Neurochemical Orchestration of Sleep: Biochemical Pathways, Therapeutic Targets, and Drug Development
This article provides a comprehensive synthesis of the biochemical mechanisms governing sleep-wake regulation and their disruption in sleep disorders.
Neurochemical Orchestration of Sleep: Biochemical Pathways, Therapeutic Targets, and Drug Development
Abstract
This article provides a comprehensive synthesis of the biochemical mechanisms governing sleep-wake regulation and their disruption in sleep disorders. Targeting researchers and drug development professionals, it details the foundational roles of neurotransmitters like GABA, adenosine, and serotonin, and hormones such as melatonin. It further explores methodological approaches for investigating sleep biochemistry, analyzes the mechanistic basis of common sleep pathologies, and evaluates current and emerging therapeutic strategies. The review aims to bridge fundamental neurochemistry with clinical application, highlighting promising targets for novel pharmacotherapies in sleep medicine.
Core Neurochemical Systems and Hormonal Regulators of the Sleep-Wake Cycle
The regulation of sleep and wakefulness is a complex neurological process governed by the precise interplay of inhibitory and excitatory neurotransmitter systems. The balanced activity of GABA (gamma-aminobutyric acid), the principal inhibitory neurotransmitter, and glutamate, the major excitatory neurotransmitter, forms the fundamental basis of sleep-wake control [1] [2]. Acetylcholine (ACh) serves a crucial neuromodulatory role, promoting cortical activation during both wakefulness and REM sleep [3] [4]. Disruptions in these intricate systems are implicated in the pathogenesis of various sleep disorders, including insomnia, obstructive sleep apnea, and REM sleep behavior disorder [5] [1]. This whitepaper provides an in-depth technical analysis of the mechanisms by which GABA, glutamate, and acetylcholine regulate sleep states, supported by recent experimental findings and quantitative data, framed within the context of sleep disorder biochemistry and therapeutic development.
Neurotransmitter Systems and Sleep-Wake Regulation
GABA: The Primary Inhibitory System
Gamma-aminobutyric acid (GABA) is the main inhibitory neurotransmitter in the central nervous system and plays a fundamental role in sleep initiation and maintenance. GABAergic neurotransmission promotes sleep by inhibiting wake-promoting brain regions [2].
- Receptors and Mechanisms: GABA exerts its effects through ionotropic GABAA and metabotropic GABAB receptors. Activation of GABAA receptors, which form chloride channels, results in fast hyperpolarization and functional inhibition of postsynaptic neurons. The GABAA receptor is a heteropentamer, with the most common isoform composed of α1, β2, and γ2 subunits, and is a primary target for sedative-hypnotic drugs like benzodiazepines, barbiturates, and z-drugs [1] [2]. GABAB receptors act indirectly through G-proteins, mediating a slower, prolonged inhibitory response [6].
- Therapeutic Application: Most clinically used hypnotics are based on enhancing GABAergic inhibition. Three generations of hypnotics—barbiturates, benzodiazepines, and imidazopyridines/cyclopyrrolones (Z-drugs)—primarily act as positive allosteric modulators of the GABAA receptor, thereby decreasing waking and increasing slow-wave sleep [6] [4]. Emerging research also implicates neuroinflammation and oxidative stress in insomnia pathophysiology, which GABAergic therapies may help ameliorate [2].
Glutamate: The Primary Excitatory System
Glutamate is the major excitatory neurotransmitter in the CNS and is essential for maintaining wakefulness and regulating synaptic plasticity during sleep [7] [8].
- Receptors and Signaling Pathways: Glutamate acts through ionotropic (iGlu) and metabotropic (mGlu) receptors. Ionotropic receptors (NMDA, AMPA, kainate) are ligand-gated ion channels that mediate fast excitatory transmission. Metabotropic receptors (mGluR1-8) are G-protein coupled receptors that modulate slower, neuromodulatory processes [8]. The NMDA receptor, particularly its essential NR1 subunit, is critically involved in sleep regulation [7].
- Pathophysiology in Sleep Disorders: Recent clinical evidence indicates glutamatergic dysregulation in insomnia. A 2024 study found significantly elevated serum glutamate levels and upregulated mRNA expression of the NMDA receptor NR1 subunit in the peripheral blood of patients with Insomnia Disorder (ID) compared to healthy controls [7]. This hyperglutamatergic state may contribute to the hyperarousal characteristic of chronic insomnia. The glutamate-glutamine cycle, which involves astrocytes, is crucial for preventing glutamate excitotoxicity and maintaining excitatory homeostasis [1].
Acetylcholine: A Key Neuromodulator
Acetylcholine (ACh) is a key neuromodulator of the sleep-wake cycle, with a distinct activity profile that promotes cortical activation [3] [4].
- Dual Role in Wakefulness and REM Sleep: Cholinergic neurons in the basal forebrain (BF) and brainstem (e.g., the pedunculopontine nucleus) are highly active during both wakefulness and REM sleep, driving cortical desynchronization and EEG activation seen in these states. During NREM sleep, the firing rates of these neurons are significantly reduced [3] [4].
- Receptors and Functional Connectivity: ACh acts through muscarinic (mAChR) and nicotinic (nAChR) receptors. A key mechanism by which ACh influences brain state is by enhancing neuronal excitation and reducing long-range cortico-cortical interactions, thereby promoting a desynchronized EEG pattern [3]. Advanced neuroimaging studies show that functional connectivity between the basal forebrain and the cortex decreases during deeper NREM sleep stages (N3), reflecting reduced cholinergic neuromodulation [3].
Quantitative Data and Experimental Findings
Recent clinical and preclinical studies provide quantitative insights into neurotransmitter dysregulation in sleep disorders.
Table 1: Serum Glutamate and NMDA Receptor NR1 Subunit Expression in Insomnia Disorder (ID)
| Parameter | ID Patient Group | Healthy Control Group | Statistical Significance | Assessment Method |
|---|---|---|---|---|
| Serum Glutamate | Significantly Elevated | Lower | p < 0.05 | Enzyme-linked immunosorbent assay (ELISA) |
| NR1 Subunit mRNA | Significantly Upregulated | Lower | p < 0.05 | Real-time PCR |
| NR1 Diagnostic Power | AUC: 0.758 | - | Sensitivity: 73.3%, Specificity: 76.7% | Receiver Operating Characteristic (ROC) Analysis |
| Glutamine & GAD | No Significant Difference | No Significant Difference | p > 0.05 | ELISA |
A 2024 clinical study demonstrated that the mRNA expression levels of the NMDA receptor NR1 subunit could serve as a potential biomarker for ID, with a diagnostic accuracy shown by an area under the curve (AUC) of 0.758 [7].
Table 2: Changes in Functional Connectivity (FC) of Neuromodulatory Nuclei During NREM Sleep
| Sleep Stage | Basal Forebrain (BF) FC | Locus Coeruleus (LC) FC | Brain-Wide Network State |
|---|---|---|---|
| Wakefulness (W) | Baseline | Baseline | Balanced |
| N1 | No significant change | No significant change | More Integrated |
| N2 | No significant change | Significant Decrease | Transitional |
| N3 | Significant Decrease | Significant Decrease | More Segregated |
A 2025 fMRI study analyzing NREM sleep substages found that the functional connectivity of key neuromodulatory nuclei with the cortex changes distinctly as sleep deepens. The study also reported a global shift in brain network organization, moving towards a more integrated state in light sleep (N1) and a more segregated state in deep sleep (N3) [3].
Experimental Models and Methodologies
Preclinical Animal Models
Research on sleep disorders utilizes several well-established animal models to investigate the neurobiological consequences of sleep disruption:
- Total Sleep Deprivation (TSD) and Selective Sleep Deprivation (SSD): These models are used to study pathological changes in experimental animals after REM sleep deprivation or overall sleep loss [5].
- Forced Locomotion Technique (FLT): This method involves forcing experimental animals to move continuously within a powered device, such as a rotary cylinder or horizontal turntable, to achieve sleep deprivation [5].
Clinical and Translational Research Protocols
Table 3: Key Methodologies for Investigating Neurotransmitter Systems in Human Sleep
| Methodology | Application | Key Outputs |
|---|---|---|
| Enzyme-Linked Immunosorbent Assay (ELISA) | Quantification of serum concentrations of glutamate, glutamine (Gln), and glutamic acid decarboxylase (GAD) [7]. | Concentration levels (e.g., in μg/mL or pg/mL). |
| Real-time Polymerase Chain Reaction (qPCR) | Detection of mRNA expression levels of receptor subunits (e.g., NMDAR NR1) in peripheral blood [7]. | Cycle threshold (Ct) values, fold-change in expression. |
| Functional Magnetic Resonance Imaging (fMRI) | Assessment of changes in functional connectivity (FC) between brain regions (e.g., BF, LC) across sleep stages [3]. | Pearson correlation matrices, network integration/segregation metrics. |
| Polysomnography (PSG) | Gold-standard for objective sleep staging and ruling out other sleep-wake disorders like sleep apnea [7]. | Sleep stages (W, N1, N2, N3, REM), sleep latency, arousal index. |
Integrated Signaling Pathways and Neurotransmitter Interactions
The transition between sleep and wake states emerges from dynamic interactions between multiple neurotransmitter systems. The following diagram illustrates the key pathways and nuclei involved in regulating the sleep-wake cycle through GABA, glutamate, and acetylcholine.
The flip-flop switch model between sleep and wake involves mutual inhibition between wake-promoting monoaminergic nuclei (e.g., Locus Coeruleus releasing Noradrenaline) and sleep-promoting GABAergic neurons in the ventrolateral preoptic nucleus (VLPO). Acetylcholine from the basal forebrain and brainstem promotes both wakefulness and REM sleep, while glutamate provides widespread excitatory drive for arousal. The state of this network is reflected in measurable changes in functional connectivity, such as decreased LC and BF connectivity with the cortex during deep NREM sleep [3] [4].
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Reagents and Materials for Sleep Neurotransmitter Research
| Reagent / Material | Primary Function | Example Application |
|---|---|---|
| Selective Receptor Agonists/Antagonists | To probe the function of specific receptor subtypes (e.g., mGluR2/3 agonists, GABAA α1-subunit selective ligands) in sleep architecture [8]. | Pharmacological dissection of sleep-wake circuits in preclinical models. |
| ELISA Kits | To quantitatively measure concentrations of neurotransmitters (Glu, Gln) and related enzymes (GAD) in serum, CSF, or tissue homogenates [7]. | Clinical biomarker discovery and validation in patient cohorts. |
| qPCR Assays | To detect and quantify mRNA expression levels of receptor subunits and synthetic enzymes in patient blood or post-mortem brain tissue [7]. | Investigation of transcriptional regulation in sleep disorders. |
| Radioligands (for PET) | To enable in vivo imaging and quantification of receptor availability and density in the human brain. | Translational studies linking receptor changes to sleep phenotype. |
The intricate balance and temporal coordination between GABAergic, glutamatergic, and cholinergic systems form the neurochemical cornerstone of sleep-wake regulation. Dysregulation within these systems is a key pathophysiological feature of sleep disorders. The identification of elevated peripheral glutamate and NMDA NR1 subunit expression in insomnia points toward excitatory-inhibitory imbalance as a promising therapeutic target [7]. Future drug development is moving beyond non-selective GABAergic drugs toward more precise targets, including specific GABA receptor subunits, metabotropic glutamate receptors (e.g., mGluR2, mGluR5), and cholinergic receptors, aiming to correct underlying imbalances with fewer side effects [2] [8]. Integrating multimodal methodologies—from molecular biology and advanced neuroimaging to computational modeling—will continue to deepen our understanding of these neurotransmitter systems, ultimately paving the way for novel, targeted therapies in sleep medicine.
