitors | Prevent proteolytic degradation of the target protein during sample preparation and processing. | Critical for maintaining protein integrity, especially during slow Native PAGE runs and elution steps. |
| Electroelution Apparatus | Equipment used to apply an electric field to extract proteins from excised gel pieces post-electrophoresis. | Preferred method for efficient recovery of native proteins from gels with high yield and concentration. |
Downstream Functional Validation of Recovered Proteins
The successful recovery of proteins via Native PAGE is ultimately validated through functional assays. The applicability of these assays depends entirely on the nature of the recovered protein.
- Enzymatic Assays: For enzymes, functionality is confirmed by measuring catalytic activity. The recovered protein is incubated with its specific substrate under optimal pH and temperature conditions, and the formation of the product or consumption of the substrate is measured over time [5] [7]. A successful outcome demonstrates that the enzyme's active site remained intact throughout the separation and recovery process.
- Protein-Protein Interaction Studies: Recovered proteins can be used in techniques like co-immunoprecipitation, surface plasmon resonance (SPR), or isothermal titration calorimetry (ITC) to study binding kinetics and affinity with partner proteins [5]. This confirms the preservation of conformational epitopes and interaction domains.
- Cell-Based Assays: In drug development, functionally recovered proteins (e.g., cytokines, growth factors) can be applied to cell cultures to study downstream signaling pathways, proliferation, or other phenotypic responses, verifying their bio-relevance [7].
The convergence of sophisticated separation science and robust downstream processing is vital for advancing functional proteomics and drug development. This guide has delineated the critical pathway from selecting the appropriate electrophoretic method (SDS-PAGE vs. Native PAGE) to the successful recovery and validation of functional proteins. While SDS-PAGE remains an unparalleled tool for analytical characterization, Native PAGE and its advanced variants, such as NSDS-PAGE, are indispensable for researchers aiming to probe the dynamic, functional landscape of proteins in their native state. The implementation of the detailed protocols and best practices outlined herein—from careful sample preparation and temperature control to gentle elution techniques—enables scientists to effectively bridge the gap between protein separation and functional analysis, thereby unlocking deeper insights into protein mechanics and accelerating therapeutic discovery.
Solving Common Problems and Optimizing Results
In the realm of protein separation research, the choice between SDS-PAGE and Native PAGE is fundamental, dictated by the scientific question at hand. SDS-PAGE, a denaturing technique, separates proteins based primarily on molecular weight by masking intrinsic charges and disrupting higher-order structures [5] [43]. In contrast, Native PAGE separates proteins in their native, folded state, preserving their function, complex formation, and intrinsic charge, with migration dependent on a combination of size, charge, and shape [6] [12]. While Native PAGE is indispensable for studying functional protein complexes and enzymatic activity, SDS-PAGE remains the workhorse for determining molecular weight, assessing purity, and analyzing subunit composition due to its high resolution and simplicity [5] [43]. However, this powerful technique is prone to specific artifacts like smiling bands, smearing, and poor resolution. This guide provides an in-depth troubleshooting framework for these issues, contextualized within the core principles of protein separation.
Core Principles: SDS-PAGE vs. Native PAGE
Understanding the fundamental differences between these two techniques is crucial for effective troubleshooting, as many SDS-PAGE artifacts stem from deviations in its denaturing principles.
Comparative Analysis of Separation Techniques
The table below summarizes the key operational and outcome differences between SDS-PAGE and Native PAGE.
| Criterion | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight (mass) of polypeptide chains [6] [12] | Native size, overall charge, and 3D shape of the protein [6] [12] |
| Gel Condition | Denaturing [6] | Non-denaturing [6] |
| SDS Presence | Present (denaturant) [6] | Absent [6] |
| Sample Preparation | Heated with SDS and reducing agents [6] [4] | Not heated; no denaturing agents [6] |
| Protein State | Denatured and linearized [5] [43] | Native, folded conformation [5] [43] |
| Protein Function Post-Separation | Lost [5] [43] | Retained [5] [43] |
| Protein Recovery | Not recoverable in functional form [6] | Recoverable in active form [6] |
| Primary Applications | Molecular weight determination, purity check, subunit analysis [43] [6] | Studying oligomeric state, protein-protein interactions, and enzymatic activity [5] [10] |
The Scientist's Toolkit: Essential Reagents for SDS-PAGE
A successful experiment relies on high-quality reagents. Key components and their functions are listed below.
| Reagent | Function |
|---|---|
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers a uniform negative charge [4] [12]. |
| Acrylamide/Bis-acrylamide | Forms the cross-linked polymer matrix that acts as a molecular sieve [12]. |
| APS (Ammonium Persulfate) & TEMED | Catalyze the free-radical polymerization of the acrylamide gel [12]. |
| Tris-based Buffers | Provide the conductive ionic medium and maintain stable pH during electrophoresis [4]. |
| Reducing Agents (e.g., DTT, β-ME) | Cleave disulfide bonds to ensure complete protein denaturation and subunit separation [4]. |
| Tracking Dye (e.g., Bromophenol Blue) | Visualizes the migration progress during the run [4]. |
| Glycerol | Increases sample density for easy loading into wells [19]. |
Troubleshooting Common SDS-PAGE Artifacts
The following section details the most common SDS-PAGE issues, their causes, and proven solutions. The provided workflow offers a systematic approach to diagnosing problems.
Logical Troubleshooting Workflow
The diagram below outlines a step-by-step diagnostic process for the issues discussed in this guide.
Quantitative Troubleshooting Guide
For efficient problem-solving, the table below consolidates specific issues, their root causes, and corrective actions.
| Problem & Visual Indicator | Primary Cause | Recommended Solution |
|---|---|---|
| Smiling Bands (Curved Bands) [44] [45] | Excessive heat generation during electrophoresis, causing uneven gel expansion [44]. | Run the gel at a lower voltage for a longer duration. Perform electrophoresis in a cold room or use a cooling apparatus to dissipate heat [44]. |
| Smeared Bands [44] [45] | 1. Voltage too high, causing overheating and distorted migration [44].2. Protein overload, exceeding the gel's capacity [45].3. High salt concentration in the sample [45]. | 1. Decrease voltage by 25-50% [45].2. Load less protein onto the gel [45].3. Desalt the sample via dialysis, TCA precipitation, or a desalting column [45]. |
| Poor Band Resolution (Blurry Bands) [44] [45] | 1. Insufficient run time [44].2. Improper running buffer (wrong ion concentration/pH) [44].3. Gel concentration inappropriate for target protein size [45]. | 1. Run the gel until the dye front nears the bottom; longer for high MW proteins [44].2. Remake running buffer to exact specifications [44].3. Optimize acrylamide percentage or use a gradient gel [45]. |
| Artifact Bands (e.g., Doublets, Streaking) [45] | 1. Incomplete reduction of disulfide bonds, leading to re-oxidation [45].2. Protein aggregation or precipitation [45].3. Edge effect from empty peripheral wells [44]. | 1. Use fresh reducing agents (DTT, β-mercaptoethanol) and ensure proper heating [45].2. Centrifuge samples before loading; add urea for hydrophobic proteins [45].3. Do not leave wells empty; load ladder or dummy samples in peripheral wells [44]. |
| Weak or Missing Bands [45] | 1. Protein ran off the gel (over-run) [44].2. Protein degradation by proteases [45].3. Protein quantity below detection limit [45]. | 1. Stop run promptly; use a higher % acrylamide gel for low MW proteins [45].2. Use protease inhibitors and avoid repeated freeze-thaw cycles [45].3. Concentrate the sample or use a more sensitive stain (e.g., silver stain) [45]. |
Advanced Experimental Considerations
Sample Preparation Protocol
Robust sample preparation is the most critical step for preventing artifacts.
- Procedure: Mix protein sample with 4X SDS-PAGE sample buffer (containing Tris, glycerol, SDS, bromophenol blue, and a reducing agent like DTT) to a 1X final concentration [4] [12]. Heat the mixture at 70°C for 10 minutes or 95°C for 5 minutes to ensure complete denaturation [4]. Centrifuge at >10,000 x g for 1 minute to pellet any insoluble debris before loading [45].
- Rationale: Heating disrupts hydrogen bonds and unfolds proteins, while SDS binds linearly at a constant ratio (1.4g SDS/g protein), masking intrinsic charge [4] [12]. DTT cleaves disulfide bonds, ensuring subunits dissociate. This creates uniformly charged, linear molecules that migrate strictly by size.
Gel Electrophoresis Protocol
- Standard Conditions: For a discontinuous Tris-glycine system, use a stacking gel (pH ~6.8) and a resolving gel (pH ~8.8) [4]. Apply a constant voltage of 150-200V for a mini-gel until the dye front reaches the bottom (typically 45-60 minutes) [44] [4].
- Troubleshooting Modifications: If smiling occurs, reduce voltage to 100-125V and extend run time [44]. For poor resolution of high molecular weight proteins, prolong the electrophoresis time even after the dye front has migrated off the gel [44].
Mastering SDS-PAGE troubleshooting requires a solid understanding of its denaturing principles, which stand in stark contrast to the structure-preserving nature of Native PAGE. While SDS-PAGE provides unparalleled resolution for molecular weight analysis, its susceptibility to artifacts like smiling, smearing, and poor resolution often stems from deviations in protocol—excessive heat, improper sample prep, or suboptimal running conditions. By applying the systematic diagnostic and corrective strategies outlined in this guide, researchers can ensure the generation of high-quality, reproducible data. This reliability is fundamental not only for basic protein characterization but also for downstream applications in drug development, such as validating biopharmaceutical purity and consistency.
In the landscape of protein separation research, polyacrylamide gel electrophoresis (PAGE) serves as a foundational technique, with Sodium Dodecyl Sulfate PAGE (SDS-PAGE) and Native PAGE representing two philosophically distinct approaches. SDS-PAGE, a denaturing method, unravels proteins to their primary structure, separating polypeptides based almost exclusively on molecular weight [6] [12]. In contrast, Native PAGE is a sophisticated non-denaturing technique designed to preserve the intricate architecture and biological function of proteins throughout the separation process [5] [19]. This preservation is paramount for researchers and drug development professionals who require more than just molecular weight information; they need insights into protein-protein interactions, oligomeric states, and enzymatic activity—features all obliterated by SDS-PAGE. This guide details the systematic optimization of Native PAGE to maintain protein stability and activity, providing a critical methodology for functional proteomics within the broader context of protein separation research.
Core Principles: Native PAGE vs. SDS-PAGE
The fundamental divergence between Native PAGE and SDS-PAGE lies in their treatment of protein structure. SDS-PAGE employs the anionic detergent SDS and often reducing agents like DTT or β-mercaptoethanol to fully denature proteins, mask their intrinsic charge, and ensure separation is determined solely by molecular mass [6] [26] [17]. This process irrevocably destroys the native conformation, quaternary structure, and thus, the biological activity of the protein [12].
Native PAGE, however, omits all denaturing agents. Separation occurs based on the protein's intrinsic charge, size, and three-dimensional shape as it migrates through the gel matrix in its native, folded state [5] [12]. This allows multimeric proteins to retain their subunit interactions, enables the recovery of active proteins post-electrophoresis, and facilitates the study of functional complexes [19] [12]. The following table summarizes the critical distinctions between these two techniques.
Table 1: Fundamental Differences Between SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight only [6] [17] | Size, intrinsic charge, and 3D shape [6] [5] |
| Gel Nature | Denaturing [6] | Non-denaturing [6] |
| Protein State | Denatured, linearized polypeptides [12] | Native, folded conformation [6] |
| Quaternary Structure | Disrupted; multimers dissociate [26] | Preserved; multimers remain intact [12] |
| Biological Activity | Lost [6] [12] | Retained [7] [5] |
| Protein Recovery | Typically not functional [6] | Functional proteins can be recovered [6] [19] |
| Key Applications | Molecular weight determination, purity checks [6] [12] | Studying oligomeric state, protein-protein interactions, in-gel activity assays [6] [46] |
Key Optimization Parameters for Native PAGE
Optimizing Native PAGE requires careful attention to several experimental parameters to ensure proteins remain stable and active. The following workflow outlines the critical decision points and optimization steps.
Buffer Composition and pH
The buffer system is the most critical factor in Native PAGE. Unlike SDS-PAGE, which uses buffers containing SDS, Native PAGE relies on mild, non-denaturing buffers.
- Running Buffer: Standard Native PAGE often uses Tris-glycine or Tris-borate buffers at a basic pH (typically 8.0-9.0) to ensure most proteins carry a net negative charge and migrate toward the anode [26]. For more specialized applications, Blue Native (BN)-PAGE uses Coomassie G-250 dye in the cathode buffer, which confers a negative charge to proteins, while Clear Native (CN)-PAGE relies solely on the protein's intrinsic charge [6] [46].
- pH Considerations: The buffer pH must be carefully selected to maintain protein solubility and prevent aggregation or denaturation. Avoid pH extremes that could irreversibly damage the protein's native structure [12].
Temperature Control
To minimize denaturation and proteolysis during the run, Native PAGE is typically performed at 4°C [6]. Keeping the apparatus cool helps maintain the integrity of the protein's three-dimensional structure and its associated biological activity.
Gel Composition and Pore Size
The polyacrylamide gel matrix acts as a molecular sieve.
- Gradient Gels: Gradient gels (e.g., 4-16%) are highly effective for resolving a broad range of protein sizes and complexes in their native state, as they allow for fine separation across different molecular weights [26] [46].
- Acrylamide Percentage: The optimal acrylamide concentration depends on the size of the target protein or complex. Lower percentages (e.g., 4-8%) are better for large complexes, while higher percentages (e.g., 10-15%) provide better resolution for smaller proteins [12].
Sample Preparation
Native PAGE sample preparation must avoid any denaturing conditions.
- No Denaturants: The sample buffer must not contain SDS, urea, or other denaturing agents [6].
- No Reducing Agents: Avoid DTT or β-mercaptoethanol, as they would disrupt native disulfide bonds that may be essential for structure [6] [26].
- No Heating: The protein sample should never be heated prior to loading [6].
- Preserving Cofactors: For metalloenzymes, the use of metal chelators like EDTA in buffers must be avoided to prevent stripping essential metal ions, which is crucial for retaining activity [7].
Advanced Experimental Protocol: High-Resolution Native PAGE
The following detailed protocol, adapted from contemporary research, is designed for separating native proteins with high resolution and retained enzymatic function, as demonstrated in the study of medium-chain acyl-CoA dehydrogenase (MCAD) [46].