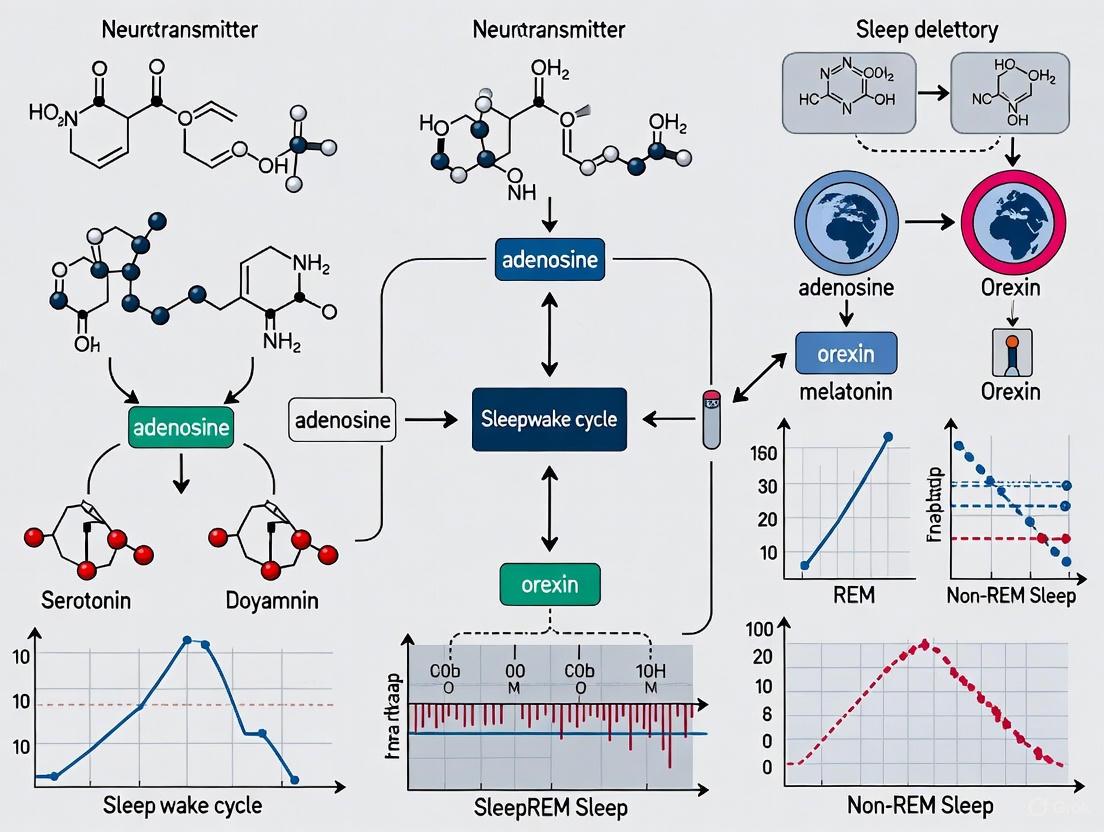
Sleep is a fundamental, recurring state of life essential for normal physiological and cognitive function. Long-term sleep disruption can lead to physical and mental fatigue, inattention, memory loss, and anxiety, imposing a significant public health and economic burden worldwide [9]. The sleep-wake cycle is regulated by a complex neural network involving numerous nuclei and neurotransmitters in the brain. Within this system, adenosine (AD) has emerged as a crucial endogenous sleep-regulatory substance that drives sleep homeostasis through specific receptor-mediated mechanisms [9]. This whitepaper examines the role of adenosine as an endogenous sleep regulator, focusing on its homeostatic functions and the receptor mechanisms through which it exerts its effects, providing researchers and drug development professionals with a comprehensive technical overview of this critical regulatory system.
Adenosine is a purine nucleoside that functions as a ubiquitous endogenous cell signal transducer and regulator in the central nervous system (CNS) [10]. Its concentrations in the brain increase during prolonged wakefulness and decrease during sleep, positioning it as a key mediator of sleep homeostasis. The sleep-promoting effects of adenosine are primarily mediated through four G protein-coupled receptors (GPCRs): A1, A2A, A2B, and A3, collectively known as P1 receptors [10]. This review synthesizes current research on the functions and mechanisms of adenosine and its receptors in sleep regulation, with implications for understanding insomnia, hypersomnia, and other sleep disorders.
Adenosine Receptor Subtypes and Signaling Mechanisms
Adenosine exerts its physiological effects by activating four principal receptor subtypes, each with distinct signaling pathways, cellular distributions, and functional roles in sleep-wake regulation.
Table 1: Adenosine Receptor Subtypes and Their Characteristics
| Receptor Subtype | G-Protein Coupling | Primary Signaling Pathway | CNS Distribution | Overall Effect on Neuronal Activity |
|---|---|---|---|---|
| A1 Receptor | Gᵢ/G₀ | Inhibits adenylate cyclase, reduces cAMP | Widespread: cortex, hippocampus, cerebellum | Inhibitory |
| A2A Receptor | Gₛ | Stimulates adenylate cyclase, increases cAMP | Striatum, nucleus accumbens, olfactory tubercle | Excitatory (in indirect pathway neurons) |
| A2B Receptor | Gₛ | Stimulates adenylate cyclase, increases cAMP | Low levels throughout CNS, glial cells | Excitatory |
| A3 Receptor | Gᵢ/G₀ | Inhibits adenylate cyclase, reduces cAMP | Low levels in CNS, predominantly peripheral | Inhibitory |
The A1 receptor (A1R) is the most widely expressed adenosine receptor in the CNS, with prominent presence in the cortex, hippocampus, and cerebellum [10]. Activation of A1Rs produces predominantly inhibitory effects through Gᵢ/G₀ protein coupling, which leads to inhibition of adenylate cyclase, reduced intracellular cyclic adenosine-3,5-monophosphate (cAMP) production, and consequent decreased protein kinase A (PKA) activity [10] [11]. This signaling cascade ultimately suppresses neuronal excitability through multiple mechanisms, including inhibition of voltage-gated calcium channels, activation of potassium channels, and reduction of neurotransmitter release [9]. In the context of sleep regulation, A1R activation in wake-promoting brain regions inhibits the release of excitatory neurotransmitters, thereby promoting sleep.
The A2A receptor (A2AR) is primarily localized in the striatum, nucleus accumbens, and olfactory tubercle [10]. In contrast to A1Rs, A2ARs couple to Gₛ proteins, stimulating adenylate cyclase activity, increasing intracellular cAMP levels, and enhancing PKA activity [11]. This excitatory signaling pathway plays a particularly important role in the striatum, where A2A receptors co-localize and functionally interact with dopamine D2 receptors in indirect pathway striatal projection neurons (iSPNs) [11]. The activation of A2A receptors in these neurons promotes sleep, particularly through actions in the ventral striatal shell of the nucleus accumbens, where they can strongly promote NREM sleep.
The A2B and A3 receptors are less abundant in the CNS but contribute to adenosine signaling under certain conditions. A2B receptors have low affinity for adenosine and primarily become activated when adenosine concentrations are elevated, such as during prolonged wakefulness or metabolic stress [12]. A3 receptors also inhibit adenylate cyclase through Gᵢ/G₀ coupling but are more prominent in peripheral tissues, though emerging evidence suggests they may play a role in torpor regulation [10].
Diagram 1: Adenosine Receptor Signaling Pathways. This diagram illustrates the primary intracellular signaling mechanisms of adenosine A1 and A2A receptors, the two principal receptor subtypes involved in sleep-wake regulation.
Homeostatic Sleep Drive and Adenosine Accumulation
The sleep homeostat represents a fundamental regulatory process that tracks time spent awake and generates increasing sleep pressure that must eventually be dissipated through sleep. Adenosine serves as a key molecular substrate of the sleep homeostat, with extracellular concentrations in specific brain regions progressively increasing during wakefulness and declining during sleep [9].
Research indicates that adenosine accumulation during extended wakefulness occurs through multiple mechanisms. Metabolic activity in the brain during wakefulness leads to increased ATP consumption and consequent generation of adenosine as a breakdown product [10]. Additionally, astrocyte-derived adenosine has been identified as playing an important role in modulating sleep homeostasis and sleep loss-induced cognitive and memory deficits [9]. The basal forebrain and cortex are particularly important sites for adenosine-mediated sleep homeostasis, with microdialysis studies demonstrating region-specific fluctuations in adenosine concentrations across the sleep-wake cycle.
The homeostatic function of adenosine is evidenced by several key observations. First, local perfusion of adenosine or adenosine transport inhibitors into the basal forebrain promotes sleep, while adenosine receptor antagonists promote wakefulness [9]. Second, the progressive increase in adenosine during wakefulness correlates with increasing slow-wave activity (SWA) during subsequent NREM sleep, which is a key electrophysiological marker of sleep homeostasis. Third, astrocyte-derived adenosine in modulating sleep homeostasis and sleep loss-induced related cognitive and memory deficits plays an important role [9]. This homeostatic accumulation of adenosine and its subsequent decline during sleep represents a crucial mechanism for restoring energy balance and clearing metabolic waste products that accumulate during wakefulness.
Neural Circuits and Brain Regions in Adenosine-Mediated Sleep Regulation
Adenosine regulates sleep-wake states through coordinated actions on multiple neural systems, both by inhibiting wake-promoting centers and facilitating sleep-promoting regions.
Inhibition of Arousal Systems
Adenosine exerts a tonic inhibitory influence on multiple components of the ascending arousal system. In the basal forebrain, adenosine inhibits cholinergic neurons through A1R activation, reducing acetylcholine release in the cortex and promoting slow-wave sleep [9] [11]. Similarly, adenosine acting via A1 receptors inhibits wake-active neurons in the tuberomammillary nucleus (histaminergic), locus coeruleus (noradrenergic), and raphe nuclei (serotonergic) [9]. This broad inhibition of arousal systems contributes to the transition from wakefulness to sleep and helps maintain sleep states.
Activation of Sleep-Promoting Systems
In addition to inhibiting arousal centers, adenosine facilitates sleep through activation of sleep-active neurons. In the ventrolateral preoptic area (VLPO), adenosine excites sleep-active neurons through both A1 and A2A receptor mechanisms, further promoting sleep initiation and maintenance [9]. The nucleus accumbens represents another important site where adenosine, particularly through A2A receptors, strongly promotes NREM sleep [10]. This dual mechanism—simultaneous inhibition of wake-promoting systems and activation of sleep-promoting systems—allows adenosine to efficiently orchestrate the complex neural transitions between sleep and wake states.
Table 2: Key Brain Regions in Adenosine-Mediated Sleep Regulation
| Brain Region | Primary Receptor Involvement | Mechanism of Action | Functional Outcome |
|---|---|---|---|
| Basal Forebrain | A1 Receptor | Inhibition of cholinergic neurons | Reduced cortical ACh, promotes SWS |
| Tuberomammillary Nucleus | A1 Receptor | Inhibition of histaminergic neurons | Reduced histamine release, promotes sleep |
| Ventrolateral Preoptic Area (VLPO) | A1 and A2A Receptors | Excitation of sleep-active neurons | Enhanced sleep initiation and maintenance |
| Nucleus Accumbens | A2A Receptor | Modulation of striatal output pathways | Promotion of NREM sleep |
| Cortex | A1 Receptor | Direct cortical inhibition | Increased slow-wave activity |
Experimental Models and Methodologies for Studying Adenosine in Sleep
Research on adenosine-mediated sleep regulation employs diverse experimental approaches, from in vitro receptor pharmacology to in vivo animal models and human studies.
Pentobarbital-Induced Sleep Model
The pentobarbital-induced sleep model is a widely used pharmacological approach for screening potential sleep-modulating compounds. In a recent study investigating Phlomoides umbrosa Turczaninow root extract (PUTRE), ICR mice were injected with pentobarbital (45 mg/kg body weight) to induce sleep [13]. The experimental protocol involved:
Animal Preparation: 5-week-old female ICR mice (18-20 g) were acclimated to controlled conditions (temperature 23 ± 2°C, relative humidity 50% ± 5%, 12h light/dark cycle) for one week [13].
Treatment Administration: Mice were orally administered test substances (PUTRE at 200 or 400 mg/kg/day, or diazepam at 2 mg/kg/day as positive control) once daily for two weeks [13].
Sleep Measurements: Sleep latency (time until disappearance of righting reflex after pentobarbital injection) and sleep duration (time between disappearance and reappearance of righting reflex) were quantified [13].
Receptor Mechanism Studies: To investigate adenosine receptor specificity, antagonists including caffeine (non-selective adenosine receptor antagonist) and 8-cyclopentyl-1,3-dipropylxanthine (DPCPX; selective A1 receptor antagonist) were co-administered with PUTRE [13].
This model demonstrated that PUTRE significantly decreased sleep latency and increased sleep duration in a manner counteracted by adenosine receptor antagonists, confirming A1 receptor-specific agonist activity [13].
Receptor Antagonism and Genetic Models
Pharmacological antagonism studies provide crucial information about receptor specificity in adenosine-mediated sleep effects. Caffeine, as a non-selective adenosine receptor antagonist, promotes wakefulness and has been used extensively to characterize adenosine's role in sleep regulation [13]. More selective antagonists, including DPCPX (A1-selective) and SCH58261 (A2A-selective), allow finer dissection of receptor subtype contributions.
Genetic approaches, including knockout mice for specific adenosine receptor subtypes, have significantly advanced understanding of receptor-specific functions. A2A receptor knockout mice show reduced sleep pressure responses, while A1 receptor knockout mice display more subtle sleep phenotypes, suggesting compensatory mechanisms may develop [10]. Conditional and region-specific knockout strategies provide further spatial and temporal precision in dissecting adenosine receptor functions.
Diagram 2: Experimental Workflow for Pentobarbital-Induced Sleep Study. This diagram outlines the methodological sequence for investigating sleep-modulating compounds using the pentobarbital-induced sleep model in mice.
Neurochemical and Molecular Analyses
Advanced neurochemical monitoring techniques enable real-time measurement of adenosine fluctuations across sleep-wake cycles. In vivo microdialysis allows sampling of extracellular adenosine in specific brain regions, with high-performance liquid chromatography (HPLC) providing quantitative analysis of adenosine concentrations [9]. Complementary electrophysiological recordings (EEG/EMG) permit correlation of adenosine dynamics with sleep-wake states and spectral characteristics.
Molecular biology techniques including in situ hybridization, immunohistochemistry, and receptor autoradiography provide detailed information about receptor distribution and density. Recent human studies have revealed disease-associated alterations in adenosine receptor expression, such as increased A2A receptor density in atrial tissue from patients with atrial fibrillation, suggesting receptor dysregulation may contribute to pathophysiology [12].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents for Adenosine and Sleep Studies
| Reagent / Material | Category | Primary Function | Example Applications |
|---|---|---|---|
| Caffeine | Non-selective adenosine receptor antagonist | Blocks A1, A2A, A2B, A3 receptors | Establishing adenosine-mediated effects in sleep-wake regulation [13] |
| 8-Cyclopentyl-1,3-dipropylxanthine (DPCPX) | Selective A1 receptor antagonist | Competitively inhibits A1 receptors | Determining A1 receptor-specific contributions to sleep [13] |
| SCH58261 | Selective A2A receptor antagonist | Competitively inhibits A2A receptors | Dissecting A2A receptor functions in sleep regulation |
| Pentobarbital | GABA-A receptor agonist | Induces surgical anesthesia and experimental sleep | Screening sleep-modifying compounds in rodent models [13] |
| Diazepam | Benzodiazepine (GABA-A receptor positive allosteric modulator) | Positive control for sleep enhancement studies | Reference compound for comparing sleep-promoting efficacy [13] |
| Adenosine Deaminase | Enzyme | Catalyzes adenosine to inosine conversion | Experimental reduction of endogenous adenosine levels |
| ELISA Kits (melatonin, GABA) | Analytical tool | Quantifies sleep-related biomarkers in biological samples | Measuring hormone/neurotransmitter concentrations in serum or brain tissue [13] |
Therapeutic Implications and Future Research Directions
The adenosine system represents a promising therapeutic target for sleep disorders, with multiple approaches currently under investigation. Traditional adenosine receptor antagonists like caffeine demonstrate the wake-promoting potential of adenosine modulation, but their clinical utility is limited by side effects and tolerance development [14]. More sophisticated approaches include selective receptor subtype agonists and antagonists, allosteric modulators, and drugs targeting adenosine metabolism or transport.
Natural products and phytochemicals represent a growing area of research, with several plant extracts demonstrating sleep-enhancing effects through adenosine receptors. Phlomoides umbrosa Turczaninow root extract (PUTRE) has shown sleep-promoting effects mediated specifically through A1 receptor agonist activity [13]. Other herbal supplements including valerian, passionflower, lemon balm, ashwagandha, and chamomile have shown benefits for sleep disorders, though their mechanisms of action may involve multiple pathways beyond adenosine signaling [14]. The demand for a new class of alternative natural products with sleep-improving effects remains high, with good research value and broad prospects for insomnia treatment [9].
Future research directions should focus on several key areas. First, biased agonism of adenosine receptors—where ligands preferentially activate specific intracellular pathways—may enable separation of therapeutic effects from unwanted side effects [12]. Second, understanding receptor density alterations in pathological conditions could guide personalized approaches to sleep disorder treatment [12]. Third, the development of brain region-specific drug delivery could enhance efficacy while minimizing systemic side effects. Finally, further exploration of non-canonical adenosine functions in epigenetic regulation, immunometabolism, and cellular homeostasis may reveal novel therapeutic avenues [12].
Adenosine serves as a crucial endogenous sleep regulator that integrates metabolic signals with neural regulatory systems to maintain sleep-wake homeostasis. Through its actions on A1 and A2A receptors in key brain regions, adenosine inhibits wake-promoting systems while facilitating sleep-active nuclei, creating a coordinated transition between behavioral states. The progressive accumulation of adenosine during wakefulness provides a molecular basis for the sleep homeostat, linking prior waking duration to subsequent sleep intensity. Current research continues to elucidate the complex receptor mechanisms and neural circuits through which adenosine regulates sleep, providing innovative approaches for treating sleep disorders. The development of selective adenosine receptor modulators, including compounds derived from traditional medicinal plants, offers promising avenues for therapeutic intervention with potentially fewer side effects than conventional sleep medications. As our understanding of adenosine signaling in sleep regulation continues to expand, so too will opportunities for translating these insights into improved treatments for sleep disorders and related conditions.
The monoaminergic systems, comprising neurotransmitters such as serotonin (5-HT) and norepinephrine (NE), represent fundamental neuromodulatory networks within the central nervous system (CNS). These systems regulate a diverse array of neurological functions, from emotional processing and cognition to fundamental homeostatic processes including sleep-wake regulation [15] [16]. As primary CNS neuromodulators, monoamines do not primarily conduct neural signals but instead fine-tune the flow of information carried by amino acid-based neurotransmitters like glutamate and GABA, thereby shaping neural circuit dynamics in accordance with behavioral and physiological states [16].
The critical role of monoaminergic systems in sleep physiology and pathology is well-established. Disruptions in these pathways are implicated in various sleep disorders, which frequently co-occur with neuropsychiatric conditions such as depression and anxiety [17] [18]. This review provides an in-depth examination of the dual roles of serotonin and norepinephrine pathways, with particular emphasis on their implications for sleep biochemistry and disorders. We synthesize recent advances in structural biology, receptor mapping, and clinical studies to present a comprehensive resource for researchers and drug development professionals working at the intersection of monoaminergic neurotransmission and sleep medicine.
Molecular Architecture of Monoaminergic Systems
Structural Biology of Monoamine Transporters and Receptors
Monoamine transporters (MATs), including serotonin transporters (SERT) and norepinephrine transporters (NET), belong to the solute carrier 6 (SLC6) family of neurotransmitter sodium symporters [19]. These transporters regulate the spatial and temporal extent of monoaminergic signaling by mediating the reuptake of neurotransmitters from the synaptic cleft back into presynaptic neurons, thus maintaining neurotransmitter homeostasis [19].
Recent structural characterization of MATs using X-ray crystallography and cryo-electron microscopy has revealed their molecular architecture in unprecedented detail. Both SERT and NET contain twelve transmembrane (TM) helices, with ten organized into two inverted-topological repeats (TM1-TM5 and TM6-TM10) [19]. The first helix of each repeat is broken into two segments (TM1a-TM1b and TM6a-TM6b), creating a structural framework that harbors the primary substrate-binding site (S1) for neurotransmitters and co-transported ions near the broken regions of TM1 and TM6 [19].
Table 1: Key Structural Features of Monoamine Transporters
| Feature | Description | Functional Significance |
|---|---|---|
| Overall Fold | Twelve TM helices with two inverted repeats | Conservative structural framework supporting transport mechanism |
| Primary Binding Site (S1) | Located near broken regions of TM1 and TM6 | Binds neurotransmitter substrate and sodium ions |
| Sodium Binding Sites | Na1 (stabilizes outward-facing form) and Na2 (triggers inward-facing transition) | Couples neurotransmitter transport to sodium electrochemical gradient |
| Chloride Binding Site | Coordinated by residues in TM2, TM6, and TM7 | Required for transport activity in eukaryotic MATs |
| Extracellular Gates | Formed by TM1b and TM10a | Controls access to central binding site from extracellular milieu |
| C-terminal Latch | Associated with TM1a through intracellular loop IL1 | Potential role in modulating transporter activity and oligomerization |
The transport process is energetically coupled to the co-transport of Na+ ions down their electrochemical gradient. Eukaryotic MATs are also chloride-dependent, requiring Cl- co-transport for proper function [19]. The binding of neurotransmitters, along with Na+ and Cl- ions, elicits cooperative conformational changes in the transmembrane domain that facilitate substrate translocation across the membrane [19].
Receptor Diversity and Signaling Pathways
Serotonin and norepinephrine exert their effects through diverse receptor families, primarily G-protein coupled receptors (GPCRs). The serotonin system includes at least seven distinct receptor families (5-HT1 to 5-HT7), while norepinephrine signals primarily through α1-, α2-, and β-adrenergic receptors [20] [21]. These receptors differ in their G-protein coupling, downstream signaling cascades, and brain distributions, enabling highly specialized functional outcomes despite using similar primary neurotransmitters.
Recent mapping efforts using positron emission tomography (PET) have quantified the distribution of 19 different neurotransmitter receptors and transporters across nine neurotransmitter systems in the human cortex [20]. This comprehensive atlas reveals that receptor profiles align with structural connectivity and mediate neurophysiological dynamics, including oscillatory activity and functional connectivity [20]. The differential distribution of these receptors across cortical regions contributes to the specialized roles of serotonin and norepinephrine in regulating sleep architecture and sleep-related disorders.
Research Methods and Experimental Approaches
Assessing Monoaminergic Function in Sleep Research
Investigating the role of monoaminergic systems in sleep disorders requires multidisciplinary approaches spanning molecular, systems, and clinical neuroscience. The following methodologies represent core techniques in contemporary sleep research.
Polysomnography with Quantitative REM Sleep Analysis: Comprehensive sleep assessment requires laboratory-based polysomnography with special emphasis on quantifying REM sleep without atonia (RSWA). This methodology involves recording electromyography (EMG) signals from both the chin and bilateral flexor digitorum superficialis (FDS) muscles during REM sleep [22]. Epochs (30-second periods) are scored according to the American Academy of Sleep Medicine Manual for the Scoring of Sleep and Associated Events (Version 2.6). The percentage of REM sleep epochs exhibiting excessive muscle activity (RSWA epochs) is calculated, with a threshold of >27% considered diagnostic for REM sleep behavior disorder (RBD) [22]. This precise quantification is essential for investigating the impact of monoaminergic drugs on REM sleep architecture.
Population Studies and Depressive Symptom Assessment: Large-scale epidemiological studies investigating sleep and monoaminergic function often employ the Patient Health Questionnaire-9 (PHQ-9) to assess depressive symptom severity [17] [18]. This validated instrument aligns with DSM criteria for depression and captures symptom frequency over a two-week period. Item 3 of the PHQ-9 specifically addresses sleep disturbances, querying the frequency of "trouble falling or staying asleep, or sleeping too much" [18]. Responses are scored on a four-point Likert scale (0="not at all" to 3="nearly every day"), providing a quantitative measure of sleep disruption in relation to depressive symptoms.
Table 2: Key Reagent Solutions for Monoamine and Sleep Research
| Research Reagent | Primary Function/Application | Key Features and Considerations |
|---|---|---|
| PET Tracers (e.g., [11C]DASB for SERT, [11C]MRB for NET) | In vivo quantification of transporter availability and density | Enables whole-brain mapping of monoamine transporters; requires radiation safety protocols |
| Selective Agonists/Antagonists (e.g., prazosin for α1-adrenergic receptors) | Pharmacological dissection of receptor subtypes in sleep-wake regulation | Target-specific interventions to establish causal relationships between receptors and sleep phenotypes |
| AASM-Compliant Polysomnography Systems | Comprehensive sleep staging and architecture analysis | Gold standard for sleep disorder diagnosis; includes EEG, EOG, EMG, and respiratory monitoring |
| PHQ-9 and GAD-7 Questionnaires | Standardized assessment of depressive and anxiety symptoms | Validated instruments for quantifying psychological correlates of sleep disturbances |
| Electromyography (EMG) for RSWA Quantification | Objective measurement of muscle atonia during REM sleep | Critical for diagnosing REM sleep behavior disorder and assessing medication effects on REM sleep |
Molecular and Genetic Techniques
Advanced molecular techniques enable researchers to investigate the genetic and transcriptional regulation of monoaminergic systems. The transcription factor AP-2β has emerged as a key regulator of multiple genes within monoaminergic pathways, including the serotonin transporter (5-HTT), catechol-O-methyltransferase (COMT), dopamine-beta-hydroxylase (DBH), and vesicular monoamine transporter 2 (VMAT2) [23]. Investigating AP-2β signaling involves techniques such as chromatin immunoprecipitation (ChIP) to identify direct gene targets, promoter-reporter assays to quantify transcriptional activity, and genetic association studies linking TFAP2B polymorphisms with sleep and neuropsychiatric disorders [23].