Materials and Reagent Solutions
Table 2: Essential Research Reagents for High-Resolution Native PAGE
| Reagent / Material | Function / Explanation |
|---|---|
| High-Resolution Clear Native (hrCN) Gel (e.g., 4-16% gradient) | Provides a non-denaturing matrix with a pore-size gradient to separate native protein complexes based on size, charge, and shape [46]. |
| Tris-Based Running Buffer (pH ~8.0) | Provides the ionic environment and pH for electrophoresis. The alkaline pH ensures most proteins are negatively charged [26]. |
| Non-Denaturing Sample Buffer (e.g., containing glycerol) | Glycerol increases sample density for easy well loading without requiring denaturants [7] [26]. |
| Protease Inhibitors (e.g., PMSF) | Prevents protein degradation by endogenous proteases during sample preparation and electrophoresis [7] [26]. |
| Coomassie Blue Staining Solution | For general protein detection after electrophoresis without denaturing proteins [26]. |
| Activity Assay Reagents (e.g., substrate, electron acceptors like NBT) | For in-gel activity staining to directly visualize and quantify the function of resolved enzymes [46]. |
Step-by-Step Methodology
Gel Preparation & Equilibration: Use a pre-cast high-resolution 4-16% Bis-Tris gradient gel or prepare one manually. Pre-run the gel with running buffer for a short period (e.g., 30 minutes) in double-distilled water to remove any unpolymerized acrylamide and storage buffers [7].
Native Sample Preparation:
- Prepare the protein sample in a non-denaturing lysis buffer (e.g., 20 mM Tris-Cl, pH 7.4) [7].
- Add protease inhibitors (e.g., 500 μM PMSF) to prevent degradation [7].
- Mix the protein sample with a 4X native sample buffer. A recommended formulation is: 100 mM Tris HCl, 150 mM Tris base, 10% (v/v) glycerol, 0.01875% (w/v) Coomassie G-250, 0.00625% (w/v) Phenol Red, pH 8.5 [7].
- Crucially, do not heat the sample.
Electrophoresis:
- Load the prepared samples into the wells.
- Conduct electrophoresis in a running buffer such as: 50 mM MOPS, 50 mM Tris Base, 0.0375% SDS, pH 7.7 (Note: trace SDS can be present in some "native" protocols to improve resolution without full denaturation [7]) or a standard Tris-glycine buffer.
- Run the gel at a constant voltage (e.g., 150-200V) at 4°C until the dye front migrates to the bottom of the gel [6] [7].
Post-Electrophoresis Analysis:
- Protein Visualization: Stain the gel with Coomassie Brilliant Blue or a compatible fluorescent stain to visualize the total protein profile [26].
- In-Gel Activity Assay: To detect functional enzymes, incubate the gel in an activity stain solution. For example, for an oxidoreductase like MCAD, the gel is incubated with its physiological substrate (e.g., octanoyl-CoA) and an electron acceptor like nitro blue tetrazolium (NBT), which produces an insoluble purple formazan precipitate at the site of activity [46].
Applications and Data Interpretation
Properly optimized Native PAGE provides unique insights that are inaccessible via SDS-PAGE. The following diagram illustrates a classic experiment that reveals a protein's quaternary structure.
A practical example of this interpretation is a protein that migrates at 120 kDa on Native PAGE but migrates at 60 kDa on non-reducing SDS-PAGE. This result strongly infers that the native protein is a dimer of 60 kDa subunits that are not linked by disulfide bonds [10]. The non-covalent interactions holding the dimer together are disrupted by SDS in the absence of a reducing agent.
Quantitative Functional Analysis
Advanced Native PAGE applications allow for precise quantitative analysis. Research on MCAD demonstrates that in-gel activity assays can show a linear correlation between the amount of protein loaded, its FAD cofactor content, and the resulting enzymatic activity, enabling the functional quantification of specific protein oligomers, such as active tetramers versus inactive aggregates [46].
Table 3: Troubleshooting Common Issues in Native PAGE
| Issue | Potential Cause | Optimization Strategy |
|---|---|---|
| Poor Resolution / Smearing | Protein aggregation, proteolysis, incorrect buffer pH. | Use fresh protease inhibitors; optimize buffer pH and ionic strength; keep samples and apparatus at 4°C [12]. |
| Low or No In-Gel Activity | Protein denatured during processing; loss of essential cofactor. | Avoid any heating; include essential cofactors (e.g., Zn²⁺, FAD) in buffers; ensure running buffer is free of metal chelators like EDTA [7] [46]. |
| Abnormal Migration | Protein not fully charged or incorrect gel pore size. | For BN-PAGE, ensure Coomassie dye is present to confer charge; switch to a gradient gel for better separation of a wide size range [46]. |
| Protein Aggregation at Well | Sample too concentrated or contains aggregates. | Centrifuge sample before loading; use a gel with a lower percentage acrylamide stacking layer [12]. |
Within the dualistic framework of protein separation methodologies, Native PAGE stands as an indispensable technique for functional analysis. Its optimization—through meticulous control of buffer conditions, temperature, and sample integrity—allows researchers to move beyond simple molecular weight determination and into the realm of native structure, complex interaction, and biological activity. For drug discovery professionals targeting protein-protein interactions or studying the functional impact of pathogenic variants, mastering Native PAGE is not merely an academic exercise but a critical step toward generating biologically relevant data. As the drive to understand and drug the proteome in its native state intensifies, the refined application of Native PAGE will continue to be a cornerstone of rigorous biochemical research.
Gel Percentage Selection for Different Protein Size Ranges
Protein gel electrophoresis is a foundational technique in biological research and biopharmaceutical development for separating complex protein mixtures. The selection between SDS-PAGE (denaturing) and native PAGE (non-denaturing) separation principles represents a critical branching point in experimental design, each with distinct advantages and limitations for protein characterization [47] [26]. This technical guide provides an in-depth framework for researchers on selecting appropriate gel percentages based on target protein size ranges, with particular emphasis on practical applications in drug development workflows.
The core distinction between these techniques lies in their treatment of protein structure. SDS-PAGE employs the anionic detergent sodium dodecyl sulfate to denature proteins and impart a uniform negative charge, effectively separating polypeptides based primarily on molecular mass [47] [4]. In contrast, native PAGE preserves protein higher-order structure, enzymatic activity, and protein complexes, separating molecules based on both charge and hydrodynamic size [47] [26] [7]. This fundamental difference dictates not only the type of information obtained but also the strategic approach to gel matrix selection.
Fundamental Principles of Gel-Based Separation
Polyacrylamide Gel Matrix Formation
Polyacrylamide gels form through a copolymerization reaction between acrylamide monomers and bisacrylamide cross-linkers, typically catalyzed by ammonium persulfate (APS) and TEMED [47] [4]. The resulting three-dimensional network creates pores that serve as a molecular sieve during electrophoresis. The pore size is inversely related to the total acrylamide concentration, with higher percentages creating smaller pores that better resolve lower molecular weight proteins [47] [26].
The gel percentage refers to the total concentration of acrylamide and bisacrylamide in the solution. For example, a 10% gel contains 10g of acrylamide-bisacrylamide mixture per 100mL of solution [47]. The ratio of bisacrylamide to acrylamide typically ranges from 1:20 to 1:40, affecting the rigidity and pore size distribution of the resulting matrix [47]. This matrix density directly impedes protein migration during electrophoresis, with smaller proteins traversing the gel more rapidly than larger counterparts [26].
Discontinuous Gel Systems
Most high-resolution electrophoretic separations employ a discontinuous system consisting of two distinct gel regions: a stacking gel and a resolving gel [4] [26]. The stacking gel typically has a lower acrylamide concentration (4-5%), larger pore size, and different pH (approximately 6.8) that concentrates protein samples into tight bands before they enter the resolving gel [47]. The resolving gel contains higher acrylamide concentration (ranging from 8-20% depending on target protein sizes) with a basic pH (8.8) where actual size-based separation occurs [4]. This dual-layer system significantly enhances band sharpness and resolution compared to continuous buffer systems.
Table 1: Standard Gel Percentage Recommendations for Protein Separation
| Gel Percentage | Optimal Protein Separation Range | Primary Applications |
|---|---|---|
| 8% | 70 - 200 kDa | Large proteins and protein complexes |
| 10% | 40 - 100 kDa | Standard range for most routine analyses |
| 12% | 20 - 80 kDa | Intermediate molecular weight proteins |
| 15% | 10 - 50 kDa | Small proteins and polypeptides [26] |
SDS-PAGE: Denaturing Separation by Molecular Mass
Principles and Mechanisms
In SDS-PAGE, proteins are denatured and linearized by heating in the presence of SDS and reducing agents (β-mercaptoethanol or DTT) that cleave disulfide bonds [4]. SDS binds to polypeptides at a constant weight ratio (approximately 1.4g SDS per 1g protein), masking intrinsic protein charges and creating a uniform negative charge-to-mass ratio [47] [4]. Under these conditions, protein migration through the polyacrylamide matrix depends primarily on molecular size, with smaller polypeptides migrating faster than larger ones [47].
The electrophoresis running buffer typically contains Tris-glycine with 0.1% SDS at pH 8.3-8.6, maintaining the denatured state and negative charge on proteins throughout separation [4]. The inclusion of SDS in both sample and running buffers ensures continuous denaturation during electrophoresis. The bromophenol blue dye front allows visual monitoring of separation progress, with electrophoresis typically completed before the dye front exits the gel [4].
Gel Percentage Selection for SDS-PAGE
Selection of appropriate acrylamide concentration is critical for achieving optimal resolution in SDS-PAGE. As shown in Table 1, different gel percentages provide optimal separation for specific molecular weight ranges [26]. For proteins spanning a broad molecular weight range, gradient gels (e.g., 4-20% or 10-20% acrylamide) offer enhanced resolution across multiple size classes by creating a pore size gradient from top to bottom [47] [4]. This gradient allows proteins to migrate until they reach a pore size that restricts further movement, effectively sharpening bands and improving resolution across a wider mass range than single-percentage gels.
Table 2: Detailed Gel Percentage Guidelines for SDS-PAGE
| Target Protein Size | Recommended Gel % | Separation Characteristics | Special Considerations |
|---|---|---|---|
| Very large proteins(100-500 kDa) | 4-8% | Larger pore size facilitates entry and migration of big macromolecules | May require extended run times; lower resolution for small proteins |
| Standard separation(30-150 kDa) | 10-12% | Optimal balance between resolution and separation range | Most commonly used for general protein analysis |
| Small proteins/peptides(5-50 kDa) | 15-20% | Small pore size provides better resolution of low MW species | May prevent migration of very large proteins [47] [26] |
SDS-PAGE Experimental Protocol
Sample Preparation:
- Dilute protein samples in SDS sample buffer (62.5 mM Tris-HCl pH 6.8, 2% SDS, 10% glycerol, 0.01% bromophenol blue) [4]
- Add reducing agent (50 mM DTT or 5% β-mercaptoethanol) to break disulfide bonds [4]
- Heat samples at 95°C for 5 minutes or 70°C for 10 minutes to ensure complete denaturation [4]
- Centrifuge briefly to collect condensed sample before loading
Gel Preparation and Electrophoresis:
- Prepare resolving gel solution with desired acrylamide percentage (Table 2) in Tris-HCl pH 8.8
- Add APS and TEMED to initiate polymerization, pour between glass plates, and overlay with isopropanol to create flat interface [4]
- After polymerization, prepare stacking gel (4-5% acrylamide in Tris-HCl pH 6.8) with comb to create wells
- Assemble gel in electrophoresis chamber filled with Tris-glycine-SDS running buffer [4]
- Load samples and molecular weight markers (10-20 μg protein per lane for Coomassie staining)
- Run electrophoresis at 100-200V constant voltage until dye front approaches bottom of gel [4]
Post-Electrophoresis Analysis:
- Stain with Coomassie Brilliant Blue or silver stain for total protein visualization [4]
- For specific detection, transfer to membrane for western blotting [26]
- Image and analyze band patterns and intensities using densitometry software
Native PAGE: Separation of intact Proteins and Complexes
Principles and Methodological Considerations
Native PAGE separates proteins based on their intrinsic charge, size, and three-dimensional structure under non-denaturing conditions [47] [26]. Without SDS denaturation, proteins maintain their native conformation, enzymatic activity, and participation in multimeric complexes [47] [7]. Separation depends on both the protein's net charge at the running buffer pH and its hydrodynamic size, which is influenced by molecular mass and three-dimensional shape [47].
The electrophoresis buffer for native PAGE typically lacks SDS and may use Tris-glycine at pH 8.8 or specialized buffers like Tris-Bicine [26]. To maintain protein stability and prevent denaturation during separation, the electrophoresis apparatus is often kept cool (4°C), and proteolysis inhibitors may be included in samples [47]. Unlike SDS-PAGE, the relationship between migration distance and molecular mass is not linear in native PAGE, as each protein's unique charge and structure influence its mobility [47] [26].
Advanced Native Electrophoresis Techniques
Blue Native PAGE (BN-PAGE) represents a specialized native electrophoresis technique particularly valuable for studying multiprotein complexes, especially mitochondrial oxidative phosphorylation complexes [37]. In this method, the anionic dye Coomassie Blue G-250 binds to protein complexes, imparting negative charge without causing significant denaturation [37] [7]. This allows separation of intact protein complexes according to their mass and shape, with the dye providing both charge and color for visualization [37].
Native Agarose Gel Electrophoresis offers an alternative matrix for separating very large protein complexes or proteins that might be retained in the stacking gel of polyacrylamide systems [48]. Agarose matrices with larger pore sizes (typically 0.5-2%) are suitable for separating macromolecular assemblies that would be excluded from polyacrylamide gels [47] [48]. Recent advancements demonstrate the application of agarose-based systems for analyzing therapeutic proteins and antibodies while maintaining baseline stability and resolution [49].
Table 3: Native PAGE Variations and Applications
| Method | Matrix | Separation Basis | Optimal Protein Size | Key Applications |
|---|---|---|---|---|
| Standard Native PAGE | Polyacrylamide (4-10%) | Charge, size, shape | 10-500 kDa | Enzyme activity assays, protein interactions |
| Blue Native PAGE | Polyacrylamide gradient (4-16%) | Mass with charge shift | 50-10,000 kDa | Mitochondrial complexes, oligomeric states |
| Native Agarose Electrophoresis | Agarose (0.5-2%) | Charge, hydrodynamic size | >500 kDa | Antibodies, large complexes, therapeutic proteins [47] [49] [48] |
Hybrid and Advanced Methodologies
NSDS-PAGE: A Bridge Between Techniques
Native SDS-PAGE (NSDS-PAGE) represents an innovative hybrid approach that modifies standard SDS-PAGE conditions to preserve certain functional properties while maintaining high resolution [7]. This method eliminates or reduces SDS concentration (0.0375% vs. standard 0.1%), removes EDTA from buffers, and omits the heating step during sample preparation [7]. These modifications allow proteins to retain bound metal ions and, in many cases, enzymatic activity while still achieving excellent separation resolution.
Experimental data demonstrates that Zn²⁺ retention in proteomic samples increases from 26% in standard SDS-PAGE to 98% in NSDS-PAGE conditions [7]. Furthermore, seven of nine model enzymes, including four Zn²⁺ proteins, retained activity after NSDS-PAGE separation, whereas all were denatured during conventional SDS-PAGE [7]. This technique offers a valuable compromise when researchers need high-resolution separation while preserving certain functional aspects of proteins.