Additionally, modern phylogenomic approaches have illuminated the evolutionary origins of monoaminergic systems. Reconciliation analysis using tools like GeneRax, which employs maximum likelihood approaches to optimize duplication and loss events given gene and species trees, has demonstrated that key enzymatic machinery for monoamine synthesis originated in the bilaterian stem group [15]. This evolutionary perspective informs our understanding of the conserved nature of monoaminergic signaling across species commonly used in sleep research.
Signaling Pathways and Neural Circuitry
The following diagram illustrates the key pathways through which serotonin and norepinephrine systems regulate sleep-wake states and how their dysregulation contributes to sleep disorders:
This pathway diagram illustrates several key relationships: (1) the fundamental role of monoaminergic neurotransmission in regulating sleep-wake states; (2) how disruption of these systems contributes to various sleep disorders; and (3) the modulatory effects of pharmacological interventions and comorbidities. The transcription factor AP-2β serves as a master regulator of multiple genes within monoaminergic systems, including SERT and NET, thereby influencing overall system function [23]. Serotonin predominantly promotes NREM sleep, while norepinephrine plays a critical role in REM sleep regulation and the maintenance of muscle atonia during REM periods [22]. Disruption of these pathways, whether through genetic factors, medication effects, or comorbidities like cardiovascular disease, can lead to sleep disturbances and conditions such as REM sleep behavior disorder (RBD) [17] [22].
Clinical Implications and Therapeutic Applications
Monoaminergic Dysregulation in Sleep and Neuropsychiatric Disorders
The intimate relationship between monoaminergic systems and sleep disorders is particularly evident in neuropsychiatric conditions. Recent population-based studies demonstrate a dose-response relationship between depressive symptom severity and sleep disorders in vulnerable populations such as postmenopausal women [17]. Each unit increase in PHQ-9 score was associated with a 10% higher risk of sleep disorders (OR=1.10, 95% CI: 1.07-1.13), with this association further amplified in individuals with comorbid cardiovascular disease [17].
Among patients with mental health disorders, sleep disturbances are exceptionally prevalent. A recent study of patients preparing for discharge from psychiatric units found that 79.6% experienced significant sleep disturbances, with key risk factors including relationship status, specific mental health diagnoses, anxiety, and poor wellbeing [18]. This remarkable prevalence underscores the tight coupling between monoaminergic dysregulation in psychiatric disorders and concomitant sleep disruption.
Pharmacological Modulation and Considerations
Antidepressants that target monoaminergic systems significantly impact sleep architecture, particularly REM sleep. Quantitative analysis reveals that specific medication classes differentially increase REM sleep without atonia (RSWA) [22]:
- SSRIs: Increase RSWA epochs by 4.1%
- SNRIs: Increase RSWA epochs by 5.6%
- SSRI+SNRI combinations: Increase RSWA epochs by 13.6%
- SNRI+TCA combinations: Increase RSWA epochs by 18.7%
These medications may unmask underlying predisposition to RBD rather than directly causing the disorder, as discontinuation often leads to transient resolution of symptoms followed by later recurrence, suggesting the medications reveal an underlying neuropathological process [22]. This has significant implications for both clinical management and drug development, highlighting the need for careful assessment of sleep-related side effects in psychopharmacological research.
Table 3: Monoamine-Targeting Antidepressants and Effects on Sleep Parameters
| Drug Class | Primary Mechanism | Key Effects on Sleep Architecture | Clinical Considerations |
|---|---|---|---|
| SSRIs (e.g., citalopram) | Selective inhibition of serotonin reuptake | Increased sleep latency, reduced REM sleep percentage, increased RSWA | May initially worsen insomnia; sedating variants preferred for anxious depression |
| SNRIs (e.g., venlafaxine) | Inhibition of serotonin and norepinephrine reuptake | Significant REM suppression, pronounced RSWA increases | Dose-dependent noradrenergic effects may impact sleep continuity |
| TCAs (e.g., clomipramine) | Broad monoamine reuptake inhibition with anticholinergic effects | Variable effects on sleep continuity; some agents are sedating | Anticholinergic properties may exacerbate RBD; not consistently linked to RSWA when used alone |
| MAOIs (e.g., phenelzine) | Inhibition of monoamine oxidase | Profound REM suppression; may improve sleep continuity in some cases | Dietary restrictions required; may modulate AP-2β transcription factor levels |
Future Research Directions and Therapeutic Innovations
The complex interplay between serotonin and norepinephrine pathways in sleep regulation presents both challenges and opportunities for therapeutic innovation. Future research directions should include:
Development of Circuit-Specific Therapeutics: Advanced receptor mapping indicates that neurotransmitter receptors are heterogeneously distributed across cortical regions and aligned with intrinsic functional networks [20]. This suggests potential for developing more targeted interventions that modulate specific neural circuits rather than employing broad monoaminergic modulation.
Transcriptional Regulation Approaches: The transcription factor AP-2β represents a promising target for modulating monoaminergic systems more harmoniously, as it coordinately regulates multiple genes within these pathways [23]. Pharmaceutical interventions targeting AP-2β activity or expression could potentially restore balanced monoaminergic function with fewer side effects than current reuptake inhibitors.
Biomarker Development: Integrating neurotransmitter receptor maps with genetic and proteomic data may yield biomarkers for predicting individual susceptibility to sleep side effects from monoaminergic medications, enabling personalized treatment approaches [20] [23].
Evolutionary Perspectives: Understanding the bilaterian origins of monoaminergic systems [15] provides fundamental insights into the conserved nature of these pathways across species, potentially informing translational research and animal models of sleep disorders.
The dual roles of serotonin and norepinephrine in sleep regulation underscore the complexity of these systems and the need for sophisticated approaches to therapeutic intervention. As our understanding of their molecular mechanisms, circuit functions, and interactions with other neurotransmitter systems deepens, so too will our ability to develop more effective and targeted treatments for sleep disorders rooted in monoaminergic dysregulation.
Melatonin, or N-acetyl-5-methoxytryptamine, is a neuroendocrine hormone that serves as a primary chronobiological regulator in vertebrates [24]. First isolated in 1958 from the bovine pineal gland, it is often termed the "hormone of darkness" due to its distinct diurnal secretion pattern, which is pivotal for synchronizing the body's internal physiology with the external light-dark cycle [25] [24]. Its synthesis, primarily within the pineal gland, is tightly controlled by the suprachiasmatic nucleus (SCN), the master circadian pacemaker in the hypothalamus [26] [27] [25]. Beyond its canonical role in circadian rhythm and sleep regulation, melatonin exerts pleiotropic effects, including potent antioxidant, anti-inflammatory, and immunomodulatory actions [26] [24]. This whitepaper details the molecular machinery of melatonin biosynthesis, its regulatory pathways, receptor-mediated signaling, and its integral function within the biochemistry of sleep, providing a technical foundation for researchers and drug development professionals.
Biosynthesis and Secretion of Melatonin
The Pineal Synthesis Pathway
Melatonin biosynthesis is a four-step enzymatic process originating from the essential amino acid tryptophan [24]. This pathway, summarized in the table below, is confined to specific tissues, with the pineal gland being the primary source of rhythmically secreted melatonin [26].
Table 1: Enzymatic Pathway for Melatonin Biosynthesis
| Step | Substrate | Enzyme | Product | Key Regulation |
|---|---|---|---|---|
| 1 | Tryptophan | Tryptophan Hydroxylase (TPH) | 5-Hydroxytryptophan | Rate-limiting step [26] |
| 2 | 5-Hydroxytryptophan | Aromatic L-amino acid decarboxylase | Serotonin (5-HT) | -- |
| 3 | Serotonin | Arylalkylamine N-acetyltransferase (AANAT) | N-Acetylserotonin (NAS) | Key rate-limiting enzyme; activated by darkness via norepinephrine; critical for circadian rhythm generation [26] [24] |
| 4 | N-Acetylserotonin | Acetylserotonin O-methyltransferase (ASMT) / Hydroxyindole-O-methyltransferase (HIOMT) | Melatonin | Final synthesis step [26] |
The activity of AANAT is the critical control point. During the day, light-induced signals from the SCN inhibit its activity, suppressing melatonin production. At night, norepinephrine release from sympathetic nerve terminals stimulates AANAT, leading to a surge in melatonin synthesis and secretion that peaks between 2:00 and 4:00 a.m. [25] [28]. The resulting melatonin is amphiphilic, allowing it to diffuse easily and cross physiological barriers [24].
Extrapineal Synthesis and Systemic Regulation
While the pineal gland is the central source of circulatory melatonin, numerous extrapineal sites, including the retina, gastrointestinal tract, skin, and immune cells, also synthesize it [26] [24]. Crucially, the mammalian gastrointestinal tract can contain over 400 times more melatonin than the pineal gland, but this extrapineal melatonin acts primarily as a local autocrine or paracrine signal; it does not exhibit a circadian rhythm and does not contribute significantly to the circulating levels that regulate sleep and circadian functions [24]. The SCN, receiving light input via the retinohypothalamic tract, serves as the master regulator. It relays signals through a multisynaptic pathway to the pineal gland, ensuring melatonin secretion is exquisitely synchronized with the environmental photoperiod [27] [25].
Diagram 1: Regulatory pathway of melatonin synthesis.
Melatonin Receptors and Signaling Mechanisms
Melatonin exerts its effects through receptor-dependent and receptor-independent pathways, enabling a diverse range of physiological actions.
Receptor-Mediated Signaling
The primary receptors for melatonin are MT1 and MT2, which are G-protein coupled receptors (GPCRs) with distinct signaling cascades [26] [25]. A third binding site, MT3, has been identified as the enzyme quinone reductase 2 [25]. The table below summarizes the key properties and signaling pathways of these receptors.
Table 2: Melatonin Receptors and Downstream Signaling
| Receptor | G-Protein Coupling | Key Signaling Pathways | Major Physiological Roles |
|---|---|---|---|
| MT1 | Gi | ↓ cAMP, ↓ PKA; ↑ PKC | SCN phase-shifting; vasoconstriction; sleep promotion; inhibition of neuronal firing in SCN [25] |
| MT2 | Gi | ↓ cAMP; ↑ PKC; ↑ cGMP | SCN phase-shifting; regulation of retinal dopamine; vasodilation; immune modulation [25] |
| MT3 (QRM2) | N/A (Enzyme) | Antioxidant defense | Detoxification; considered a low-affinity melatonin binding site [25] |
These receptors are widely expressed in the SCN, retina, pituitary gland, cardiovascular system, and immune cells, facilitating melatonin's role as a key synchronizer of circadian rhythms and other functions [26] [25] [28].
Non-Receptor-Mediated Actions
A significant aspect of melatonin's bioactivity is its potent, receptor-independent antioxidant capability. Melatonin and its metabolic derivatives act as direct scavengers of reactive oxygen species (ROS) and reactive nitrogen species (RNS) [25] [28]. Furthermore, it upregulates endogenous antioxidant enzymes like glutathione peroxidase and superoxide dismutase, while downregulating pro-oxidant enzymes [28]. This function is crucial for protecting mitochondrial integrity and reducing cellular oxidative stress, a mechanism implicated in its neuroprotective and anti-aging effects [29] [25].
Diagram 2: Melatonin signaling mechanisms.
Experimental Analysis of Melatonin Pathways
Research into melatonin's function relies on a suite of molecular, cellular, and biochemical techniques. The following protocol, derived from recent studies on hair follicle stem cells (HFSCs), exemplifies a comprehensive approach to dissecting melatonin signaling.
Experimental Objective: To characterize the dose-dependent effect of melatonin on HFSC viability and its molecular mechanism via the RORA/FOXC1 pathway [30].
1. Cell Culture and Treatment:
- Primary Cell Isolation: Isplicate the bulge region from rat whiskers and establish primary HFSC cultures using established enzymatic digestion and culture methods [30].
- Melatonin Treatment: Treat HFSCs with varying final concentrations of melatonin (e.g., 500 ng/L, 1000 ng/L [low dose], and 2000 ng/L [high dose]) dissolved in an appropriate vehicle (e.g., DMSO). Include a vehicle-only control group. Incubate for a predetermined period (e.g., 24-72 hours) [30].
2. Gene and Protein Expression Analysis:
- Real-Time qPCR: Extract total RNA and synthesize cDNA. Use SYBR Green-based qPCR with primers for Rorα and Foxc1 to quantify mRNA expression levels. Analyze data using the 2^–ΔΔCt method [30].
- Western Blotting: Extract total cellular proteins. Separate proteins by SDS-PAGE, transfer to a membrane, and probe with primary antibodies against RORA and FOXC1, followed by HRP-conjugated secondary antibodies. Detect using chemiluminescence [30].
- Chromatin Analysis: Perform CUT&Tag or CUT&RUN assays to investigate RORA binding to the Foxc1 promoter. Use an anti-RORA antibody to pull down DNA fragments, which are then sequenced or quantified by qPCR/ddPCR [30].
- Luciferase Reporter Assay: Clone the putative Foxc1 promoter region into a luciferase reporter vector. Co-transfect HFSCs with this construct and a RORA overexpression plasmid. Measure luciferase activity to confirm direct transcriptional regulation [30].
3. Functional Phenotype Assay:
- Cell Viability (CCK-8 Assay): After melatonin treatment, add CCK-8 reagent to culture wells. Incubate and measure the absorbance at 450 nm. The amount of formazan dye generated is directly proportional to the number of living cells [30].
- Gene Knockdown: Transfert HFSCs with Foxc1-specific siRNAs to knock down gene expression. Repeat the high-dose melatonin treatment and CCK-8 assay to determine if the phenotypic effect is reversed [30].
Diagram 3: Experimental workflow for melatonin signaling.
The Scientist's Toolkit: Key Research Reagents
The following table catalogues essential reagents and their applications for investigating melatonin synthesis, signaling, and function, based on cited experimental protocols.
Table 3: Research Reagent Solutions for Melatonin Studies
| Reagent / Kit | Function / Application | Example Use Case |
|---|---|---|
| Melatonin (HY-B0075) [30] | Primary agonist for MT1/MT2 receptors; used for in vitro and in vivo treatment. | Investigating dose-dependent effects on cell viability and gene expression [30]. |
| SR1075 (RORA Agonist) [30] | Chemical activator of the nuclear receptor RORA, a potential downstream target. | Probing the RORA-FOXC1 pathway independent of melatonin stimulation [30]. |
| SR3335 (RORA Inhibitor) [30] | Chemical inhibitor of RORA activity. | Validating the specific role of RORA in the melatonin signaling cascade [30]. |
| AANAT Antibodies | Detect and quantify the key rate-limiting enzyme in melatonin synthesis. | Immunohistochemistry or Western Blotting to localize and measure AANAT protein levels [26]. |
| MT1/MT2 Selective Agonists & Antagonists (e.g., ramelteon, luzindole) | Pharmacological tools to dissect receptor-specific functions. | Determining whether a physiological effect is mediated by MT1, MT2, or both receptors [25]. |
| CUT&Tag / CUT&RUN Kits | Map transcription factor binding sites on chromatin with high sensitivity. | Identifying direct binding of RORA to the promoter region of the Foxc1 gene [30]. |
| Droplet Digital PCR (ddPCR) | Absolute quantification of nucleic acids with high precision. | Accurately measuring copy number of genomic regions bound by transcription factors post-CUT&Tag [30]. |
| CCK-8 Assay Kit | Colorimetric measurement of cell viability and proliferation. | Assessing the impact of different melatonin doses on HFSC survival [30]. |
Melatonin in Sleep and Neurodegenerative Pathology
The role of melatonin in sleep is fundamental. Its secretion profile directly promotes sleep initiation and maintenance by reducing sleep latency and modulating sleep architecture. Abnormal melatonin secretion is a recognized factor in various sleep disorders, which are themselves risk factors for chronic diseases [25]. In the context of neurodegenerative diseases, circadian disruption and sleep-wake cycle alterations are now recognized as core features, often preceding clinical symptoms and contributing to disease pathophysiology [31] [29].
Research indicates that melatonin is dysregulated in conditions like Alzheimer's and Parkinson's disease. For instance, the endogenous onset of melatonin secretion (DLMO) is undetectable in a significant proportion of patients, indicating severe circadian dysfunction [32]. Therapeutically, melatonin's chronobiotic (rhythm-resetting), antioxidant, and anti-inflammatory properties make it a promising candidate for adjunct therapy. It is hypothesized to improve mitochondrial function, reduce oxidative damage, and help restore circadian alignment, thereby potentially slowing disease progression and improving sleep quality in patients [31] [29] [25].
This whitepaper provides a comprehensive analysis of the complex crosstalk between four pivotal hormonal regulators—cortisol, orexin, leptin, and ghrelin—within the context of sleep biochemistry and associated disorders. These hormones form an intricate signaling network that integrates metabolic status, stress response, and sleep-wake regulation, with significant implications for drug development targeting sleep pathologies and metabolic disorders. We synthesize current experimental evidence, delineate underlying molecular mechanisms, and present standardized methodological approaches for investigating this multifaceted hormonal interplay. The conceptual and mechanistic insights presented herein aim to facilitate the development of targeted therapeutic strategies that restore hormonal equilibrium in sleep-related disorders.
The regulation of sleep and wakefulness is a complex biological process orchestrated by numerous neurochemical and hormonal systems. Among these, cortisol, orexin, leptin, and ghrelin form a critical regulatory quadrad that integrates energy homeostasis, stress response, and sleep architecture. Cortisol, the primary glucocorticoid produced by the hypothalamic-pituitary-adrenal (HPA) axis, exhibits a robust circadian rhythm that influences sleep-stage distribution and promotes wakefulness. Orexin (hypocretin), a neuropeptide produced in the lateral hypothalamus, is a crucial stabilizer of wakefulness whose deficiency underlies narcolepsy pathogenesis. Leptin, an adipocyte-derived hormone, communicates energy sufficiency to the brain and promotes sleep, while ghrelin, primarily secreted from the stomach, stimulates appetite and influences sleep architecture. The bidirectional communication between these hormonal systems creates a complex network that modulates sleep-wake cycles, energy balance, and stress adaptation, with dysregulation in this crosstalk contributing significantly to various sleep disorders.
Hormonal Profiles and Physiological Roles
Individual Hormonal Characteristics
Table 1: Fundamental Characteristics of Key Hormones in Sleep Regulation
| Hormone | Primary Secretion Site | Major Receptors | Primary Sleep/Wake Role | Circulating Patterns |
|---|---|---|---|---|
| Cortisol | Adrenal cortex | Glucocorticoid receptor (GR) | Wake-promoting; suppresses REM sleep | Diurnal rhythm: peaks at waking, nadir at night |
| Orexin | Lateral hypothalamus | OX1-R, OX2-R | Wake stabilization; sleep-wake transitions | Highest during active period; minimal during sleep |
| Leptin | Adipose tissue | LepRb (long form) | Sleep promotion; increases SWS | Higher during sleep; proportional to fat mass |
| Ghrelin | Stomach (fundus) | GHS-R1a | REM sleep enhancement; orexigenic | Increases pre-prandially; decreases postprandially |
Detailed Hormonal Mechanisms
Cortisol secretion follows a circadian pattern regulated by the suprachiasmatic nucleus (SCN), with levels gradually increasing throughout the night and peaking during the early morning hours [33]. This hormone not only increases the duration and intensity of non-rapid eye movement (NREM) sleep but also inhibits REM sleep. Sleep disorders, including sleep deprivation and disruption, activate the HPA axis, resulting in elevated cortisol levels that perpetuate arousal states and can exacerbate insomnia [33].
Orexin exists in two forms, orexin A and B, which bind to G protein-coupled receptors OX1-R and OX2-R. OX1-R selectively binds orexin A, while OX2-R is nonselective for both orexins [34]. The orexin system projects widely throughout the brain, promoting wakefulness through interactions with monoaminergic and cholinergic systems. Notably, orexin A stimulates cortisol secretion from human adrenocortical cells through activation of the adenylate cyclase-dependent signaling cascade, demonstrating a direct endocrine interaction between these systems [34].
Leptin is a 167-amino acid peptide hormone produced predominantly by adipocytes and encoded by the obese (ob) gene on chromosome 7 [35]. Its circulating concentration correlates with adipose tissue mass and exhibits higher levels during sleep. Leptin regulates energy balance by decreasing appetite and increasing energy expenditure through activation of the JAK2/STAT3 signaling pathway in the hypothalamus, where it stimulates pro-opiomelanocortin (POMC) expression while inhibiting neuropeptide Y (NPY) expression [35].
Ghrelin is a 28-amino-acid peptide hormone predominantly secreted from the stomach, with approximately 60-70% of circulating ghrelin originating from gastric sources [36]. A unique post-translational modification—esterification of an n-octanoic acid to serine-3 by the enzyme ghrelin O-acyltransferase (GOAT)—is essential for its binding to the growth hormone secretagogue receptor 1a (GHS-R1a) and subsequent biological activity [36] [37]. The majority of circulating ghrelin exists in the unacylated form (UAG), which does not activate GHS-R1a but may modulate metabolic activities independently or in opposition to acylated ghrelin [36].
Bidirectional Hormonal Crosstalk
Leptin-Ghrelin antagonism
Leptin and ghrelin operate in a reciprocal manner to regulate energy balance and feeding behavior, creating what has been described as the "ghrelin-leptin tango" [38]. These hormones exhibit opposing roles in appetite control: ghrelin stimulates appetite through activation of orexigenic neurons in the arcuate nucleus, while leptin suppresses appetite by inhibiting these same neurons while simultaneously activating anorexigenic pathways [39]. The leptin/ghrelin ratio serves as a potential hunger signal, with research demonstrating this ratio is significantly higher in overweight/obese men compared to normal-weight individuals in both fasting and postprandial states [38].
In obesity, a paradoxical state of leptin resistance often develops, characterized by elevated leptin levels that fail to properly suppress appetite [35]. This resistance may occur at the blood-brain barrier level, where uncontrolled transport of the hormone from blood to brain contributes to dysregulated energy homeostasis. Meanwhile, ghrelin levels typically show an inverse relationship with body mass index (BMI), with obese individuals often exhibiting lower circulating ghrelin concentrations [39]. This hormonal imbalance creates a feed-forward cycle where leptin resistance promotes further weight gain, which in turn exacerbates the hormonal dysregulation.
Cortisol-Orexin Interactions
Orexin A demonstrates a specific capacity to stimulate cortisol secretion from human adrenocortical cells through activation of the adenylate cyclase-dependent signaling pathway [34]. This effect is concentration-dependent, with maximal effective concentration at 10^(-8) mol/L. Orexin A also enhances the cortisol response to maximal effective concentrations of angiotensin II and endothelin-1, and potentiates the response to low concentrations of ACTH (10^(-12)/10^(-11) mol/L) [34]. Reverse transcription polymerase chain reaction (RT-PCR) analyses have demonstrated high levels of OX1-R messenger RNA in the human adrenal zona fasciculata-reticularis, providing a molecular basis for this direct action [34].
Simultaneously, cortisol regulates orexin signaling through feedback mechanisms, with stress-induced increases in cortisol appearing to increase ghrelin in the periphery [37]. This creates a triangular relationship where cortisol influences both orexin and ghrelin signaling, potentially explaining how stressful life events trigger motivation for rewards and influence sleep-wake states.
Integrated Hormonal Network in Sleep Regulation
Diagram 1: Hypothalamic Integration of Hormonal Signals in Sleep Regulation
The hormonal crosstalk converges primarily within hypothalamic nuclei, particularly the arcuate nucleus (ARC), where ghrelin activates neuropeptide Y (NPY) and agouti-related peptide (AgRP) neurons to stimulate feeding and influence sleep architecture [36]. Leptin exerts opposing effects by inhibiting these orexigenic pathways while stimulating anorexigenic POMC neurons [35]. Simultaneously, orexin neurons in the lateral hypothalamus receive inputs from metabolic sensors and project widely to cortical and brainstem regions to stabilize wakefulness. Cortisol modulates this entire network through its widespread receptors and influences on gene expression, creating a tightly regulated system that aligns sleep-wake states with metabolic needs and environmental demands.