Two-Dimensional Electrophoresis
For the most comprehensive analysis of complex protein mixtures, two-dimensional (2D) PAGE combines separation by native isoelectric point in the first dimension with SDS-PAGE separation by mass in the second dimension [47]. This technique provides the highest resolution currently available for protein analysis, capable of resolving thousands of proteins on a single gel [47]. The first dimension typically uses isoelectric focusing (IEF) in immobilized pH gradient (IPG) strips to separate proteins according to their isoelectric points, followed by standard SDS-PAGE in the second dimension for mass-based separation [47].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for Protein Electrophoresis
| Reagent/Category | Specific Examples | Function in Electrophoresis |
|---|---|---|
| Gel Matrix Components | Acrylamide, bisacrylamide, agarose | Forms porous sieving matrix for size-based separation |
| Polymerization Agents | Ammonium persulfate (APS), TEMED | Initiates and catalyzes gel matrix formation |
| Denaturing Agents | SDS (sodium dodecyl sulfate) | Denatures proteins, confers uniform negative charge |
| Reducing Agents | DTT, β-mercaptoethanol, TCEP | Cleaves disulfide bonds for complete denaturation |
| Buffering Systems | Tris-glycine, Tris-tricine, Bis-Tris | Maintains pH for optimal charge and separation |
| Tracking Dyes | Bromophenol blue | Visualizes electrophoresis progress |
| Molecular Weight Standards | Prestained markers, unstained ladders | Calibrates gel for molecular weight estimation |
| Detection Reagents | Coomassie Blue, Silver stain, SYPRO Ruby | Visualizes separated protein bands |
| Crosslinking Reagents | Glutaraldehyde, DSS | Stabilizes protein complexes for native analysis [47] [4] [48] |
Application in Biopharmaceutical Development
The selection of appropriate electrophoresis conditions has particular significance in biopharmaceutical development, where characterization of therapeutic proteins requires precise assessment of size, purity, and structural integrity [49]. Recent advancements in capillary gel electrophoresis methods address longstanding challenges in traditional slab gel electrophoresis, particularly for analyzing complex biologics like monoclonal antibodies and fusion proteins [49] [50].
Innovative methodologies such as SDS capillary agarose gel electrophoresis (SDS-CAGE) have demonstrated effectiveness in eliminating baseline disturbances that frequently complicate the analysis of higher molecular weight biopharmaceuticals [49]. These approaches enable rapid purity testing and subunit integrity analysis of therapeutic proteins across a wide molecular weight range, providing robust platforms for quality control in biopharmaceutical manufacturing [49]. The development of tetrahydroxyborate-stabilized agarose gels represents particular progress in baseline hump-free therapeutic protein characterization, facilitating more accurate quantification [49].
Strategic selection of gel percentages and electrophoresis conditions remains fundamental to successful protein separation across diverse research applications. The choice between SDS-PAGE and native PAGE methodologies should be guided by experimental objectives: SDS-PAGE provides superior mass-based resolution of denatured polypeptides, while native techniques preserve structural and functional characteristics. Gradient gels offer versatile solutions for samples spanning broad molecular weight ranges, while specialized techniques like BN-PAGE and NSDS-PAGE address specific research needs for complex analysis and functional preservation.
For researchers in drug development, ongoing methodological advancements continue to enhance the resolution, reproducibility, and analytical capabilities of electrophoretic protein separation. By aligning gel selection strategies with protein characteristics and experimental goals, scientists can optimize separation efficiency and data quality in both basic research and biopharmaceutical applications.
Within the foundational research on protein separation techniques, the optimization of buffer systems represents a pivotal step in achieving high-resolution results. Both SDS-PAGE (Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis) and native PAGE (Polyacrylamide Gel Electrophoresis) serve as cornerstone methodologies in proteomic research, drug development, and biopharmaceutical characterization. While SDS-PAGE denatures proteins to separate them primarily by molecular weight, native PAGE maintains proteins in their folded state, preserving their functional characteristics and separating them based on size, charge, and shape [51] [6]. The buffer system employed in these techniques directly governs the electrophoretic conditions, including pH maintenance, ion conductivity, and ultimately, the quality of separation. The Tris-glycine buffer system has long served as the traditional workhorse in countless laboratories worldwide. However, emerging research demonstrates that specialty buffers, including Tris-acetate and Tris-tricine, offer significant advantages for specific applications, particularly when analyzing complex proteins such as monoclonal antibodies or when employing native PAGE techniques [52] [53] [54]. This technical guide provides an in-depth comparison of these buffer systems, grounded in recent experimental evidence, to inform method optimization for researchers and drug development professionals.
Fundamental Principles: SDS-PAGE vs. Native PAGE
Understanding the core principles of each electrophoretic technique is essential for contextualizing buffer system selection. The following table outlines the primary differences between these two fundamental approaches.
Table 1: Core Differences Between SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight [51] [6] | Size, overall charge, and shape [51] [6] |
| Gel Nature | Denaturing [6] | Non-denaturing [6] |
| SDS Presence | Present (denatures proteins) [51] | Absent [6] |
| Sample Preparation | Heated with reducing agents [6] | Not heated; no denaturants [6] |
| Protein State | Denatured and linearized [51] | Native, folded conformation [6] |
| Protein Function | Lost [6] | Retained [55] |
| Primary Applications | Molecular weight determination, purity checks [6] | Studying native structure, subunit composition, enzyme activity [6] [55] |
The selection between these techniques dictates the required buffer chemistry. SDS-PAGE relies on SDS to impart a uniform negative charge to all proteins, allowing separation based virtually solely on molecular size as proteins migrate through the polyacrylamide gel matrix [51]. In contrast, native PAGE separates proteins based on their intrinsic charge within the buffer's alkaline pH range, combined with the sieving effect of the gel, which is influenced by the protein's size and three-dimensional structure [51] [55]. This fundamental distinction directly influences the choice of an optimal buffer system.
Comparative Analysis of Buffer Systems
Tris-Glycine: The Traditional Workhorse
The Tris-glycine discontinuous buffer system, foundational to the Laemmli method, operates in a pH range of approximately 8.3 to 9.5 [55]. In this system, chloride ions from the gel buffer act as the highly mobile leading ion, while glycinate ions from the running buffer serve as the trailing ions. This setup creates a sharp boundary that stacks proteins into thin bands before they enter the separating gel. While this system is versatile and has been successfully used for decades, it has demonstrated limitations, particularly with high molecular weight proteins. Studies on monoclonal antibodies (mAbs) have revealed that the Tris-glycine system can produce skewed or distorted bands, band spreading, and low resolution of sub-fragments [52] [54]. The relatively high operational pH may also contribute to gel-induced protein modifications [54].
Specialty Buffers: Tris-Acetate and Tris-Tricine
Tris-Acetate is a specialty buffer designed to address the shortcomings of Tris-glycine, particularly for large proteins. It operates at a significantly lower pH (7.2-8.5) compared to Tris-glycine, which minimizes gel-induced protein modifications [54] [55]. In this system, acetate acts as the leading ion and tricine as the trailing ion. Research has demonstrated its superior performance for proteins over 150 kDa, making it especially valuable in the biopharmaceutical industry for analyzing monoclonal antibodies like IgG1 and IgG2 [54] [55]. A 2021 study by Sonboli et al. found that a modified Tris-acetate SDS-PAGE system provided sharper bands, more accurate molecular weight determination, higher resolution, and better estimation of sub-fragments for mAbs compared to the traditional Tris-glycine method, with results more closely aligned with capillary gel electrophoresis [52] [54].
Tris-Tricine buffers are another alternative, particularly effective for the separation of low to medium molecular weight proteins and peptides. A comparative study on the separation of major serum proteins using native-PAGE showed that a modified Tris-tricine system was superior to both Tris-glycine and traditional Tris-tricine methods [53]. This buffer system is also recommended as an alternative in SDS-PAGE when resolving very low molecular weight proteins/peptides, as it provides better resolution in this size range [26].
Table 2: Operational Characteristics and Recommended Uses of Different Buffer Systems
| Buffer System | Operational pH Range | Key Features | Ion Roles | Ideal Use Cases |
|---|---|---|---|---|
| Tris-Glycine | 8.3 - 9.5 [55] | Traditional Laemmli system; widely established | Leading: ChlorideTrailing: Glycinate [54] | General purpose SDS-PAGE; smaller proteins (20-500 kDa) in native PAGE [55] |
| Tris-Acetate | 7.2 - 8.5 [54] [55] | Lower pH for less modification; superior for large proteins | Leading: AcetateTrailing: Tricine [54] | Large proteins (>150 kDa), mAbs [52] [55]; can be used with LDS for sharper bands [54] |
| Tris-Tricine | ~7.4 - 8.8 (buffering range) [26] | Superior for low MW proteins and peptides; effective in native-PAGE | N/A (often used in continuous systems) | Low MW proteins/peptides [26]; separation of serum proteins in native PAGE [53] |
| NativePAGE Bis-Tris | ~7.5 [55] | Uses Coomassie G-250 to impart charge; ideal for membrane proteins | Charge modifier: Coomassie G-250 [55] | Membrane proteins, hydrophobic proteins; studying native protein complexes [55] |
Experimental Evidence and Performance Data
Case Study: Monoclonal Antibody Analysis with SDS-PAGE
A definitive 2021 comparative study published in Protein Expression and Purification directly evaluated Tris-glycine and Tris-acetate SDS-PAGE systems for the analysis of IgG1 and IgG2 monoclonal antibodies [52] [54]. The researchers employed a 6-20% gradient gel and compared the results with those obtained from capillary gel electrophoresis (CE-SDS), an orthogonal method known for its precision.
Key Experimental Findings:
- Band Sharpness and Resolution: The Tris-acetate system demonstrated markedly sharper bands and higher resolution, allowing for a clearer distinction between the main antibody bands and lower molecular weight sub-fragments (e.g., light and heavy chains) [52]. In contrast, IgG2 samples appeared as a single sharp band in Tris-acetate gels but showed smearing in Tris-glycine gels [54].
- Molecular Weight Accuracy: The apparent molecular weight of mAbs determined using the Tris-glycine system was inaccurately high. This inaccuracy was shown to be unrelated to protein glycosylation, as deglycosylation experiments with PNGase-F enzyme yielded no significant improvement in the Tris-glycine results [52] [54]. The Tris-acetate system provided a more accurate molecular weight estimation.
- Correlation with CE-SDS: The quantification and profile of protein sub-fragments obtained with the Tris-acetate SDS-PAGE method showed a closer correlation to the results from the CE-SDS method, establishing it as a more reliable and accurate technique for quality control of mAbs [54].
The study concluded that while Tris-glycine may be suitable for smaller proteins, it is not optimal for large proteins like mAbs, and recommended Tris-acetate for more accurate quality control in biotechnology and pharmaceutical settings [54].
Performance in Native PAGE Applications
The choice of buffer is equally critical in native PAGE, where the goal is to maintain the protein's native structure and function. Thermo Fisher Scientific outlines three primary gel chemistry systems for native PAGE, each with distinct advantages [55]:
- Novex Tris-Glycine: The traditional system, best suited for studying smaller molecular weight proteins (20-500 kDa) where maintaining the native net charge is important [55].
- NuPAGE Tris-Acetate: Recommended for the analysis of larger molecular weight proteins (>150 kDa) in their native state, providing better resolution for these complexes [55].
- NativePAGE Bis-Tris: This system, based on Blue Native (BN)-PAGE, uses Coomassie G-250 dye in the cathode buffer to bind proteins and confer a net negative charge without denaturation. Its near-neutral pH (~7.5) and detergent compatibility make it ideal for studying membrane protein complexes and hydrophobic proteins, and it allows for separation by molecular weight regardless of a protein's intrinsic isoelectric point (pI) [55].
A hybrid method known as native SDS-PAGE (NSDS-PAGE) has also been developed, which reduces the SDS concentration in the running buffer to 0.0375% and omits EDTA and heating steps. This protocol maintains excellent protein resolution while allowing many enzymes to retain their activity and metal cofactors, bridging the gap between fully denaturing and fully native conditions [7].
The Scientist's Toolkit: Essential Reagents and Materials
Successful experimentation requires not only the correct buffer choice but also a suite of high-quality supporting reagents. The following table catalogs the essential research solutions referenced in the studies discussed.
Table 3: Key Research Reagent Solutions for Buffer Optimization Experiments
| Reagent/Material | Function/Description | Example Application in Cited Studies |
|---|---|---|
| LDS Sample Buffer | A denaturing agent similar to SDS but often reported to provide sharper bands and better resolution [54]. | Used in Tris-acetate SDS-PAGE for improved denaturation of monoclonal antibodies [54]. |
| Coomassie G-250 | A dye used in BN-PAGE and NativePAGE Bis-Tris systems to bind proteins non-specifically, imparting a negative charge while maintaining native state [55]. | Serves as the charge-shift molecule in NativePAGE Bis-Tris gels, enabling analysis of membrane proteins and basic proteins [55]. |
| PNGase-F Enzyme | An enzyme that catalyzes the removal of N-linked glycan chains from glycoproteins. | Used in parallel experiments to investigate the effect of glycosylation on mAb migration in SDS-PAGE [52] [54]. |
| DTT or 2-Mercaptoethanol | Reducing agents that cleave disulfide bonds between cysteine residues. | Added to SDS-PAGE sample buffer to ensure complete protein denaturation into subunits [51] [54]. |
| Precast Gels (Gradient) | Polyacrylamide gels with a gradient of increasing density (e.g., 4-20%, 6-20%), enabling separation of a broader range of protein sizes. | Employed in both Tris-glycine and Tris-acetate comparative studies for mAb analysis [52] [54]. |
| Capillary Gel Electrophoresis | An automated, orthogonal analytical method with high quantitative precision for protein purity and heterogeneity. | Used as a reference method to validate the results obtained from Tris-acetate and Tris-glycine SDS-PAGE [54]. |
Strategic Selection Guide
The decision to use Tris-glycine versus a specialty buffer should be guided by the specific experimental objectives and the nature of the target protein(s). The following diagram illustrates the key decision points and considerations in this selection process.
Diagram Title: Buffer System Selection Workflow
Concluding Synthesis
In the broader context of protein separation research, the optimization of buffer systems moves beyond mere tradition to a deliberate, evidence-based strategy. The long-standing use of Tris-glycine buffers is well-earned for general-purpose applications, particularly with small to medium-sized proteins. However, as protein therapeutics and complex biological molecules evolve, the limitations of this traditional system become more apparent.
The integration of specialty buffers like Tris-acetate and Tris-tricine into the researcher's toolkit addresses these limitations directly. The experimental data is clear: for large proteins such as monoclonal antibodies, Tris-acetate SDS-PAGE provides superior resolution, accuracy, and reliability [52] [54]. For native PAGE applications, the choice is more nuanced, with Tris-glycine suitable for smaller proteins, Tris-acetate excelling with larger complexes, and NativePAGE Bis-Tris being indispensable for membrane proteins and those with basic pIs [55].
Therefore, buffer system optimization is not a one-size-fits-all endeavor but a critical parameter in experimental design. By aligning the operational characteristics of the buffer—its pH, ion composition, and compatibility with detergents or dyes—with the specific physicochemical properties of the target protein and the analytical goal, researchers and drug developers can achieve the high-quality separations necessary for advancing biopharmaceutical characterization and proteomic discovery.
Sample Preparation Pitfalls and Best Practices for Both Techniques
In protein separation research, the critical importance of sample preparation cannot be overstated. The fundamental differences between SDS-PAGE and Native PAGE separation principles necessitate distinct sample preparation strategies, where even minor deviations can compromise data integrity, experimental reproducibility, and biological relevance. SDS-PAGE employs denaturing conditions to separate proteins based primarily on molecular weight, while Native PAGE utilizes non-denaturing conditions to separate proteins based on their inherent charge, size, and three-dimensional structure [5] [6]. This methodological divergence means that preparation pitfalls specific to one technique can yield misleading results, incorrect molecular weight determinations, loss of enzymatic activity, or erroneous conclusions about protein complex formation [56] [10]. Within the broader thesis of understanding the distinctions between these separation techniques, this guide provides researchers and drug development professionals with a comprehensive framework for navigating the complexities of sample preparation to ensure optimal electrophoretic results.
Fundamental Principles: SDS-PAGE vs. Native PAGE
The core distinction between these electrophoretic techniques lies in their treatment of protein structure. SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis) utilizes the anionic detergent SDS, which denatures proteins by binding to hydrophobic regions, unfolds them into linear chains, and imparts a uniform negative charge proportional to their molecular weight [5] [57]. This process masks the proteins' intrinsic charge and eliminates the influence of shape, ensuring separation occurs almost exclusively based on polypeptide chain size [26]. Consequently, SDS-PAGE is ideal for determining molecular weight, assessing purity, and analyzing subunit composition, but it destroys native conformation and biological activity [5] [6].
In contrast, Native PAGE maintains proteins in their native, folded conformation by omitting denaturing agents like SDS and reducing agents [5] [57]. Separation depends on the protein's intrinsic net charge at the gel pH and its hydrodynamic size (influenced by both mass and shape) [26]. This preservation enables the study of functional properties, protein-protein interactions, oligomeric states, and enzymatic activity post-separation [5] [57]. A classic illustration of these complementary roles is a protein that migrates as a 60 kDa band on non-reducing SDS-PAGE but as a 120 kDa band on Native PAGE, indicating it is a non-covalent dimer of 60 kDa subunits in its native state [10].
Table 1: Core Principles and Applications of SDS-PAGE and Native PAGE
| Feature | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight | Size, charge, and shape |
| Protein State | Denatured (unfolded) | Native (folded) |
| Detergent (SDS) | Present | Absent |
| Sample Heating | Required (typically 70-100°C) | Avoided |
| Reducing Agents | Often used (DTT, BME) | Omitted |
| Protein Function | Lost post-separation | Retained post-separation |
| Primary Applications | Molecular weight determination, purity analysis, western blotting | Studying oligomeric state, protein complexes, enzymatic activity |
Sample Preparation for SDS-PAGE: Protocols and Pitfalls
Standard Denaturing Protocol
The goal of SDS-PAGE sample preparation is complete denaturation, reduction, and uniform SDS coating of all proteins.
- Lysis and Solubilization: Use an appropriate lysis buffer containing 1-2% SDS to ensure complete solubilization and denaturation [58]. For hydrophobic proteins like membrane proteins, supplementation with 6-8 M urea may be necessary [56].
- Inhibit Proteolysis: Immediately add a fresh, broad-spectrum protease inhibitor cocktail to the lysis buffer upon contact with cells or tissues to prevent protein degradation [56] [58].
- Nucleic Acid Digestion: For viscous crude extracts, treat with Benzonase Nuclease to shear nucleic acids, which otherwise can impede migration [56].
- Denaturation and Reduction: Mix the protein lysate with a 2X or 4X SDS-PAGE sample buffer (e.g., Laemmli buffer) containing SDS, a buffer (e.g., Tris), glycerol (for density), a tracking dye, and a reducing agent like Dithiothreitol (DTT) or β-mercaptoethanol (BME) to break disulfide bonds [56] [26].
- Heat Denaturation: Heat samples at 70-100°C for 5-10 minutes to ensure complete unfolding and protease inactivation [56] [26].
- Clarification: Centrifuge heated samples briefly (e.g., 2 minutes at 17,000 x g) to remove any insoluble debris, which can cause streaking [56].
- Quantification and Loading: Determine protein concentration using an assay compatible with detergents (e.g., BCA assay) [58]. Load an appropriate amount per well; for a standard mini-gel, 10-20 µg of total protein for crude samples or 0.5-4 µg for purified proteins is a typical starting point when using Coomassie staining [56].
Common SDS-PAGE Pitfalls and Solutions
Table 2: Troubleshooting SDS-PAGE Sample Preparation
| Problem/Observation | Potential Causes | Solutions |
|---|---|---|
| Protein Degradation (multiple lower molecular weight bands) | • Inactive protease inhibitors• Delay between lysis and heating• Proteases active in sample buffer | • Use fresh protease inhibitors• Keep samples on ice; heat immediately after adding sample buffer [56] [58] |
| Smeared Bands | • Incomplete denaturation/reduction• Protein aggregation• High salt concentration | • Add fresh DTT/BME• Boil samples adequately [26]• Ensure SDS is in excess (3:1 SDS:protein ratio) [56]• Desalt samples or dilute load |
| Sample Leaking from Wells | • Insufficient glycerol in loading buffer• Air bubbles in wells• Overloaded wells | • Ensure adequate glycerol (e.g., 10%) for density [59]• Rinse wells with running buffer before loading• Do not fill wells more than 3/4 capacity [59] |
| Horizontal Smiling/Bulging Bands | • Buffer of incorrect composition/pH• Gel running too hot | • Check running buffer recipe and pH [26]• Run gel at lower voltage or with cooling |
| No or Faint Bands | • Underloaded protein• Protein precipitation before loading | • Accurately quantify protein [58]• Check solubility; consider urea or non-ionic detergents [56] |
| Unexpected Bands (e.g., ~55-65 kDa) | • Keratin contamination from skin or dust | • Wear gloves, use clean equipment, aliquot sample buffers [56] |
Sample Preparation for Native PAGE: Protocols and Pitfalls
Standard Non-Denaturing Protocol
The overarching principle for Native PAGE is to preserve the native state of the protein throughout the entire process.
- Gentle Lysis: Use mild, non-denaturing detergents (e.g., Triton X-100, digitonin) or physical methods (e.g., Dounce homogenization) in a suitable physiological buffer to lyse cells while preserving protein complexes [58] [6].
- Protect Native Structure: Maintain samples at 4°C at all times to minimize denaturation and proteolysis [6]. Protease inhibitors remain essential.
- Avoid Denaturants: Crucially, DO NOT add SDS, urea, or other denaturing agents to the sample or buffer [6].
- No Reduction or Heating: DO NOT add reducing agents (DTT, BME) and DO NOT heat the samples [6] [57].
- Sample Buffer: Use a native sample buffer containing a non-denaturing charged dye (like Coomassie G-250 in Blue Native PAGE), glycerol, and a buffer at an appropriate pH [7].
- Clarification: Centrifuge samples at high speed (e.g., 47,000 x g for 30 minutes) to remove any aggregates or insoluble material that could enter the gel [7].
- Control Buffer Composition: Keep ionic strength low (e.g., < 50 mM NaCl) to prevent disruption of the stacking gel system and band distortion [7] [26]. Dialysis or desalting columns may be necessary.
Common Native PAGE Pitfalls and Solutions
- Loss of Protein Complexes: The use of harsh detergents like SDS or high salt during lysis will dissociate non-covalent complexes. Solution: Employ mild detergents optimized for native complex isolation and keep ionic strength low [58].
- Protein Aggregation and Precipitation: Without SDS to solubilize them, some hydrophobic proteins may aggregate. Solution: Optimize detergent-to-protein ratio and include mild solubilizing agents compatible with native states [59].
- Poor Resolution due to Charge Heterogeneity: Since separation depends on intrinsic charge, proteins with similar mass but different charges may not resolve well. Solution: Consider alternative native techniques like Clear Native PAGE (CN-PAGE) or BN-PAGE for complex mixtures [6].
- Altered Migration from Buffer Conditions: The pH and composition of the running buffer directly impact a protein's net charge and thus its migration. Solution: Carefully design and consistently use the appropriate discontinuous buffer system for Native PAGE [26].
The Scientist's Toolkit: Essential Reagents for Electrophoresis
Table 3: Key Research Reagent Solutions and Their Functions
| Reagent | Function | Key Considerations |
|---|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins; confers uniform negative charge for SDS-PAGE. | Use high-purity grade; ensure constant excess over protein (1.4:1 ratio minimum) [56]. |
| DTT (Dithiothreitol) / BME (β-Mercaptoethanol) | Reduces disulfide bonds in SDS-PAGE. | DTT is more stable and has less odor; add fresh before heating [56] [26]. |
| Protease Inhibitor Cocktail | Prevents proteolytic degradation of samples. | Must be added fresh to lysis buffer; select cocktails based on sample type [58]. |
| Benzonase Nuclease | Degrades DNA and RNA to reduce sample viscosity. | Crucial for viscous lysates; prevents smearing and poor resolution [56]. |
| Urea | Aiding solubilization of difficult proteins (e.g., membrane proteins). | Can carbamylate proteins; use fresh solutions or treat with mixed-bed resins [56]. |
| Coomassie G-250 Dye | Imparts charge for protein migration in Blue Native (BN)-PAGE. | Provides a mild anionic charge without significant denaturation [7]. |
| Tris-Glycine / Bis-Tris Buffers | Common buffer systems for controlling pH during electrophoresis. | Bis-Tris is more stable and avoids reactive amine groups at high pH [7] [57]. |
| Molecular Weight Markers | Calibrate gel for protein size determination. | Use pre-stained for tracking; use unstained or broad-range for accuracy. |
Advanced Considerations and Hybrid Techniques
A notable advancement in the field is the development of Native SDS-PAGE (NSDS-PAGE), a hybrid approach that modifies standard SDS-PAGE conditions to retain some native properties while maintaining high resolution [7]. This method involves removing SDS and EDTA from the sample buffer, omitting the heating step, and significantly reducing the SDS concentration in the running buffer (e.g., to 0.0375%) [7]. Research has demonstrated that this modification dramatically increases the retention of bound metal ions in metalloproteins (e.g., from 26% to 98% for Zn²⁺) and allows most model enzymes to retain their activity post-electrophoresis, bridging the gap between the high resolution of SDS-PAGE and the functional preservation of BN-PAGE [7].
Furthermore, artifacts like the cleavage of Asp-Pro bonds can occur during prolonged heating at 100°C, and chemicals leaching from disposable plasticware can interfere with electrophoresis [56]. Awareness of these subtle issues and adherence to best practices, such as controlled heating times and rinsing plasticware, are hallmarks of a meticulous researcher.
Workflow and Decision Pathway
The following workflow diagram summarizes the key decision points and steps for preparing samples for SDS-PAGE and Native PAGE.
Mastering sample preparation is a prerequisite for generating reliable and interpretable data in protein electrophoresis. The distinct requirements of SDS-PAGE and Native PAGE demand a disciplined and informed approach from the researcher. By understanding the underlying principles, meticulously following optimized protocols, and proactively troubleshooting common pitfalls, scientists can confidently prepare samples that accurately reflect their biological questions. This rigorous approach to sample preparation ensures that the powerful techniques of SDS-PAGE and Native PAGE continue to yield valid insights into protein structure, function, and interaction, thereby advancing research and drug development efforts.
Data Interpretation and Technique Selection
In protein separation research, the choice between Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Native PAGE fundamentally dictates the type of information that can be obtained about a protein's structure and assembly. These techniques serve as critical tools for researchers and drug development professionals seeking to understand protein function, purity, and quaternary structure. The core distinction lies in their treatment of the native protein state: SDS-PAGE denatures proteins into their constituent polypeptides, while Native PAGE preserves the protein's higher-order structure and biological activity [6] [12].
The interpretation of banding patterns, especially for complex structures like dimers, requires a thorough understanding of these underlying principles. When a protein sample isolated from a natural source is analyzed using both techniques, the differential migration patterns reveal essential information about subunit composition, molecular weight, and the nature of the forces holding the subunits together [10]. This case study examines dimer analysis to illustrate how the complementary use of denaturing and non-denaturing electrophoresis provides a more complete structural picture, which is indispensable for characterizing therapeutic proteins, enzymes, and other biologically relevant macromolecules.
Technical Comparison: SDS-PAGE versus Native PAGE
The fundamental differences between SDS-PAGE and Native PAGE stem from their sample preparation methods, buffer compositions, and operational conditions. These variables directly impact whether a protein retains its native conformation or becomes denatured into polypeptide chains.
Table 1: Key Technical Differences Between SDS-PAGE and Native PAGE
| Parameter | SDS-PAGE | Native PAGE |
|---|---|---|
| Gel State | Denaturing [6] [5] | Non-denaturing [6] [5] |
| SDS Presence | Present (anionic detergent) [6] | Absent [6] [19] |
| Sample Preparation | Heated with SDS and reducing agents [6] [12] | Not heated; no denaturants [6] |
| Separation Basis | Molecular weight of polypeptides [6] [12] | Size, charge, and shape of native protein [6] [12] |
| Protein Net Charge | Uniformly negative [6] [60] | Intrinsic charge (positive or negative) [6] |
| Protein State | Denatured/unfolded [6] [5] | Native/folded [6] [5] |
| Protein Function Post-Separation | Lost [6] | Retained [6] |
| Protein Recovery | Typically not recoverable functionally [6] | Can be recovered in functional form [6] |
| Common Running Temperature | Room Temperature [6] | 4°C [6] |
| Primary Applications | Molecular weight determination, purity checks, expression analysis [6] [60] | Studying native structure, subunit composition, functional assays, protein purification [6] |
The Role of SDS and Denaturation
In SDS-PAGE, the anionic detergent sodium dodecyl sulfate (SDS) plays a pivotal role by binding to hydrophobic regions of proteins at a nearly constant ratio of 1.4 g SDS per 1 g of protein [12] [60]. This binding confers a uniform negative charge density, effectively masking the protein's intrinsic charge. The sample is also heated in the presence of reducing agents like β-mercaptoethanol or dithiothreitol (DTT), which cleave disulfide bonds [6] [60]. Consequently, proteins unfold into linear polypeptides, and their migration through the polyacrylamide gel matrix depends almost exclusively on molecular weight [12]. Smaller polypeptides migrate faster, while larger ones migrate more slowly.
Preservation of Native Structure
In contrast, Native PAGE is performed without denaturing agents. The gel lacks SDS, and samples are not heated [6]. This preserves the protein's secondary, tertiary, and quaternary structures. During electrophoresis, migration depends on the protein's intrinsic charge, size, and three-dimensional shape [12]. Proteins with a higher negative charge density migrate faster toward the anode, while the gel matrix creates a sieving effect that retards larger proteins more than smaller ones [12]. Because functionality is retained, proteins separated by Native PAGE can be excised from the gel and used in subsequent activity assays [6].