Molecular Signaling Pathways
Key Signal Transduction Mechanisms
Diagram 2: Intracellular Signaling Pathways of Hormonal Crosstalk
Leptin signaling initiates when leptin binds to the long form of its receptor (LepRb), triggering transphosphorylation of three tyrosine residues (Y985, Y1077, and Y1138) that activate receptor-associated kinase JAK2 [35]. This leads to phosphorylation of STAT3 and STAT5, which translocate to the nucleus and modulate gene expression—increasing POMC expression while decreasing NPY expression to suppress appetite [35]. The LepRb-Y985 signaling pathway specifically regulates hepatic insulin sensitivity, with ablation of this signaling improving whole-body insulin sensitivity through enhanced suppression of hepatic glucose production [35].
Ghrelin signaling primarily occurs through the GHS-R1a, a G-protein-coupled receptor with remarkable constitutive activity—signaling at approximately 50% of its maximal capacity even without hormone presence [36]. Upon binding acylated ghrelin, GHS-R1a activates multiple downstream pathways, including phospholipase C, protein kinase C, and inositol trisphosphate, ultimately stimulating GH release and activating hypothalamic NPY/AgRP neurons to promote feeding [36] [37].
Orexin signaling in the adrenal cortex occurs primarily through OX1-R receptors coupled to the adenylate cyclase-dependent pathway [34]. Orexin A binding increases cAMP production, which activates protein kinase A (PKA) and subsequently stimulates cortisol secretion. This effect can be blocked by the adenylate cyclase inhibitor SQ-22536 or the PKA inhibitor H-89, confirming the centrality of this pathway in orexin-induced cortisol release [34].
Cortisol signaling involves binding to glucocorticoid receptors (GR) that function as ligand-activated transcription factors. Upon cortisol binding, GR translocates to the nucleus and regulates gene expression by binding to glucocorticoid response elements (GREs) in promoter regions of target genes. This genomic action underlies cortisol's widespread effects on metabolic, immune, and neuronal functions relevant to sleep physiology.
Experimental Methodologies and Protocols
Standardized Approaches for Hormonal Analysis
Table 2: Experimental Protocols for Hormonal Crosstalk Investigation
| Experimental Goal | Recommended Protocol | Key Parameters Measured | Technical Considerations |
|---|---|---|---|
| Hormone Level Assessment | Blood collection in EDTA-aprotinin tubes; sample acidification; specific ELISA or RIA | Circulating levels of acylated ghrelin, leptin, cortisol, orexin | For ghrelin: add esterase inhibitors; process samples on ice; use specific assays for acylated vs. unacylated forms |
| Meal Response Testing | Crossover design with isocaloric meals (450 kcal) of varying macronutrient composition | Leptin/ghrelin ratio at 0, 30, 60, 120, 180, 240 min postprandial | Standardize meal timing; control for nutritional status; account for diurnal variations |
| Receptor Signaling Analysis | RT-PCR for receptor mRNA; EMSA for transcription factor binding; DLR assays for promoter activity | Receptor expression patterns; transcription factor activation; promoter responsiveness | Validate antibody specificity; include appropriate controls for constitutive activity |
| Sleep Architecture Assessment | Polysomnography with hormone sampling at sleep stages | Hormone levels across NREM (N1, N2, N3) and REM sleep | Control for circadian influences; standardized sleep conditions |
Detailed Protocol: Hormonal Response to Macronutrient Composition
Based on the crossover study design examining the leptin/ghrelin ratio following meals with varying macronutrient contents [38]:
- Participant Selection: Recruit age-matched groups with normal body weight (BMI < 25 kg/m²) and overweight/obese (BMI > 25 kg/m²) participants
- Study Design: Randomized crossover with 1-2 week washout periods between test meals
- Test Meals: Isocaloric meals (450 kcal) with different macronutrient compositions:
- High-carbohydrate/fat-free meal (HC-meal)
- Normo-carbohydrate/high-protein meal (NC-meal)
- High-fat/low-carbohydrate meal (HF-meal)
- Blood Sampling: Collect venous blood after a 12-hour fast immediately before meal intake (0 min) and at 30, 60, 120, 180, and 240 min postprandially
- Hormone Analysis:
- Measure leptin concentration using enzyme immunoassay (Human Leptin ELISA)
- Measure total ghrelin concentration using radioimmunoassay (Ghrelin (total) RIA)
- Calculate leptin/ghrelin ratio at each timepoint
- Statistical Analysis: Use one-way ANOVA or Wilcoxon signed-rank tests for paired samples with false discovery rate p-value adjustment for multiple comparisons
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Investigating Hormonal Crosstalk
| Reagent/Category | Specific Examples | Research Application | Functional Role |
|---|---|---|---|
| Enzyme Inhibitors | SQ-22536, H-89 | Signaling pathway dissection | SQ-22536 inhibits adenylate cyclase; H-89 inhibits PKA |
| Hormone Assays | Human Leptin ELISA, Ghrelin (total) RIA | Hormone quantification | Specific detection and measurement of hormone concentrations |
| Antibodies | Anti-leptin receptor, Anti-GHS-R1a | Receptor localization and expression | Immunohistochemistry, Western blotting for receptor characterization |
| Molecular Biology Tools | ABI5 knockout mutants (CR-abi5), Ubi::ABI5 overexpression constructs | Genetic manipulation of signaling pathways | CRISPR/Cas9-generated mutants; overexpression models for pathway analysis |
| Hormone Analogs | Synthetic GHS, Orexin A and B | Receptor activation studies | Selective receptor agonists for pathway stimulation |
| Signal Transduction Assays | EMSA, DLR assays, CUT&Tag | Transcription factor activity and DNA binding | Electrophoretic mobility shift assays; dual-luciferase reporter systems |
Implications for Sleep Disorders and Therapeutic Development
The intricate crosstalk between cortisol, orexin, leptin, and ghrelin has profound implications for understanding and treating sleep disorders. Insomnia and sleep fragmentation are frequently associated with HPA axis dysregulation, characterized by elevated cortisol levels and a flattened circadian rhythm [33]. Simultaneously, insufficient sleep itself can directly impact hormonal signaling, with sleep deprivation reducing leptin levels and increasing ghrelin concentrations—creating an endocrine environment that promotes appetite and weight gain [33] [39]. This bidirectional relationship between sleep loss and metabolic hormones may explain the strong epidemiological association between sleep disorders and metabolic conditions like obesity and type 2 diabetes.
Therapeutic strategies targeting this hormonal quadrad are emerging across multiple approaches. Leptin sensitizers represent a promising avenue for addressing leptin resistance in obesity-related sleep disturbances, potentially restoring appropriate satiety signaling and improving sleep quality. Ghrelin antagonists and GOAT inhibitors are being explored to block the orexigenic and wake-promoting effects of ghrelin, particularly in conditions where excessive hunger disrupts sleep maintenance. Orexin receptor antagonists have already demonstrated clinical utility for insomnia treatment, with dual orexin receptor antagonists (DORAs) like suvorexant promoting sleep without disrupting sleep architecture in the manner of traditional sedative-hypnotics. Finally, HPA axis modulators, including CRF1 receptor antagonists and glucocorticoid receptor antagonists, offer potential for normalizing cortisol rhythms in conditions of stress-related sleep disruption.
The complex interplay between these hormonal systems necessitates sophisticated therapeutic approaches that consider the entire regulatory network rather than isolated targets. Combination therapies that simultaneously address multiple components of this system may offer enhanced efficacy for treating complex sleep disorders with metabolic comorbidities. Furthermore, chronotherapeutic approaches that account for circadian variations in hormone sensitivity may optimize treatment timing to maximize benefit while minimizing side effects.
The crosstalk between cortisol, orexin, leptin, and ghrelin represents a fundamental regulatory network that integrates sleep-wake regulation, energy homeostasis, and stress adaptation. Understanding the bidirectional communication between these systems provides crucial insights into the pathophysiology of sleep disorders and their frequent comorbidities with metabolic conditions. The experimental methodologies and reagents outlined in this whitepaper provide a foundation for systematic investigation of this complex hormonal interplay.
Future research should prioritize several key areas: First, the development of more sophisticated experimental models that capture the dynamic, multi-hormonal interactions in physiologically relevant contexts. Second, the application of advanced techniques such as single-cell transcriptomics to delineate cell-type-specific responses to hormonal signals within sleep-regulatory circuits. Third, longitudinal clinical studies that track how age-related changes in these hormonal systems contribute to sleep fragmentation in elderly populations. Finally, the translation of mechanistic insights into targeted therapeutic strategies that restore balance to this regulatory network in specific sleep and metabolic disorders.
As our understanding of this hormonal crosstalk deepens, we move closer to personalized approaches for sleep medicine that account for individual variations in hormonal signaling, circadian physiology, and metabolic status. This integrated perspective promises more effective interventions for the multitude of disorders that arise from dysregulation in the intricate dance between cortisol, orexin, leptin, and ghrelin.
Advanced Techniques for Probing Sleep Biochemistry and Identifying Drug Targets
Sleep disorders represent a significant global health burden, necessitating advanced preclinical research to elucidate their underlying mechanisms and develop novel therapeutics. The biochemistry of sleep and its disorders involves complex interactions between neural circuits, genetic factors, and molecular pathways. Preclinical models are indispensable tools for deciphering these complexities and accelerating the translation of basic research into clinical applications. This technical guide provides a comprehensive overview of current approaches in preclinical sleep research, focusing on genetic, pharmacological, and electroencephalographic assessment methodologies. The integration of these approaches within a multidisciplinary framework is essential for addressing the multifaceted nature of sleep disorders and advancing mechanism-based drug discovery [40].
The field is currently undergoing a paradigm shift toward integrated, multidimensional models that better reflect the chronic, heterogeneous, and comorbid nature of human sleep disorders. This evolution is driven by recognition that oversimplified paradigms and limited modeling of comorbidity constrain clinical applicability. Emerging tools including optogenetics, chemogenetics, CRISPR, wearable EEG, and artificial intelligence are enabling unprecedented high-resolution mapping of sleep-wake mechanisms [40]. This guide examines how these advanced technologies are being incorporated into contemporary preclinical models to enhance their translational relevance for drug development professionals and basic researchers.
Classification and Selection of Preclinical Models
Genetic Models
Genetic models enable researchers to investigate the molecular and circuit-level basis of sleep disorders through targeted manipulation of specific genes or pathways. These models range from single gene mutations to complex polygenic models that better represent the genetic architecture of human sleep disorders.
Table 1: Genetic Models in Sleep Research
| Model Type | Key Genes/Pathways | Sleep Phenotype | Translational Relevance |
|---|---|---|---|
| Circadian Rhythm Models | Clock, Bmal1, Per1/2/3, Cry1/2 [41] | Altered sleep-wake cycle timing, disrupted rhythmicity | Familial advanced sleep phase syndrome (FASPS), circadian rhythm disorders |
| Narcolepsy Models | Hypocretin/orexin system, HLA genes [42] | Cataplexy, excessive daytime sleepiness, disrupted REM sleep | Narcolepsy with cataplexy, hypersomnia disorders |
| Neurodegenerative Models | Prion protein (PrP) gene [42] | Progressive insomnia, loss of sleep architecture | Fatal familial insomnia (FFI), synucleinopathies [43] |
| Down Syndrome Model | NKCC1 overexpression [44] | Sleep instability, fragmentation, reduced delta power | Down syndrome-associated sleep disturbances |
| Complex Trait Models | MEIS1, BTBD9, MAP2K5, SKOR1 [41] | Periodic limb movements, sensory symptoms | Restless legs syndrome (RLS)/periodic limb movement disorder |
Genetic models have been instrumental in elucidating the molecular basis of circadian rhythms. The core circadian mechanism involves a transcription-translation feedback loop where CLOCK and BMAL1 proteins form heterodimers that activate transcription of Per and Cry genes. PER and CRY proteins then accumulate, form complexes, and translocate back to the nucleus to inhibit CLOCK-BMAL1 activity, completing the approximately 24-hour cycle [41]. Additional regulatory layers include post-translational modifications by casein kinases that regulate protein stability and nuclear localization.
For narcolepsy research, genetic models have identified the critical role of the hypocretin/orexin system. The majority of human narcolepsy with cataplexy cases are associated with loss of hypocretin neurons, and genetic studies in autosomal recessive canine models and gene-targeted mice have demonstrated that mutations in hypocretin receptor 2 produce similar phenotypes [42]. These findings have been translated to human patients, where cerebrospinal fluid hypocretin levels are typically low [42].
Pharmacological Models
Pharmacological models involve administering compounds that either induce sleep disturbances or evaluate potential therapeutic effects. These models are particularly valuable for assessing drug efficacy, pharmacokinetics, and pharmacodynamics during preclinical development.
GABAergic modulators represent a well-established class of compounds used to study sleep architecture. Benzodiazepines like diazepam and non-benzodiazepine hypnotics such as zolpidem are commonly used to enhance GABAergic inhibition, producing characteristic changes in EEG power spectra including increased beta activity and decreased theta power [45]. These compounds serve as positive controls for evaluating novel hypnotics and studying the mechanisms underlying sleep maintenance.
Stimulants and wake-promoting agents including modafinil and caffeine are utilized to investigate the neurobiology of arousal and vigilance state control. Caffeine, an adenosine receptor antagonist, demonstrates well-characterized pro-wakefulness effects with increased time spent in wakefulness and delayed REM and NREM sleep onset [45]. These compounds are essential tools for modeling excessive daytime sleepiness and testing wake-promoting therapeutics.
Orexin receptor antagonists such as suvorexant represent a newer class of hypnotics that work by blocking the wake-promoting orexin system. These compounds produce distinct EEG signatures characterized by reduced wakefulness and specific alterations in REM sleep architecture [45]. They are valuable for studying the role of the orexin system in sleep-wake regulation and modeling insomnia phenotypes.
Disease-specific pharmacological models enable researchers to investigate sleep disturbances associated with neurological disorders. For example, bumetanide, an NKCC1 inhibitor, has been shown to improve sleep architecture and EEG information content in the Ts65Dn mouse model of Down syndrome by restoring inhibitory GABAergic transmission [44]. This approach demonstrates how targeting specific molecular pathways can rescue sleep abnormalities in genetic disorders.
Circuit Manipulation Models
Advanced techniques for precise manipulation of neural circuits provide unprecedented spatial and temporal control over specific sleep-wake regulatory systems. These approaches bridge the gap between genetic models and pharmacological interventions by enabling cell-type-specific and circuit-specific manipulations.
Optogenetics utilizes light-sensitive opsins to activate or inhibit specific neuronal populations with millisecond precision. This technique has been particularly valuable for dissecting the roles of various hypothalamic, brainstem, and basal forebrain nuclei in sleep-wake regulation. By targeting specific neural circuits, researchers can establish causal relationships between neuronal activity and sleep-wake states.
Chemogenetics employing Designer Receptors Exclusively Activated by Designer Drugs (DREADDs) allows for remote control of neuronal activity over longer timescales ranging from hours to days. This approach is well-suited for modeling chronic sleep disturbances and investigating the long-term consequences of circuit manipulation on sleep architecture and related physiological processes.
These circuit manipulation techniques are increasingly being integrated with polysomnographic recordings to establish direct links between specific neuronal populations and vigilance state control. When combined with genetic models, they enable researchers to determine whether sleep phenotypes result from developmental effects or ongoing physiological processes, providing crucial insights into disease mechanisms and potential therapeutic targets.
Quantitative Assessment Methodologies
Electroencephalographic (EEG) Biomarkers
Advanced EEG analysis provides non-invasive, high-temporal-resolution readouts of brain activity during sleep and wakefulness. These measures serve as essential biomarkers for characterizing sleep phenotypes and evaluating therapeutic interventions in preclinical models.
Table 2: Quantitative EEG Biomarkers in Preclinical Sleep Research
| Biomarker Category | Specific Measures | Biological Interpretation | Application Examples |
|---|---|---|---|
| Spectral Power Analysis | Delta (δ: 0.5-4 Hz), Theta (θ: 4-8 Hz), Alpha (α: 8-12 Hz), Beta (β: 12-30 Hz), Gamma (γ: 30-80 Hz) power bands [46] | Neural oscillatory activity balance, sleep depth, cortical arousal | OSA severity assessment, sleep depth quantification [47] |
| Sleep Architecture Parameters | Wake/NREM/REM latency, bout duration and number, stage transitions, sleep fragmentation index | Sleep stability, continuity, and organization | Sleep quality assessment, treatment efficacy evaluation [45] |
| Novel Computational Biomarkers | Odds Ratio Product (ORP) [47], EEG entropy/complexity, Slowing Ratio (SR) [46] | Sleep depth, cortical arousability, information processing capacity | Arousability assessment in adolescents [47], cognitive function evaluation |
| State-Dependent Spectral Fingerprinting | Power spectrum differences across wake, NREM, and REM states [45] | Compound-specific EEG signatures, mechanism of action | Pharmacological profiling, drug discovery |
The Odds Ratio Product (ORP) represents a recently developed EEG biomarker that provides a continuous measure of sleep depth, ranging from 0 (deepest sleep) to 2.5 (full wakefulness). ORP has demonstrated significant value in quantifying cortical arousability and its relationship to cardiac autonomic modulation. Longitudinal studies have shown that increased ORP during NREM sleep and in the 9 seconds following cortical arousals (ORP-9) is associated with altered heart rate variability, suggesting a coupling between sleep depth and cardiovascular regulation [47].
Sleep architecture analysis provides fundamental metrics for evaluating sleep quality and organization. Key parameters include the timing and duration of wake, NREM, and REM episodes, as well as transition patterns between states. In disease models, characteristic alterations in sleep architecture emerge, such as the increased fragmentation and reduced delta power observed in Ts65Dn mice modeling Down syndrome [44]. These parameters serve as primary endpoints for assessing therapeutic efficacy in preclinical trials.
State-dependent spectral fingerprinting enables detailed characterization of how pharmacological compounds affect brain activity across different vigilance states. Each vigilance state (wake, NREM, REM) exhibits distinct EEG patterns, and compounds with different mechanisms of action produce unique spectral signatures within each state. For example, benzodiazepines typically increase beta power during wakefulness and NREM sleep, while orexin receptor antagonists produce specific alterations in REM sleep architecture [45]. This approach facilitates mechanism-of-action studies and comparative profiling of novel therapeutics.
Molecular and Proteomic Profiling
Mass spectrometry-based proteomics technologies have emerged as powerful tools for unraveling the molecular correlates of sleep and sleep deprivation. These approaches provide comprehensive, unbiased assessments of protein abundance changes associated with sleep disturbances, enabling identification of novel pathways and therapeutic targets.
Recent proteomic analyses of chronic sleep restriction (CSR) in mouse cortex have revealed significant alterations in 149 proteins related to sleep and 1,269 proteins related to aging, with 96 proteins common to both conditions [48]. Pathway analysis of these overlapping proteins identified significant enrichment in complement and coagulation cascades, specifically involving complement component 3 (C3), alpha-2-macroglobulin (A2M), and fibrinogen alpha and beta chains (FGA and FGB) [48]. These findings provide the first proteomic evidence linking chronic sleep restriction to accelerated brain aging through shared molecular pathways.
Proteomic studies also enable detailed characterization of biochemical pathways affected by sleep disruption. In the CSR study, sleep restriction predominantly altered proteins involved in cytoplasmic translation, vesicle-mediated transport, and metallothionein binding, while aging specifically affected proteins associated with neuronal projections, synaptic organization, mitochondrial fatty acid β-oxidation, and neutrophil degranulation [48]. These distinct yet overlapping molecular signatures offer insights into potential mechanisms through which sleep loss may accelerate age-related neurological changes.
Circadian Rhythm Assessment
Comprehensive evaluation of circadian function is essential for modeling sleep disorders with circadian components. Multiple assessment methodologies provide complementary information about circadian regulation at behavioral, physiological, and molecular levels.
Actigraphy continuously monitors rest-activity cycles using wearable devices, providing objective measures of circadian rhythm strength, timing, and consolidation. In synucleinopathy research, actigraphy has revealed progressive disruption of rest-activity profiles across the disease spectrum from isolated REM sleep behavior disorder (iRBD) to Parkinson's disease (PD) and dementia with Lewy bodies (DLB) [43]. Specifically, the mesor (mean activity level) and amplitude (rhythm strength) of rest-activity rhythms show ordered decreases from healthy controls to iRBD, PD, and DLB, correlating with motor symptom severity [43].
Melatonin rhythm assessment evaluates the timing and amplitude of melatonin secretion, providing a physiological measure of circadian phase. Studies across synucleinopathies have demonstrated progressive disruption of melatonin rhythms, with DLB patients showing complete loss of circadian melatonin variation [43]. This hormonal measure complements behavioral assessments and offers insights into the integrity of the central circadian clock and its outputs.
Peripheral clock gene expression profiling using easily accessible tissues like oral mucosa enables assessment of molecular circadian rhythms. The expression of core clock genes such as Bmal1 shows rhythmic patterns that progressively dampen across the synucleinopathy spectrum, with significantly reduced amplitude in DLB patients compared to healthy controls [43]. This approach facilitates translational research by allowing repeated sampling in both preclinical models and human subjects.
Experimental Design and Protocols
Integrated Polysomnography Recording in Rodents
Comprehensive sleep assessment in preclinical models requires simultaneous recording of multiple physiological parameters to accurately identify vigilance states and quantify sleep architecture.
Surgical implantation: Animals are surgically prepared with EEG and electromyography (EMG) electrodes under general anesthesia. EEG electrodes are typically placed in frontal and parietal cortices to record electrical brain activity, while EMG electrodes are implanted in the nuchal muscle to monitor muscle tone. Proper electrode placement is verified post-surgery and before experimental recordings.
Habituation and baseline: Following post-surgical recovery (typically 7-10 days), animals are habituated to recording conditions, including tethering to recording equipment and the experimental environment. Baseline recordings are conducted for 24-48 hours to establish individual sleep-wake patterns and minimize stress-related artifacts.
Experimental recordings: Continuous polysomnographic recordings are performed throughout the experimental paradigm, which may include sleep restriction, drug administration, or other interventions. Recordings typically span multiple light-dark cycles to capture both active and inactive period sleep. High-quality signal acquisition requires proper cable management, electrical shielding, and regular monitoring.
Data analysis: Recordings are scored in epochs (typically 4-10 seconds) by trained personnel or validated machine learning algorithms to identify wake, NREM, and REM states based on characteristic EEG and EMG patterns. Subsequently, quantitative analyses are performed to extract sleep architecture parameters, spectral power distributions, and other relevant biomarkers.
Chronic Sleep Restriction Protocol
Modeling the effects of insufficient sleep, a hallmark of modern society, requires carefully controlled protocols that restrict sleep without causing excessive stress.
Apparatus setup: The sleep restriction apparatus typically consists of a cage with a rotating bar floor or multiple small home cages placed on activity wheels. The system is programmed to deliver gentle mechanical stimulation when animals attempt to sleep during restriction periods.
Restriction paradigm: Young adult mice (3-4 months old) are subjected to 20 hours of sleep deprivation followed by 4 hours of sleep opportunity for 5 consecutive days, typically during their active (dark) phase. This paradigm simulates the chronic sleep debt experienced by many humans.
Control groups: Appropriate control groups include normal sleep controls (undisturbed sleep), recovery sleep groups (allowed extended sleep opportunity after restriction), and sometimes sleep-deprived groups using different protocols for comparison.
Tissue collection and analysis: Following the restriction period, brain tissue (e.g., cerebral cortex) is collected at a standardized time point (e.g., ZT4) for molecular analyses including proteomics, transcriptomics, or other biochemical assays. Comparison with age-matched control groups enables identification of sleep restriction-specific molecular changes [48].
Pharmaco-EEG Profiling Protocol
Systematic evaluation of compound effects on sleep and vigilance states requires standardized protocols that enable state-dependent analysis of EEG signatures.
Baseline recording: Animals undergo 24-hour baseline EEG/EMG recordings to establish individual sleep-wake patterns and spectral profiles before compound administration.
Compound administration: Test compounds are administered at multiple doses following a cross-over or between-subjects design. Appropriate vehicle controls and reference compounds (e.g., modafinil for wake promotion, zolpidem for sleep enhancement) are included for comparison.