Case Study: Dimer Analysis Through Comparative Electrophoresis
A classic example in protein analysis involves a sample that migrates as a 60 kDa band on a non-reducing SDS-PAGE gel but migrates at a position corresponding to 120 kDa on a Native PAGE gel [10]. This discrepancy provides a clear opportunity to interpret banding patterns for quaternary structure determination.
Experimental Observation and Inferences
The observation of different apparent molecular weights in the two techniques leads to a reasonable inference: the protein exists as a dimer of 60 kDa subunits that are not linked by disulfide bonds [10].
The logical flow for this interpretation is as follows:
- SDS-PAGE Analysis (Non-reducing conditions): The migration at 60 kDa indicates that the fundamental polypeptide unit has a molecular weight of 60 kDa. The use of "non-reducing" conditions is critical; it means no reducing agent (like DTT) was added, so any disulfide bonds within or between subunits would remain intact. The fact that the protein still runs as a 60 kDa monomer under these conditions demonstrates that the dimer is not stabilized by covalent disulfide linkages [10].
- Native PAGE Analysis: The migration corresponding to 120 kDa confirms that in its native, functional state, the protein exists as a larger complex. The doubling of the molecular weight strongly suggests a dimeric quaternary structure [10].
The conclusion is that the two 60 kDa monomers associate into a 120 kDa dimer through non-covalent interactions, such as hydrophobic forces, hydrogen bonding, or electrostatic attractions. These weak interactions are disrupted by the denaturing action of SDS in SDS-PAGE, but they remain intact in the gentle conditions of Native PAGE [10].
Detailed Experimental Protocols
To obtain the results described in the case study, the following standardized methodologies can be employed.
Non-Reducing SDS-PAGE Protocol
- Sample Preparation: Mix the protein sample with an SDS-based loading buffer (e.g., LDS sample buffer). Crucially, omit reducing agents like DTT or β-mercaptoethanol. Heat the sample at 70°C for 10 minutes to ensure complete denaturation by SDS [7].
- Gel Preparation: Use a standard SDS-PAGE gel, such as a precast NuPAGE Novex 12% Bis-Tris gel [7]. The gel is cast in a buffer containing SDS.
- Running Buffer: Use an SDS-running buffer, for example, 50 mM MOPS, 50 mM Tris Base, 0.1% SDS, pH 7.7 [7].
- Electrophoresis Conditions: Load the prepared samples and molecular weight markers. Run the gel at a constant voltage of 200V for approximately 45 minutes at room temperature until the dye front reaches the bottom of the gel [7].
- Staining: After electrophoresis, visualize the protein bands using a compatible stain such as Coomassie Blue, GelCode Blue, or SimplyBlue SafeStain [12] [61].
Native PAGE Protocol
- Sample Preparation: Mix the protein sample with a non-denaturing loading buffer. A typical Native PAGE sample buffer may contain 50 mM BisTris, 50 mM NaCl, 10% glycerol, and 0.001% Ponceau S, pH 7.2 [7]. Do not heat the sample.
- Gel Preparation: Use a gel that contains no SDS or other denaturants. A Native-PAGE Novex 4-16% Bis-Tris gradient gel is suitable for resolving the native protein [7].
- Running Buffer: Use non-denaturing anode and cathode buffers. For example, the cathode buffer may contain 50 mM BisTris, 50 mM Tricine, and 0.02% Coomassie G-250, while the anode buffer contains 50 mM BisTris and 50 mM Tricine, pH 6.8 [7].
- Electrophoresis Conditions: Load the samples and native protein standards. Run the gel at a constant voltage of 150V for 90-95 minutes at room temperature [7]. To minimize denaturation during the run, the apparatus can be kept cool or run in a cold room [6] [12].
- Staining: Detect proteins using a compatible stain, ensuring the staining and destaining process does not denature the proteins if functional recovery is desired.
Table 2: Key Reagents for Dimer Analysis Experiments
| Research Reagent | Function in Experiment |
|---|---|
| SDS (Sodium Dodecyl Sulfate) | Denatures proteins and imparts uniform negative charge for SDS-PAGE [6] [60]. |
| Bis-Tris Gels | Common gel matrix providing stable pH environment for both SDS-PAGE and Native PAGE [7]. |
| DTT or β-Mercaptoethanol | Reducing agent that breaks disulfide bonds (used in reducing SDS-PAGE, omitted in non-reducing) [6] [60]. |
| Coomassie G-250 | A dye used in Native PAGE running buffer (e.g., BN-PAGE) and for staining proteins post-electrophoresis [7]. |
| Molecular Weight Markers | Pre-stained or unstained protein ladders for estimating the molecular weight of separated proteins [12] [61]. |
| Glycerol | Added to samples to increase density, preventing diffusion from wells during loading [6] [7]. |
Advanced Applications and Hybrid Techniques
Beyond Dimer Analysis: The Role in Drug Development
The principles of dimer analysis extend directly to the development and quality control of biopharmaceuticals, particularly monoclonal antibodies (MAbs). MAbs are large, multi-subunit proteins, and their purity, integrity, and glycosylation status are critical quality attributes. SDS-PAGE is routinely used to assess antibody purity and detect fragments or aggregates [61]. For example, analysis of a heat-stressed IgG antibody by SDS-PAGE can reveal degradation bands corresponding to heavy chains (∼50 kDa), light chains (∼25 kDa), and various aggregates [61]. Capillary electrophoresis-SDS (CE-SDS) has emerged as a quantitative, high-resolution automated alternative to traditional slab gel SDS-PAGE for these applications in quality control labs [61].
Native SDS-PAGE (NSDS-PAGE): A Hybrid Approach
To address the limitation of standard SDS-PAGE, which destroys native structure, a modified method called Native SDS-PAGE (NSDS-PAGE) has been developed. This technique reduces the SDS concentration in the running buffer (e.g., to 0.0375%) and omits SDS and EDTA from the sample buffer along with the heating step [7]. This protocol represents a middle ground, maintaining the high resolution of traditional SDS-PAGE while allowing many proteins to retain their enzymatic activity and bound metal cofactors. In one study, this method retained 98% of Zn²⁺ bound in proteomic samples, and seven out of nine model enzymes remained active after electrophoresis [7].
The case study of dimer analysis underscores the critical importance of selecting and correctly interpreting protein separation techniques. SDS-PAGE and Native PAGE are not interchangeable but are powerfully complementary. SDS-PAGE provides information about the molecular weight of polypeptide subunits under denaturing conditions, while Native PAGE reveals the size, charge, and oligomeric state of the functional, native protein. For researchers and drug development professionals, a strategic combination of these methods, and an understanding of advanced techniques like NSDS-PAGE, provides a robust framework for elucidating protein structure, function, and stability—a foundation essential for successful biomedical research and therapeutic development.
Protein electrophoresis is a foundational technique in molecular biology and biochemistry that enables the separation of complex protein mixtures using an electric field applied through a porous gel matrix. The most common support matrix for protein separation is polyacrylamide, which forms a tunable molecular sieve [12]. Within this domain, Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Native PAGE represent two fundamentally different approaches to protein separation, each with distinct mechanisms, applications, and outcomes. SDS-PAGE, developed by Ulrich K. Laemmli, separates proteins based primarily on molecular weight under denaturing conditions [6]. In contrast, Native PAGE, with origins attributed to Ornstein and Davis, separates proteins based on their intrinsic charge, size, and three-dimensional shape under non-denaturing conditions, thereby preserving their native conformation and biological activity [6] [12]. This whitepaper provides an in-depth technical comparison of these two critical techniques, framing their differences within the context of protein separation research for scientists and drug development professionals.
Core Comparison: Separation Mechanism and Outcomes
The fundamental difference between these techniques lies in the state of the protein during separation. SDS-PAGE employs a denaturing approach that linearizes proteins and masks their inherent charge, while Native PAGE maintains proteins in their native, folded state, allowing separation based on a combination of properties.
Table 1: Fundamental Differences Between SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight (mass) [6] [12] | Size, overall charge, and shape [6] [12] |
| Gel Type | Denaturing [6] | Non-denaturing [6] |
| SDS Presence | Present (denaturing agent) [6] | Absent [6] |
| Reducing Agent | Present (e.g., DTT, BME) [6] | Absent [6] |
| Sample Preparation | Heated [6] | Not heated [6] |
| Protein State | Denatured/unfolded [6] [12] | Native/folded [6] [12] |
| Net Protein Charge | Uniformly negative (from SDS coating) [6] [12] | Positive, negative, or neutral (intrinsic charge) [6] |
| Protein Function Post-Separation | Lost [6] | Retained [6] |
| Protein Recovery | Typically not recovered functionally [6] | Can be recovered in functional form [6] |
| Primary Applications | Molecular weight determination, purity check, protein expression analysis [6] [11] | Studying protein structure, subunit composition, enzyme activity, protein-protein interactions [6] [12] |
Table 2: Technical and Practical Considerations
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Typical Running Temperature | Room Temperature [6] | 4°C (to maintain stability) [6] |
| Buffer System | Discontinuous (Stacking & Resolving gels) [12] | Varies; can be continuous or discontinuous |
| Interpretation of MW | Straightforward; relative to protein ladder [12] | Complex; influenced by charge and shape [12] |
| Key Limitations | Destroys native structure/function; no info on oligomeric state [62] | Less accurate for MW determination; complex migration [62] |
| Common Variants | Reducing vs. Non-reducing SDS-PAGE [11] | Blue Native (BN)-PAGE, Clear Native (CN)-PAGE [6] |
The following workflow illustrates the key procedural differences between SDS-PAGE and Native PAGE, from sample preparation to final analysis:
Underlying Mechanisms: A Deeper Dive
The Role of SDS in Denaturation and Charge Manipulation
In SDS-PAGE, the anionic detergent sodium dodecyl sulfate (SDS) plays a pivotal role. When a protein sample is heated (typically between 70-100°C) in the presence of SDS and a reducing agent like Dithiothreitol (DTT) or 2-Mercaptoethanol (BME), several transformative events occur [6] [12]. The reducing agent cleaves disulfide bonds, while SDS denatures the protein by wrapping around the polypeptide backbone. This process dissociates protein complexes into their subunits and unfolds the tertiary structure. Crucially, SDS binds to the denatured polypeptides in a constant weight ratio (approximately 1.4 g SDS per 1 g of protein), imparting a uniform negative charge density along the entire length of the polypeptide chain [12]. This charge masking is the key to separation by molecular weight alone. The resulting SDS-polypeptide complexes have similar shapes and charge-to-mass ratios, ensuring that during electrophoresis, their migration through the polyacrylamide gel is inversely proportional to the logarithm of their molecular mass, with smaller polypeptides moving faster [12].
Native PAGE and the Preservation of Protein Structure
Native PAGE operates on the principle of separating proteins based on their intrinsic properties without the use of denaturing agents. The migration of a protein in a native gel is a complex function of its size (hydrodynamic radius), its inherent net charge at the running buffer's pH, and its three-dimensional shape [12]. The gel matrix acts as a sieve, creating a frictional force that retards the movement of larger or more irregularly shaped proteins. Because the protein's net charge is not masked, it interacts directly with the electric field; proteins with a higher negative charge density (more charge per unit mass) will migrate faster toward the anode [12]. This technique is exceptionally valuable for studying multimeric proteins, as subunit interactions within a protein complex are generally retained [12]. Furthermore, the preservation of the native structure allows proteins to be recovered from the gel post-electrophoresis via passive diffusion or electro-elution, often with retained enzymatic activity, making it a powerful tool for functional studies and purification [6] [12].
Experimental Protocols and Methodologies
Standard SDS-PAGE Protocol
The following protocol is widely used for analytical SDS-PAGE, based on the Laemmli system [6] [12].
- Gel Preparation: A discontinuous gel system is used, comprising a resolving gel (e.g., 12% acrylamide for proteins ~10-60 kDa) and a stacking gel (lower acrylamide percentage, e.g., 4-5%). The resolving gel has a higher pH (e.g., ~8.8) and is responsible for separating proteins by size. The stacking gel, with a lower pH (~6.8), serves to concentrate all protein samples into a sharp band before they enter the resolving gel, thereby improving resolution [12].
- Sample Preparation: Protein samples are diluted in a buffer containing SDS, a reducing agent (DTT or 2-ME), glycerol (for density), and a tracking dye like Bromophenol Blue. This mixture is then heated at 95-100°C for 5-10 minutes to ensure complete denaturation and reduction [6] [12] [63].
- Electrophoresis: The prepared samples and a molecular weight marker (protein ladder) are loaded into the wells. The gel is run in a buffer containing Tris, glycine, and SDS (e.g., Tris-Glycine-SDS) at a constant voltage (e.g., 120-200 V) until the tracking dye front reaches the bottom of the gel [12].
- Post-Electrophoresis Analysis: The gel is typically stained with Coomassie Brilliant Blue, silver stain, or fluorescent dyes to visualize the separated protein bands. Alternatively, proteins can be transferred to a membrane for Western blot analysis [6] [12].
Standard Native PAGE Protocol
The Native PAGE protocol differs significantly in its sample and buffer composition to preserve protein function [6].
- Gel Preparation: The polyacrylamide gel is cast without SDS or other denaturants. The buffer system is chosen to maintain a pH that preserves protein activity and charge; common choices are Tris-Glycine or Tris-Borate systems at a mildly alkaline pH [6] [12].
- Sample Preparation: The critical distinction is that the sample buffer contains no SDS, no reducing agents, and the sample is not heated. The buffer usually includes glycerol to facilitate loading and may contain a non-denaturing tracking dye [6]. To maintain protein integrity, the sample should be kept cold throughout preparation.
- Electrophoresis: The gel apparatus is often run in a cold room or with a cooling unit to prevent heat-induced denaturation during the run (typically at 4°C) [6]. The running buffer also lacks SDS. Since proteins retain their native charge, the polarity of the electrodes must be considered; most native proteins are negatively charged at alkaline pH and will migrate toward the anode, but for basic proteins, the electrode polarity may need to be reversed [12].
- Post-Electrophoresis Analysis: Proteins can be detected using gentle staining methods. A significant advantage is that functional assays, such as in-gel activity staining for enzymes, can be performed to confirm the protein is active [12].
Advanced Applications and Hybrid Techniques
Applications in Food Science and Disease Research
The choice of electrophoresis technique directly impacts the type of biological information that can be obtained. SDS-PAGE is a workhorse in food science for protein profiling, allergen detection, and quality assessment across various products like cereals, pulses, dairy, and meats [11]. It is indispensable for verifying the integrity of proteins in raw materials and finished products and for detecting adulteration [11]. In medical research, SDS-PAGE, particularly in its non-reducing form (without DTT/2-ME), is crucial for analyzing disulfide-bonded proteins. For instance, it has been used to study proinsulin misfolding in pancreatic beta cells, where the formation of aberrant intermolecular disulfide-linked complexes is a feature investigated in the context of diabetes [63].
Native PAGE, particularly Blue Native PAGE (BN-PAGE), is the method of choice for studying intact protein complexes, such as those involved in mitochondrial oxidative phosphorylation [7]. Its ability to separate proteins based on native mass and charge makes it ideal for analyzing protein-protein interactions, oligomeric states, and for purifying active proteins for downstream functional assays [6] [62].
Native SDS-PAGE (NSDS-PAGE): A Hybrid Approach
To address the limitations of both standard techniques, a hybrid method called Native SDS-PAGE (NSDS-PAGE) has been developed. This approach seeks to combine the high resolution of SDS-PAGE with the functional preservation of Native PAGE [7]. The modifications include:
- Removing SDS and EDTA from the sample buffer.
- Omitting the heating step during sample preparation.
- Drastically reducing the SDS concentration in the running buffer (e.g., from 0.1% to 0.0375%) [7].
Research has shown that this method can retain Zn²⁺ bound in proteomic samples with high efficiency (increasing from 26% in standard SDS-PAGE to 98% in NSDS-PAGE) and preserve the activity of many model enzymes post-electrophoresis, offering a powerful compromise for metalloprotein analysis [7].
The Scientist's Toolkit: Essential Research Reagents
Successful protein separation requires a suite of specific reagents, each with a defined role in the process.
Table 3: Key Reagents for SDS-PAGE and Native PAGE
| Reagent | Function | Key Consideration |
|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked polyacrylamide gel matrix; pore size determines separation range [12]. | Total concentration and cross-linker ratio dictate gel porosity and resolution. |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers uniform negative charge (for SDS-PAGE) [6] [12]. | Must be in excess relative to protein for consistent binding. |
| DTT or β-Mercaptoethanol | Reducing agents that break disulfide bonds (for reducing SDS-PAGE) [6] [11]. | Essential for analyzing subunit composition. Omitted in non-reducing SDS-PAGE. |
| Tris-based Buffers | Provides the conductive and pH environment for electrophoresis [12]. | Discontinuous systems (stacking/resolving) use different pH and composition. |
| Ammonium Persulfate (APS) & TEMED | Catalyzes the polymerization of acrylamide to form the gel [12]. | Fresh APS is critical for efficient and consistent gel polymerization. |
| Tracking Dye (e.g., Bromophenol Blue) | Anionic dye that migrates ahead of proteins to visualize electrophoresis progress [64]. | Must be anionic to migrate toward the anode with the proteins. |
| Coomassie Brilliant Blue | Used in Blue Native (BN)-PAGE; confers negative charge to native proteins [6] [7]. | Allows rough molecular weight estimation under native conditions. |
| Molecular Weight Markers | A mixture of proteins of known sizes for calibrating the gel and estimating protein mass [12]. | Must be chosen to match the separation range and conditions (denaturing vs. native). |
The selection between SDS-PAGE and Native PAGE is a fundamental strategic decision in experimental design. SDS-PAGE remains the gold standard for determining molecular weight, assessing purity, and analyzing complex mixtures where high resolution is the primary goal. Native PAGE is indispensable for all studies requiring the preservation of a protein's native conformation, quaternary structure, and biological activity, such as in enzymology, studies of protein complexes, and functional proteomics. The continued evolution of these techniques, including the development of hybrid methods like NSDS-PAGE, ensures that gel electrophoresis will remain a cornerstone of protein research and biopharmaceutical development.
Selecting the Right Technique for Your Research Objective
Protein gel electrophoresis is a foundational technique in molecular biology and biochemistry laboratories for separating complex protein mixtures based on their physical properties. When transported through a polyacrylamide gel matrix under the influence of an electrical field, charged protein molecules migrate at rates dependent on their charge, size, and shape [12]. Two predominant methodologies have emerged for protein separation: SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis), which denatures proteins to separate them primarily by molecular weight, and Native-PAGE, which maintains proteins in their native state to separate them based on charge, size, and three-dimensional structure [5] [6]. The critical choice between these techniques directly determines the type of biological information obtainable, making selection based on research objectives paramount for experimental success. This guide provides an in-depth technical comparison to inform this decision, framed within the broader context of protein separation research.
Technical Comparison: SDS-PAGE vs. Native PAGE
The fundamental distinction between these techniques lies in their treatment of protein structure. SDS-PAGE employs the anionic detergent sodium dodecyl sulfate (SDS) to denature proteins, masking their intrinsic charge and unfolding them into linear chains [5] [12]. This results in separation based almost exclusively on polypeptide size. In contrast, Native PAGE uses non-denaturing conditions without SDS, preserving proteins' secondary, tertiary, and quaternary structures, native charge, and biological activity [65] [6]. This preservation allows separation based on the combined factors of intrinsic charge and hydrodynamic size.
Table 1: Fundamental Differences Between SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Basis | Molecular weight [12] [6] | Size, charge, and shape [12] [6] |
| Protein State | Denatured and linearized [5] | Native, folded conformation [5] |
| SDS in Gel | Present [6] | Absent [6] |
| Sample Preparation | Heated with SDS and reducing agents [66] [6] | Not heated; no denaturing agents [65] [6] |
| Net Charge on Proteins | Uniformly negative [5] | Intrinsic charge (can be positive or negative) [6] |
| Protein Function Post-Separation | Destroyed [7] [5] | Largely retained [7] [5] |
| Key Applications | Molecular weight determination, purity assessment, western blotting [7] [12] [26] | Studying oligomeric state, protein-protein interactions, enzymatic activity [5] [12] |
Table 2: Performance and Practical Considerations
| Aspect | SDS-PAGE | Native PAGE |
|---|---|---|
| Resolution | High resolution by mass [7] | Lower resolution for complex mixtures [7] |
| Typical Running Temperature | Room Temperature [6] | 4°C (to maintain protein stability) [6] |
| Protein Recovery & Downstream Use | Not suitable for functional studies; used for MS, sequencing [5] [67] | Can be recovered for activity assays, interaction studies [5] [12] |
| Compatibility with Mass Spectrometry | Excellent following destaining [67] | Possible, but cross-linking from some stains (e.g., silver) can interfere [67] |
| Comparative Quantitation | Advantageous for visualizing quantity differences between samples [68] | Advantageous for visualizing native protein interactions [68] |
Experimental Protocols and Methodologies
SDS-PAGE Standard Protocol
The following protocol is adapted from standard laboratory procedures [66] [69].
1. Gel Preparation:
- Decide an appropriate acrylamide percentage (e.g., 12% for 40-100 kDa proteins, 8% for larger proteins) [26].
- For a 12% resolving gel, mix 14 mL of 30% acrylamide/bis-acrylamide stock, 8.75 mL of Tris-HCl pH 8.8, 12.25 mL of double-distilled water, 175 µL of 10% ammonium persulfate (APS), and 15 µL of TEMED. Pour between glass plates and overlay with water or isopropanol to ensure even polymerization [69].
- Once set, prepare a 5% stacking gel (e.g., 2 mL acrylamide stock, 3 mL Tris-HCl pH 6.8, 6.9 mL water, 100 µL 10% APS, 10 µL TEMED). Insert a comb to create sample wells [69].
2. Sample Preparation:
- Mix protein sample with an equal volume of 2X Laemmli sample buffer, which contains SDS and a reducing agent like β-mercaptoethanol (BME) or DTT [66].
- Heat the samples at 95°C for 5-10 minutes to fully denature the proteins [66] [69].
- Centrifuge briefly to pellet any debris [66].
3. Electrophoresis:
- Place the gel in a chamber and fill with 1X running buffer (e.g., 25 mM Tris, 192 mM glycine, 0.1% SDS, pH ~8.3) [66] [65].
- Load prepared samples and protein molecular weight markers into the wells.
- Connect to a power supply and run at a constant voltage of 150-200 V until the dye front migrates to the bottom of the gel (approximately 45-90 minutes) [66] [69].
Native PAGE Standard Protocol
1. Gel Preparation:
- Native PAGE gels contain no SDS. A typical resolving gel (e.g., 8%) can be made with 2.6 mL of 30% acrylamide/bis-acrylamide stock, 7.29 mL of 0.375 M Tris-HCl pH 8.8, 100 µL of 10% APS, and 10 µL of TEMED [65].
- A stacking gel with a lower acrylamide concentration and pH 6.8 is often used [65].
2. Sample Preparation:
- Mix the protein sample with a non-denaturing 2X sample buffer (e.g., containing 62.5 mM Tris-HCl pH 6.8, 25% glycerol, and 0.01% Bromophenol Blue) [65].
- Crucially, do not heat the samples and avoid SDS or reducing agents [65] [6].
3. Electrophoresis:
- Use a running buffer without SDS, such as Tris-Glycine (25 mM Tris, 192 mM glycine, pH ~8.3) [65].
- Load samples and run at a constant voltage, typically between 100-150 V. It is advisable to run the gel in a cold room or on ice to prevent heat-induced denaturation [65] [6].
A Hybrid Approach: Native SDS-PAGE (NSDS-PAGE)
A modified method known as Native SDS-PAGE (NSDS-PAGE) has been developed to balance the high resolution of SDS-PAGE with the functional preservation of Native PAGE. This technique involves:
- Sample Buffer: Omitting SDS and EDTA, and skipping the heating step [7].
- Running Buffer: Using a significantly reduced SDS concentration (e.g., 0.0375% instead of 0.1%) and deleting EDTA [7].
- Outcome: This method was shown to increase the retention of bound metal ions like Zn²⁺ from 26% to 98% compared to standard SDS-PAGE, and most model enzymes retained their activity post-separation while still achieving high-resolution separation [7].
Workflow and Decision Pathways
The following diagram illustrates the key decision-making process for selecting and executing the appropriate electrophoresis technique.
Electrophoresis Technique Selection Workflow
Research Reagent Solutions
Successful electrophoresis relies on a defined set of reagents, each serving a specific function in the separation process.
Table 3: Essential Reagents for Protein Electrophoresis
| Reagent / Material | Function / Purpose | Key Considerations |
|---|---|---|
| Acrylamide/Bis-acrylamide | Forms the cross-linked porous gel matrix that acts as a molecular sieve [12]. | Ratio and total concentration determine gel pore size and resolution range [12]. |
| SDS (Sodium Dodecyl Sulfate) | Anionic detergent that denatures proteins and confers a uniform negative charge [66] [12]. | Used in SDS-PAGE only. Binds ~1.4g per 1g of polypeptide [12]. |
| APS & TEMED | Ammonium persulfate (APS) and TEMED catalyze the polymerization of acrylamide [12] [69]. | Added last; TEMED stabilizes the free-radical chain reaction initiated by APS [12]. |
| Tris-based Buffers | Provides the conductive ionic environment and maintains stable pH during electrophoresis [65] [69]. | Discontinuous systems (different pH in stacker/resolver) improve band sharpness [12] [26]. |
| Reducing Agents (BME, DTT) | Breaks disulfide bonds to fully denature proteins into individual subunits [66]. | Critical for accurate MW determination in SDS-PAGE; omitted in Native PAGE [66] [6]. |
| Coomassie Brilliant Blue | A dye that binds non-covalently to proteins, enabling visualization after separation [67]. | Common, cost-effective; detects ~25 ng/band. Reversible, making it MS-compatible [67]. |
| Molecular Weight Markers | A mixture of proteins of known sizes for calibrating the gel and estimating sample protein sizes [66] [26]. | Can be prestained (for tracking) or unstained (for accuracy) [66]. |
SDS-PAGE and Native PAGE are complementary, not competing, techniques in the protein researcher's toolkit. The optimal choice is irrevocably tied to the specific research question. SDS-PAGE is the unequivocal method for determining molecular weight, assessing purity, and preparing samples for western blotting or mass spectrometry identification. Native PAGE is indispensable for probing functional aspects of proteins, including enzymatic activity, native oligomerization, and protein-protein interactions. The emergence of hybrid techniques like NSDS-PAGE [7] further blurs the lines, offering pathways to retain certain native properties while leveraging the resolution of SDS-based systems. By understanding the fundamental principles, practical protocols, and comparative strengths outlined in this guide, researchers can make an informed selection that optimally aligns with their scientific objectives, thereby ensuring robust and interpretable experimental outcomes.
Complementary Use with Other Analytical Methods (e.g., Mass Spectrometry)
In mass spectrometry (MS)-based proteomics, the high complexity of biological samples presents a significant analytical challenge. Fractionation of complex protein or peptide mixtures is an indispensable strategy to reduce this complexity and improve the sensitivity and depth of proteomic profiling [70]. Gel-based separation techniques, primarily SDS-PAGE and Native PAGE, serve as powerful, inexpensive, and straightforward front-end fractionation methods that are compatible with downstream MS analysis [70] [71]. These techniques allow researchers to separate proteins based on different physicochemical properties before MS identification, thereby increasing the total number of proteins that can be detected.
The fundamental difference between these techniques lies in their preservation of protein structure. SDS-PAGE denatures proteins, separating them primarily by molecular weight, while Native PAGE maintains proteins in their native, folded state, separating them based on size, charge, and shape [6] [12]. This core distinction dictates their complementary applications in structural proteomics, where SDS-PAGE is ideal for determining molecular weights and purity, and Native PAGE is preferred for studying functional protein complexes, interactions, and enzymatic activities [6] [5].
Fundamental Principles of SDS-PAGE and Native PAGE
Comparative Mechanism of Separation
Table 1: Core Differences Between SDS-PAGE and Native PAGE
| Criteria | SDS-PAGE | Native PAGE |
|---|---|---|
| Separation Principle | Molecular weight only [6] | Size, overall charge, and shape [6] [12] |
| Protein State | Denatured, linearized polypeptides [12] | Native conformation, folded state [6] |
| Detergent | SDS present, denatures proteins and confers uniform negative charge [12] | No SDS or other denaturing detergents [6] |
| Sample Preparation | Heated with SDS and reducing agents [6] | Not heated, no reducing agents [6] |
| Protein Function | Function lost [6] | Function often retained [6] [12] |
| Protein Recovery | Difficult to recover functional proteins post-separation [6] | Can be recovered in functional form for downstream assays [6] [72] |
| Primary Applications | Molecular weight determination, purity check, expression analysis [6] | Study of protein complexes, oligomerization, enzymatic activity [6] [5] |
Visualization of Technique Selection
The following diagram outlines the decision-making workflow for selecting the appropriate electrophoresis technique based on research goals, particularly when coupled with mass spectrometry.
SDS-PAGE in Mass Spectrometry Workflows
GeLC-MS/MS: A Cornerstone Proteomics Workflow
The GeLC-MS/MS workflow is one of the most common and efficient gel-based fractionation techniques, where a complex protein sample is first separated by 1-D SDS-PAGE, the gel is sliced into multiple bands, each band is subjected to in-gel enzymatic digestion, and the resulting peptide mixtures are analyzed by nanoLC-MS/MS [70]. This method is highly effective because SDS-PAGE fractionates proteins across a wide molecular weight range, substantially simplifying the protein mixture loaded into the mass spectrometer in any single run, which increases the total number of identified proteins [70].
Intact Protein Recovery for Top-Down Proteomics
While traditional GeLC-MS/MS relies on in-gel digestion for bottom-up proteomics, there is growing interest in top-down proteomics, which analyzes intact proteins. A major historical obstacle has been the inefficient recovery of intact proteins from polyacrylamide gels. The PEPPI-MS (Passively Eluting Proteins from Polyacrylamide Gels as Intact species for MS) workflow overcomes this [71]. PEPPI-MS uses Coomassie Brilliant Blue (CBB) as an extraction enhancer, allowing high-recovery (mean 68% for proteins <100 kDa) of intact proteins from gel pieces after just 10 minutes of shaking [71]. This enables a powerful GeLC-MS workflow for top-down proteomics, preserving information about proteoforms and post-translational modifications that is lost during peptide digestion.
Detailed SDS-PAGE Protocol for GeLC-MS/MS
Table 2: Key Research Reagents for SDS-PAGE and Downstream MS
| Reagent / Material | Function in Protocol |
|---|---|
| Polyacrylamide Gel (e.g., 4-12% gradient) | Sieving matrix for protein separation by molecular weight [12]. |
| Sodium Dodecyl Sulfate (SDS) | Denatures proteins and confers uniform negative charge, masking intrinsic charge [73] [12]. |
| Reducing Agent (DTT or β-mercaptoethanol) | Cleaves disulfide bonds to fully denature protein subunits [6] [42]. |
| Ammonium Persulfate (APS) & TEMED | Catalyzes the polymerization of acrylamide to form the gel matrix [12]. |
| Molecular Weight Markers | Provide size standards for estimating protein molecular weight and guiding gel slicing [12]. |
| Coomassie Brilliant Blue (CBB) Staining Solution | Visualizes separated protein bands for excision; in PEPPI-MS, enhances passive protein extraction [71]. |
| Trypsin (Proteomic Grade) | Proteolytic enzyme for in-gel digestion of proteins into peptides for bottom-up MS [70]. |
| Extraction Buffer (0.05% SDS/100 mM Ammonium Bicarbonate) | PEPPI-MS solution for passive elution of intact proteins from gel pieces [71]. |
Protocol Steps:
- Sample Preparation: Dilute protein samples in SDS-PAGE sample buffer (e.g., containing 63 mM Tris-HCl, 2% SDS, 10% glycerol, 0.0025% bromophenol blue, pH 6.8) supplemented with a reducing agent like 50 mM DTT [70]. Heat samples at 70-100°C for 5-10 minutes to ensure complete denaturation [12].
- Gel Electrophoresis: Load samples and molecular weight markers onto a polyacrylamide gel (e.g., a criterion 8-16% gel). Apply a constant voltage (e.g., 200V) until the dye front migrates to the bottom of the gel [70] [12].
- In-Gel Visualization and Excision: After electrophoresis, stain the gel with a compatible stain like Coomassie Brilliant Blue. Destain as needed to visualize protein bands. Using a clean scalpel, excise entire lanes into slices based on molecular weight markers. Each slice constitutes one fraction [70] [71].
- In-Gel Digestion (for Bottom-Up MS): For standard GeLC-MS/MS, destain gel pieces, reduce and alkylate cysteines in-gel, and digest proteins with trypsin overnight. Extract peptides from the gel, dry down, and reconstitute for LC-MS/MS analysis [70].
- Intact Protein Extraction (for Top-Down MS): For PEPPI-MS, place gel pieces in a disposable homogenizer. Grind the gel thoroughly with a pestle. Add extraction buffer (0.05% SDS/100 mM ammonium bicarbonate) and shake for 10 minutes to passively elute proteins. The extracted proteins can then be purified and analyzed directly by LC-MS for top-down proteomics [71].
Native PAGE in Mass Spectrometry Workflows
Preserving Native Structures for Functional Insight
Native PAGE is uniquely valuable when MS analysis must be performed on proteins in their native, functional state. Because the technique preserves higher-order structure and protein-protein interactions, it is ideal for coupling with native mass spectrometry and cross-linking mass spectrometry (XL-MS), techniques designed to probe protein complexes and quaternary structures [71]. Following Native PAGE, functional proteins can be electro-eluted or passively diffused from the gel for downstream analysis, maintaining their activity [12].
Blue Native PAGE (BN-PAGE) for Membrane Protein Complexes
A specialized form of Native PAGE, Blue Native PAGE (BN-PAGE), uses Coomassie dye to confer charge to protein complexes without causing significant denaturation. This is particularly useful for the analysis of large membrane protein complexes, which are difficult to study by other means [6]. The complexes separated by BN-PAGE can be excised, digested, and identified by LC-MS/MS, providing crucial information on the subunit composition of native complexes.
Innovative Hybrid: Native SDS-PAGE
An emerging technological innovation, termed "Native SDS-PAGE," aims to bridge the gap between the high resolution of SDS-PAGE and the functional preservation of Native PAGE. This patented buffer system allows researchers to retain the functional behavior of proteins without sacrificing resolution. Proteins separated by this method maintain their native 3-dimensional conformations and functional activity, enabling direct in-gel assays of enzymatic activity, binding studies, and subsequent isolation for mass spectral identification [72]. This robust method works with many precast gels already on the market.
Integrated Workflows and Complementary Use
The true power of integrating PAGE with MS is revealed when orthogonal techniques are combined. A study comparing common gel-based fractionation methods found that while 1-D SDS-PAGE and isoelectric focusing (IEF) each yielded the highest number of protein identifications, all techniques provided complementary results [70]. This suggests that a combination of orthogonal separation methods, such as SDS-PAGE and IEF, offers the best strategy for improved profiling sensitivity without a drastic decrease in throughput [70].
Workflow for Maximum Proteome Coverage
The following diagram illustrates an integrated structural proteomics workflow that leverages the complementary strengths of SDS-PAGE and Native PAGE to gain comprehensive protein information.
SDS-PAGE and Native PAGE are not competing techniques but rather complementary pillars in the analytical pipeline of modern proteomics. SDS-PAGE, through the GeLC-MS/MS and PEPPI-MS workflows, provides an unparalleled path for the sensitive identification of proteins and their proteoforms. In contrast, Native PAGE, including BN-PAGE, is indispensable for interrogating the functional world of native protein complexes and interactions via native MS and XL-MS. The ongoing innovation in gel-based methodologies, such as Native SDS-PAGE, promises to further blur the lines between resolution and functionality. For researchers aiming to achieve an in-depth structural and functional understanding of the proteome, the strategic integration of these electrophoretic techniques with advanced mass spectrometry is not just beneficial—it is essential.
Polyacrylamide Gel Electrophoresis (PAGE) is a foundational technique in biochemistry and molecular biology laboratories for separating protein mixtures [5]. The method relies on an electric field to move charged protein molecules through a porous polyacrylamide gel matrix, which acts as a molecular sieve [12]. The choice between its two primary forms—SDS-PAGE and Native PAGE—is critical and hinges on the specific research objectives, as each technique provides distinct information about protein characteristics [5].
SDS-PAGE (Sodium Dodecyl Sulfate-PAGE) denatures proteins into their primary polypeptide components, allowing separation based almost exclusively on molecular weight [26] [12]. In contrast, Native PAGE maintains proteins in their native, folded conformation, enabling separation influenced by the molecule's intrinsic charge, size, and three-dimensional shape [5] [12]. This fundamental distinction dictates their applications, advantages, and limitations within protein separation research.
Technical Comparison: Mechanism and Separation Basis
The divergent outcomes of SDS-PAGE and Native PAGE stem from their underlying biochemical preparation of the protein sample.
SDS-PAGE: Denaturation and Uniform Charge
In SDS-PAGE, the anionic detergent SDS binds uniformly to the polypeptide backbone in a constant weight ratio (approximately 1.4 g SDS per 1 g of protein) [12] [73]. This binding, coupled with heating and a reducing agent to break disulfide bonds, unfolds the proteins into linear chains [5] [42]. The SDS molecules effectively mask the protein's intrinsic charge, imparting a uniform negative charge density [5] [12]. Consequently, when an electric field is applied, all SDS-polypeptide complexes migrate toward the anode, and their movement through the gel matrix is determined solely by their molecular size [12].
Native PAGE: Preservation of Native Structure
Native PAGE omits denaturing agents like SDS. The protein sample is prepared in a non-denaturing buffer, preserving the protein's higher-order structure (secondary, tertiary, and quaternary), biological activity, and interactions with cofactors or other subunits [5] [12]. During electrophoresis, a protein's migration depends on its net charge at the running buffer's pH, its size (molecular weight), and its shape (hydrodynamic size) [26] [74]. A tightly folded large protein may migrate differently than a smaller but more loosely structured protein [26].
Table 1: Core Principles and Methodological Differences
| Feature | SDS-PAGE | Native PAGE |
|---|---|---|
| Protein State | Denatured and linearized [5] | Native, folded structure [5] |
| Charge | Uniform negative charge from bound SDS [12] | Intrinsic charge based on protein's pI and buffer pH [12] |
| Separation Basis | Primarily molecular mass of polypeptides [12] | Combined effect of charge, size, and shape [12] |
| Multimeric Complexes | Disassembled into subunits [5] | Often preserved [12] |
| Key Reagents | SDS, reducing agent (e.g., β-mercaptoethanol) [42] | Non-ionic detergents, Coomassie G-250 (in some systems) [74] |
Diagram 1: SDS-PAGE and Native PAGE Experimental Workflows
Advantages and Limitations: A Detailed Breakdown
The choice between these techniques represents a trade-off between high-resolution mass analysis and the preservation of functional protein information.
Advantages of SDS-PAGE
- High Resolution and Simplicity for Mass Determination: SDS-PAGE provides high-resolution separation of polypeptides by molecular weight, making it a robust method for determining subunit mass and assessing sample purity [75] [12].
- Excellent Reproducibility: The highly standardized and denaturing nature of the procedure minimizes variability caused by protein shape or aggregation, ensuring consistent results across experiments [75].
- Broad Applicability for Downstream Analysis: It is the standard precursor for western blotting, as the denatured, linearized proteins transfer efficiently to membranes and are readily recognized by antibodies [5] [26]. It is also widely used in proteomics for mass spectrometry sample preparation [5].
Limitations of SDS-PAGE
- Loss of Functional and Structural Data: The primary limitation of SDS-PAGE is protein denaturation, which destroys enzymatic activity, disrupts protein-protein interactions, and strips away non-covalently bound cofactors, including metal ions [5] [75] [7].
- Inability to Resolve Non-Mass Differences: It cannot differentiate between proteins of identical molecular weight [75]. Furthermore, highly glycosylated or charged proteins may exhibit anomalous migration.
- Restricted View of Native Complexes: By dissociating multi-subunit complexes, it provides information only on the constituent polypeptides, not the functional oligomeric assembly [5].
Advantages of Native PAGE
- Preservation of Native Structure and Function: This is the most significant advantage. Proteins separated by Native PAGE often retain their biological activity, allowing for subsequent functional assays, such as activity stains for enzymes [5] [12].
- Analysis of Protein Complexes and Interactions: Native PAGE can separate and identify stable protein-protein interactions, oligomeric states, and quaternary structures under conditions that mimic the cellular environment [5].
- Study of Labile Assemblies: Certain variants, like Clear-Native PAGE (CN-PAGE), are exceptionally mild and can retain labile supramolecular assemblies of membrane proteins that dissociate under other conditions [76].
Limitations of Native PAGE
- Complex Data Interpretation: Migration depends on multiple factors (charge, size, shape), complicating data interpretation and making native mass estimation less straightforward than in SDS-PAGE [5] [76].
- Risk of In-Gel Aggregation or Precipitation: Without denaturants, proteins may aggregate during electrophoresis, particularly hydrophobic membrane proteins, leading to poor resolution or smearing [74].
- Buffer System Dependency: The choice of buffer and pH is critical, as it must maintain protein stability and solubility. A protein's isoelectric point (pI) can cause it to precipitate if the buffer pH is not carefully selected [74].
Table 2: Comparative Advantages and Limitations for Research
| Aspect | SDS-PAGE | Native PAGE |
|---|---|---|
| Molecular Weight Determination | Excellent (subunit mass) [12] | Poor/Indirect (native mass estimation is complex) [76] |
| Preservation of Activity | No (proteins denatured) [5] | Yes (activity often retained) [12] |
| Analysis of Complexes | No (complexes dissociated) [5] | Yes (oligomeric states preserved) [12] |
| Resolution | High (based on mass) [75] | Variable (depends on protein and system) [76] |
| Ease of Interpretation | Straightforward (mass calibration) [12] | Complex (multiple migration factors) [5] |
| Downstream Western Blot | Ideal [26] | Possible (with specific protocols) [74] |
Experimental Protocols and Methodologies
A typical discontinuous SDS-PAGE protocol involves two gel layers and specific buffers:
- Gel Preparation: A resolving gel (e.g., pH 8.8, 8-12% acrylamide) is overlaid with a stacking gel (pH 6.8, 4-5% acrylamide). The discontinuous buffer system concentrates samples into sharp bands before they enter the resolving gel, enhancing resolution [77].
- Sample Preparation: Protein samples are mixed with an SDS-containing sample buffer (often Laemmli buffer), which includes SDS, a reducing agent (β-mercaptoethanol or DTT), glycerol, and a tracking dye. The mixture is heated at 70-100°C for 5-10 minutes to ensure complete denaturation [26] [12].
- Electrophoresis: Samples are loaded onto the gel and electrophoresis is performed using a running buffer containing Tris, glycine, and SDS (e.g., Tris-Glycine-SDS) at a constant voltage (e.g., 150-200 V) until the dye front reaches the bottom.
Native PAGE protocols vary based on the gel chemistry selected (Tris-Glycine, Tris-Acetate, or Bis-Tris).
- Gel and Buffer Selection:
- Tris-Glycine Systems: Traditional, operate at pH 8.3-9.5. Suitable for smaller proteins (20-500 kDa) where maintaining the native charge is desired [74].
- Bis-Tris Systems (e.g., NativePAGE): Operate at a near-neutral pH (~7.5). Use Coomassie G-250 dye in the cathode buffer to confer a negative charge on all proteins, including those with basic pI, and to prevent aggregation of membrane proteins. This provides separation by molecular weight regardless of intrinsic charge [74].
- Sample Preparation: Samples are mixed with a non-denaturing sample buffer (e.g., Tris-Glycine Native Sample Buffer) that lacks SDS and reducing agents. Glycerol is included to aid in loading.
- Electrophoresis: Pre-cast gels are run in a native running buffer without SDS. For Bis-Tris systems, the cathode buffer contains Coomassie G-250 additive. The procedure is typically performed at 4°C or in a cold room to maintain protein stability.
Advanced Methodology: Native SDS-PAGE (NSDS-PAGE)
A modified technique, Native SDS-PAGE (NSDS-PAGE), has been developed to bridge the gap between the two methods. It aims to provide the high resolution of SDS-PAGE while retaining some native functional properties, such as bound metal ions and enzymatic activity [7].
Key Protocol Modifications from Standard SDS-PAGE [7]:
- No Heating: The heating step during sample preparation is omitted.
- Reduced SDS: The SDS concentration in the running buffer is drastically reduced (e.g., to 0.0375%).
- No EDTA: Chelating agents are removed from buffers to preserve metal-protein interactions.
- Results: This milder treatment allowed for the retention of Zn²⁺ in proteomic samples (increased from 26% to 98%) and enabled seven out of nine model enzymes to retain activity post-electrophoresis [7].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for PAGE
| Reagent / Material | Function in SDS-PAGE | Function in Native PAGE |
|---|---|---|
| Sodium Dodecyl Sulfate (SDS) | Denatures proteins and imparts uniform negative charge [12] | Not used |
| Reducing Agent (e.g., DTT) | Breaks disulfide bonds for complete denaturation [42] | Not used (would disrupt native structure) |
| Coomassie G-250 Dye | Not typically used in sample buffer | Charge-shift molecule; binds proteins to confer negative charge without denaturation [74] |
| Polyacrylamide Gel | Molecular sieve; pore size determines separation range [12] | Molecular sieve; pore size determines separation range [12] |
| Tris-based Buffers | Maintains pH for electrophoresis (e.g., Tris-Glycine) [12] | Maintains pH; different systems (Tris-Glycine, Bis-Tris) available [74] |
| Non-Ionic Detergent | Not typically used | Added to samples to solubilize membrane proteins without denaturing them [74] |
Application Contexts in Protein Research and Drug Development
The strategic choice between SDS-PAGE and Native PAGE is dictated by the research question.
SDS-PAGE is ideal for:
Native PAGE is ideal for:
- Studying protein-protein interactions and oligomeric states [5].
- Detecting and quantifying functional enzymes in complex mixtures via in-gel activity assays [5] [7].
- Analyzing labile protein complexes, particularly membrane-bound complexes like mitochondrial respiratory chains [76].
- Characterizing the native state of a therapeutic protein or antibody.
Diagram 2: Decision Workflow for Selecting a PAGE Method
SDS-PAGE and Native PAGE are not competing but complementary techniques in the protein researcher's arsenal [5]. SDS-PAGE remains the undisputed method for analytical separation based on polypeptide mass, offering high resolution and reproducibility for a wide array of downstream applications. Conversely, Native PAGE is the definitive choice for any investigation requiring insight into a protein's functional, native state, including its oligomeric architecture, interactive partners, and catalytic competence.
The emergence of hybrid techniques like NSDS-PAGE [7] underscores a continuing innovation in the field, aiming to merge the benefits of both approaches. For researchers in drug development and basic science, a deep understanding of the principles, advantages, and limitations of each method is fundamental to designing robust experiments, correctly interpreting data, and ultimately making an informed choice that aligns with their strategic research goals.
Conclusion
SDS-PAGE and Native PAGE are powerful, complementary techniques that serve distinct purposes in protein analysis. SDS-PAGE is unparalleled for determining molecular weight and analyzing denatured proteins, while Native PAGE is essential for studying protein complexes, interactions, and function in their native state. The choice between them should be guided by specific research goals—whether prioritizing structural information or functional activity. As proteomics and drug discovery advance, the orthogonal use of these techniques, sometimes combined with mass spectrometry and other analytical methods, will continue to be fundamental for comprehensive protein characterization, enabling deeper insights into biological systems and therapeutic development.
References
- 1. Polyacrylamide Gel Electrophoresis, How It Works, ... [https://www.technologynetworks.com/analysis/articles/polyacrylamide-gel-electrophoresis-how-it-works-technique-variants-and-its-applications-359100]
- 2. The principle and method of polyacrylamide gel ... [https://www.mblbio.com/bio/g/support/method/sds-page.html]
- 3. SDS-PAGE guide: Sample preparation to analysis [https://www.abcam.com/en-us/knowledge-center/western-blot/sds-page-guide]
- 4. SDS-PAGE [https://en.wikipedia.org/wiki/SDS-PAGE]
- 5. How Is Native PAGE Different from SDS-PAGE? [https://synapse.patsnap.com/article/how-is-native-page-different-from-sds-page]
- 6. Differences between SDS PAGE and Native PAGE [https://www.geeksforgeeks.org/biology/differences-between-sds-page-and-native-page/]
- 7. Native SDS-PAGE: High Resolution Electrophoretic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4517606/]
- 8. Utilization of gel electrophoreses for the quantitative ... [https://www.sciencedirect.com/science/article/pii/S1319016416300767]
- 9. MatGel: A MATLAB program for quantitative analysis of 2D ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9703371/]
- 10. Interpret SDS-PAGE vs Native-PAGE Results [https://www.letstalkacademy.com/sds-page-vs-native-page-protein-dimer/]
- 11. Deciphering food proteins: The applications of SDS-PAGE ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429225003025]
- 12. Overview of Protein Electrophoresis [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-electrophoresis.html]
- 13. Protein Electrophoresis Using SDS-PAGE: A Detailed ... [https://www.goldbio.com/blogs/articles/protein-electrophoresis-sds-page-overview?srsltid=AfmBOoqGDO-KVD-APnqu0FUeqTATEdbWlDYX2wbHoGoznSk1b-PngR6v]
- 14. Tips for Optimal SDS-PAGE Separation [https://www.rockland.com/resources/tips-for-optimal-sds-page-separation/?srsltid=AfmBOooAWHx5SWMcfjSqhFhgUOt05yRki-CXjKtYBC5VPDbHlubLh1Fb]
- 15. Constant Current or Voltage in SDS-PAGE [https://bitesizebio.com/51744/constant-current-or-voltage-in-sds-page/]
- 16. Visualization of proteins in SDS PAGE gels [https://www.qiagen.com/us/knowledge-and-support/knowledge-hub/bench-guide/protein/protein-analysis/visualization-of-proteins-in-sds-page-gels]
- 17. What is the difference between PAGE and SDS-PAGE? [https://www.aatbio.com/resources/faq-frequently-asked-questions/What-is-the-difference-between-PAGE-and-SDS-PAGE]
- 18. What is SDS-PAGE? Uses, How It Works & Top Companies ... [https://www.linkedin.com/pulse/what-sds-page-uses-how-works-top-companies-2025-clearsedge-research-kztwf]
- 19. SDS PAGE vs Native PAGE [https://byjus.com/biology/difference-between-sds-page-and-native-page/]
- 20. Recent Advances in Mass Spectrometry-based Separation ... [https://link.springer.com/article/10.1007/s40242-025-4242-6]
- 21. SDS-PAGE - 蛋白质电泳 - 欣伯玉生物 [https://www.x-biotech.com/SDS-PAGE?article_id=34]
- 22. 同样是蛋白电泳,SDS-PAGE和PAGE到底有啥不一样? [https://www.uptbio.com/news/view/1270.html]
- 23. SDS-PAGE电泳的基础原理和实验步骤 [https://www.detaibio.com/topics/sds-page-technology.html]
- 24. 视频: 使用SDS-PAGE 分离蛋白质:程序和应用 [https://www.jove.com/cn/v/5058/separating-protein-with-sds-page-procedure-and-applications]
- 25. Should I Use A Native Or Denaturing Gel? [https://info.gbiosciences.com/blog/should-i-use-a-native-or-denaturing-gel]
- 26. Western blotting guide: Part 2, Protein separation by SDS- ... [https://www.jacksonimmuno.com/secondary-antibody-resource/technical-tips/western-blotting-guide-part-2/]
- 27. Validation of blue- and clear-native polyacrylamide gel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12445495/]
- 28. SDS-PAGE and Coomassie staining - QB3 Berkeley [https://qb3.berkeley.edu/education/lab-fundamentals-bootcamp/manual/proteins/protein-gels/]
- 29. A screening method for binding synthetic metallo ... [https://www.sciencedirect.com/science/article/pii/S0003269722002445]
- 30. Protein Electrophoresis Using SDS-PAGE: A Detailed ... [https://www.goldbio.com/blogs/articles/protein-electrophoresis-sds-page-overview?srsltid=AfmBOoph4TIu6hNZsamXv7GKbBoT6u7EOSdBGZEmzrZ6UhoF1ciReu3Y]
- 31. Western Blot: The Complete Guide [https://www.antibodies.com/applications/western-blotting]
- 32. Western Blot SDS-PAGE [https://www.novusbio.com/support/support-by-application/western-blot-sds-page?srsltid=AfmBOorzhITWeS1tiIAXZMjweQw_lJYgvdvq19pYlzFrD1Bi6MVFCCI8]
- 33. Western Blotting: Products, Protocols, & Applications [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-western-blotting.html]
- 34. A Native PAGE Assay for the Biochemical Characterization ... [https://bio-protocol.org/en/bpdetail?id=4266&type=0]
- 35. Native mass spectrometry of membrane proteins reconstituted ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12560048/]
- 36. Native Top–Down Analysis of Membrane Protein ... [https://www.sciencedirect.com/science/article/pii/S153594762500091X]
- 37. Blue native electrophoresis protocol [https://www.abcam.com/en-us/technical-resources/protocols/blue-native-electrophoresis]
- 38. Native electrophoresis in cell proteomic: BN-PAGE and CN ... [https://link.springer.com/article/10.1134/S1990519X11030126]
- 39. Principles of Blue Native-PAGE [https://blog.benchsci.com/principles-of-blue-native-page]
- 40. Two-dimensional Blue Native/SDS Gel Electrophoresis of ... [https://www.sciencedirect.com/science/article/pii/S1535947620334228]
- 41. Gel electrophoresis of proteins - Wikipedia [https://en.wikipedia.org/wiki/Gel_electrophoresis_of_proteins]
- 42. SDS-PAGE Analysis of proteins: Principles, methodology, ... [https://deepscienceresearch.com/dsr/catalog/book/107/chapter/407]
- 43. Advantages and Disadvantages of SDS-PAGE for Protein ... [https://www.mtoz-biolabs.com/advantages-and-disadvantages-of-sds-page-for-protein-separation.html]
- 44. Troubleshooting SDS-PAGE Gel Running Issues [https://www.goldbio.com/blogs/articles/troubleshooting-sds-page-gel-running-issues?srsltid=AfmBOoqbWB_Od4pbcuPdgqyNTmOt2Ph85ZaE9ZYMvnj3n3rwcjlShu8g]
- 45. Troubleshooting Sodium Dodecyl Sulfate- Polyacrylamide ... [https://www.hycultbiotech.com/sds-page/troubleshooting/]
- 46. High-resolution native electrophoresis in-gel activity assay ... [https://www.nature.com/articles/s41598-025-24684-3]
- 47. Overview of Protein Electrophoresis [https://www.thermofisher.com/ru/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-electrophoresis.html]
- 48. Native Agarose Gels and Contact Blotting as Means to ... [https://www.mdpi.com/2306-5354/10/10/1111]
- 49. Rapid baseline hump-free analysis of therapeutic proteins ... [https://www.sciencedirect.com/science/article/pii/S0003267025005410]
- 50. Effect of the operational parameters on the electromigration ... [https://www.sciencedirect.com/science/article/pii/S0039914025000876]
- 51. Overview of Protein Electrophoresis [https://www.thermofisher.com/de/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-electrophoresis.html]
- 52. Improving SDS-PAGE method for monoclonal antibodies [https://pubmed.ncbi.nlm.nih.gov/33596474/]
- 53. A comparison of Tris-glycine and Tris-tricine buffers for the ... [https://pubmed.ncbi.nlm.nih.gov/21818850/]
- 54. Improving SDS-PAGE method for monoclonal antibodies [https://www.sciencedirect.com/science/article/abs/pii/S1046592821000280]
- 55. Native PAGE Gels | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-gel-electrophoresis/protein-gels/specialized-protein-gels/nativepage-bis-tris-gels.html]
- 56. Common artifacts and mistakes made in electrophoresis [https://pmc.ncbi.nlm.nih.gov/articles/PMC7295095/]
- 57. Overview of Protein Electrophoresis [https://www.thermofisher.com/tr/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-electrophoresis.html]
- 58. Preparing High-Quality Samples for Electrophoresis: Best ... [https://www.labx.com/resources/preparing-high-quality-samples-for-electrophoresis-best-practices/5550]
- 59. Troubleshooting SDS-PAGE Sample Preparation Issues [https://www.goldbio.com/blogs/articles/troubleshooting-sds-page-sample-preparation-issues?srsltid=AfmBOooo5RBwY6E4UVi1P-3u-mTfgQNG9kgcEYC5QHNS0GnNLan3o0lR]
- 60. SDS-PAGE: A Cornerstone of Protein Analysis in Modern ... [https://www.metwarebio.com/sds-page-protein-analysis/]
- 61. Comparing SDS-PAGE and CE-SDS for Antibody Purity ... [https://www.bioprocessintl.com/qa-qc/comparing-sds-page-and-ce-sds-for-antibody-purity-analysis]
- 62. What's the Difference Between SDS-PAGE and Native ... [https://synapse.patsnap.com/article/whatE28099s-the-difference-between-sds-page-and-native-page]
- 63. Quantification of Folded and Misfolded Proinsulin Forms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12152102/]
- 64. Tracking Dye Used in SDS PAGE - Let's Talk Academy [https://www.letstalkacademy.com/tracking-dye-used-in-sds-page/]
- 65. Native-PAGE [http://www.assay-protocol.com/molecular-biology/electrophoresis/native-page.html]
- 66. SDS-PAGE Protocol [https://www.rockland.com/resources/sds-page-protocol/?srsltid=AfmBOop0WC2MKOPwF_OqCfk8EISn6Y9GNyWmBRp7h9tJGZGTggWLHdZO]
- 67. Protein Gel Staining: Coomassie, Silver, Fluorescent & More [https://www.creative-proteomics.com/resource/protein-gel-staining-methods.htm]
- 68. Comparison of the performance of 1D SDS-PAGE with ... [https://www.sciencedirect.com/science/article/abs/pii/S1570023218312546]
- 69. SDS-PAGE Experiment Protocol [https://microbiosci.creative-biogene.com/sds-page-experiment-protocol.html]
- 70. Comparison of in-gel protein separation techniques ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4234072/]
- 71. In-depth structural proteomics integrating mass ... [https://www.frontiersin.org/journals/analytical-science/articles/10.3389/frans.2022.1107183/full]
- 72. OTT1313 [https://uwmrf.org/technology/ott1313/]
- 73. SDS protein interactions [https://www.sciencedirect.com/science/article/abs/pii/S0301462225001462]
- 74. Gels | Thermo Fisher Scientific - IE Native PAGE [https://www.thermofisher.com/ie/en/home/life-science/protein-biology/protein-gel-electrophoresis/protein-gels/specialized-protein-gels/nativepage-bis-tris-gels.html]
- 75. Advantages and Disadvantages of SDS-PAGE Based ... [https://www.mtoz-biolabs.com/advantages-and-disadvantages-of-sds-page-based-protein-separation.html]
- 76. and Advantages of clear- limitations native PAGE [https://pubmed.ncbi.nlm.nih.gov/16220535/]
- 77. Advantage of discontinuous buffer system in SDS and native ... [https://www.letstalkacademy.com/advantage-of-discontinuous-buffer-system-in-sds-and-native-page/]