Post-administration recording: Continuous EEG/EMG recordings are performed for 6-24 hours following compound administration, depending on the compound's pharmacokinetics and expected duration of action.
State-dependent analysis: EEG power spectra are calculated separately for wake, NREM, and REM episodes, enabling detailed characterization of state-specific drug effects. Comparison with reference compounds in a comprehensive pharmacological database facilitates mechanism of action identification and profiling [45].
Dose-response relationship: Multiple dose levels are tested to establish the relationship between compound concentration and EEG effects, providing information about potency, efficacy, and therapeutic window.
Signaling Pathways and Molecular Mechanisms
The biochemical regulation of sleep involves complex interactions between multiple neurotransmitter systems, neuromodulators, and intracellular signaling pathways. The following diagrams visualize key molecular pathways and experimental workflows discussed in this guide.
Circadian Rhythm Molecular Feedback Loop
Pathway Description: The core molecular circadian clock consists of interlocking transcription-translation feedback loops. In the nucleus, CLOCK and BMAL1 proteins form heterodimers that bind to E-box elements in the promoters of Period (Per1, Per2, Per3) and Cryptochrome (Cry1, Cry2) genes, activating their transcription. PER and CRY proteins accumulate in the cytoplasm, where they form heterodimers and are phosphorylated by casein kinases (CK1δ/ε), regulating their stability and nuclear translocation. Upon entering the nucleus, PER-CRY complexes inhibit CLOCK-BMAL1-mediated transcription, completing the approximately 24-hour cycle. Additional regulatory loops involve REV-ERBα and ROR, which regulate Bmal1 expression through RORE elements [41].
Complement and Coagulation Pathway Activation in Sleep Restriction
Pathway Description: Proteomic analyses have revealed significant molecular overlap between chronic sleep restriction (CSR) and aging in the mouse cerebral cortex. Both conditions alter the abundance of 96 shared proteins, with significant enrichment in complement and coagulation cascade pathways. Key components include complement component 3 (C3), alpha-2-macroglobulin (A2M), and fibrinogen alpha and beta chains (FGA and FGB). These pathways represent novel molecular mechanisms through which chronic sleep insufficiency may accelerate brain aging processes. The complement system is known to play roles in synaptic pruning and neuroinflammation, while coagulation factors have been implicated in blood-brain barrier function and neurodegenerative processes [48].
Integrated Preclinical Sleep Assessment Workflow
Workflow Description: Comprehensive preclinical sleep assessment involves multiple integrated steps beginning with appropriate model selection based on the research question. Genetic models offer insights into molecular mechanisms, pharmacological models enable therapeutic evaluation, and circuit manipulation models provide causal insights. Polysomnography recording with simultaneous EEG and EMG monitoring enables precise identification of vigilance states and quantitative analysis of sleep architecture. Advanced computational biomarkers including the odds ratio product (ORP) and spectral power analysis provide objective measures of sleep depth and quality. Molecular analyses such as proteomics and clock gene expression profiling reveal underlying biochemical mechanisms. Finally, integrated analysis across these domains facilitates systems-level understanding of sleep physiology and pathology [40] [47] [45].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents and Solutions for Sleep Studies
| Reagent Category | Specific Examples | Research Application | Technical Considerations |
|---|---|---|---|
| Reference Compounds | Modafinil, caffeine (wake-promoting); Zolpidem, diazepam (hypnotic); Suvorexant (REM-affecting); Donepezil (procognitive) [45] | Pharmacological validation, mechanism of action studies, assay controls | Establish reference EEG signatures; dose-response critical for interpretation |
| Genetic Tools | CRISPR systems for gene editing; DREADDs (chemogenetics); Channelrhodopsins (optogenetics) [40] | Circuit manipulation, target validation, disease modeling | Require specific promoters for cell-type targeting; verification of expression and function essential |
| EEG/EMG Implants | Telemetric transmitters; Tethered electrode systems | Polysomnographic recording in freely-moving animals | Surgical expertise required; signal quality depends on proper placement and stabilization |
| Molecular Analysis Kits | Proteomic sample preparation; RNA extraction and qPCR; Melatonin immunoassays | Pathway analysis, circadian rhythm assessment, biomarker validation | Sample collection timing critical for circadian studies; proteomics requires specialized instrumentation |
| Activity Monitoring Systems | Actiwatch devices; Infrared beam breaks; Running wheels | Circadian rhythm assessment, gross motor activity quantification | Non-invasive longitudinal monitoring; complementary to EEG-based sleep analysis |
| Sleep Restriction Apparatus | Rotating drums; Multiple platform method; Gentle stimulation systems | Modeling sleep deprivation, chronic sleep restriction | Control for stress confounding; method selection depends on research question |
This toolkit represents essential resources for implementing the experimental approaches described in this guide. Proper selection and application of these reagents and systems requires careful consideration of the specific research objectives, model system characteristics, and available technical capabilities. Integration across multiple tools and approaches typically provides the most comprehensive insights into sleep-wake regulation and dysfunction.
The field of preclinical sleep research is rapidly evolving toward increasingly sophisticated, multidimensional models that better capture the complexity of human sleep disorders. The integration of genetic, pharmacological, and electroencephalographic approaches provides powerful complementary insights into the neural circuits, molecular pathways, and physiological processes underlying sleep-wake regulation. Future directions include increased incorporation of human-relevant genetic variants using CRISPR-based approaches, development of chronic disease models that better recapitulate disease progression, and application of artificial intelligence for enhanced analysis of high-dimensional data [40].
The biochemical investigation of sleep disorders increasingly recognizes the interconnectedness of neural, immune, and metabolic pathways. Recent proteomic evidence linking chronic sleep restriction to accelerated brain aging through complement and coagulation pathways highlights novel potential mechanisms and therapeutic targets [48]. Similarly, the demonstration that circadian disruption progresses across the synucleinopathy spectrum suggests opportunities for chronotherapeutic interventions in neurodegenerative diseases [43].
As these technologies and approaches continue to advance, they will undoubtedly yield deeper insights into the fundamental biochemistry of sleep and its disorders. This progress will facilitate the development of more effective, personalized therapeutics for the millions of individuals affected by sleep disorders, ultimately improving both quality of life and long-term neurological health.
The field of sleep disorder research is undergoing a transformative shift from subjective symptom assessment toward objective biomarker-driven classification. This paradigm leverages integrated profiles from peripheral fluids and neuroimaging to elucidate the complex pathophysiology underlying sleep disorders and their neurological consequences. The convergence of advanced analytical technologies—from high-sensitivity mass spectrometry to quantitative neuroimaging—now enables researchers to decode the molecular signatures of sleep disturbances and their downstream effects on brain health. Within biochemistry of sleep research, this approach provides critical insights into how sleep disruption propagates from molecular pathways to structural brain changes, creating opportunities for early intervention, patient stratification, and targeted therapeutic development.
Biomarker discovery in sleep disorders addresses a critical clinical need: the move beyond subjective questionnaires toward quantifiable, pathophysiological measures. As sleep disorders increasingly link to neurodegenerative conditions, cardiovascular disease, and metabolic syndromes, fluid and imaging biomarkers offer unprecedented windows into these relationships. For drug development professionals, these biomarkers provide objective endpoints for clinical trials and tools for identifying patient subgroups most likely to respond to targeted interventions. The integration of peripheral fluid biomarkers with neuroimaging correlates creates a comprehensive framework for understanding the bidirectional relationship between sleep pathophysiology and brain health, positioning biomarker discovery as a cornerstone of precision medicine in neurology and sleep medicine.
Emerging Fluid Biomarkers in Sleep Disorders
Circulating Transcriptomic and Metabolomic Signatures
Peripheral blood offers a accessible window into the physiological alterations associated with sleep disorders, with transcriptomic analyses revealing specific molecular patterns. A 2025 study investigated common molecular biomarkers connecting sleep disorders (SD) and stroke by analyzing transcriptome data from GEO datasets. Researchers identified differentially expressed genes (DEGs) and applied weighted gene co-expression network analysis (WGCNA) alongside machine learning algorithms (RandomForest, LASSO, and XGBoost). This integrated approach identified ARL2 as a key diagnostic biomarker with high predictive value (AUC = 0.91), subsequently validated in animal models where ARL2 was upregulated in experimental groups [49]. The enrichment analyses highlighted significant biological processes including 'response to external stimuli' and 'organic metabolic processes,' as well as metabolic pathways such as 'propionate metabolism' and 'oxidative phosphorylation,' suggesting their potential roles in the shared pathogenesis of sleep disorders and stroke [49].
Table: Key Circulating Biomarkers in Sleep Disorders Research
| Biomarker | Biological Significance | Associated Sleep Condition | Detection Method |
|---|---|---|---|
| ARL2 | Identified as common diagnostic marker for sleep disorders and stroke; high predictive value (AUC=0.91) | Sleep disorders and stroke comorbidity | Transcriptome analysis, machine learning validation |
| Glycerol | Decreased in poor sleep quality; component of random forest model | Chronic sleep disorders | CE-FTMS mass spectrometry |
| Hippuric Acid | Decreased in poor sleep quality; involved in tyrosine metabolism | Chronic sleep disorders | CE-FTMS mass spectrometry |
| 2-Hydroxybutyric Acid (2HB) | Increased in poor sleep quality; correlates with sleep length dissatisfaction | Chronic sleep disorders | CE-FTMS mass spectrometry |
| Neutrophil-to-Lymphocyte Ratio (NLR) | Indicator of systemic inflammation; associated with vascular pathology | CSVD with sleep implications | Complete blood count differential |
The analytical workflow for circulating biomarker discovery typically integrates multiple computational approaches to enhance biomarker specificity and clinical relevance, as illustrated below:
Salivary Metabolomics for Non-Invasive Assessment
Saliva has emerged as a particularly promising biofluid for sleep biomarker discovery due to its non-invasive collection and rich metabolomic profile. A 2025 investigation utilized capillary electrophoresis–Fourier transform mass spectrometry (CE-FTMS) to analyze metabolomes in saliva from 50 individuals with good sleep quality (PSQI-J ≤ 2) and 50 with poor sleep quality (PSQI-J ≥ 6) [50]. The study identified five metabolites (including glycerol and hippuric acid) that were significantly decreased, and eight metabolites (including 2-hydroxybutyric acid and gamma-glutamylthreonine) that were significantly increased in participants with poor sleep quality [50].
A random forest model incorporating six metabolites—glycerol, hippuric acid, iminodiacetic acid, phosphocreatine, trimethylamine, and 3-phenylpropionic acid—achieved a prediction accuracy of 0.866 for distinguishing sleep quality groups [50]. Correlation analyses with the Oguri-Shirakawa-Azumi sleep inventory revealed significant relationships between specific metabolites and sleep parameters: 2'-deoxyguanosine, N1-acetylspermine, and 2,4-dihydroxybenzoic acid positively correlated with sleep satisfaction, while glucosamine 6-phosphate and trimethylamine N-oxide correlated negatively [50]. These findings position salivary metabolomics as a powerful, non-invasive approach for objectively quantifying sleep quality and understanding the metabolic underpinnings of sleep disorders.
Neuroimaging Correlates of Sleep-Related Brain Pathology
Cerebral Small Vessel Disease and Sleep Disruption
Neuroimaging biomarkers provide critical insights into the structural brain changes associated with chronic sleep disorders, particularly cerebral small vessel disease (CSVD). A 2025 study investigated the relationship between peripheral blood inflammatory markers and CSVD imaging burden in 1,526 patients [51]. The research demonstrated significant correlations between neutrophil count (NC) and enlarged perivascular spaces (EPVS), as well as between systemic immune-inflammation index (SII) and deep cerebral microbleeds [51]. These findings illuminate the potential mechanistic link between sleep-related inflammation and cerebrovascular pathology, suggesting that peripheral inflammatory markers may serve as accessible proxies for sleep-related cerebrovascular damage.
The association between CSVD and sleep disorders is particularly relevant given CSVD's role in approximately 25% of ischemic stroke cases and 45% of dementia diagnoses [51]. The inflammatory pathway represents a key mechanism through which sleep disturbances may contribute to CSVD progression, involving endothelial dysfunction, impaired blood-brain barrier integrity, damage to the neurovascular unit, and low perfusion injury [51]. The infiltration of circulating neutrophils into perivascular spaces represents a direct pathway through which systemic inflammation—often elevated in sleep disorders—can compromise blood-brain barrier integrity and accelerate vascular pathology [51].
Advanced Neuroimaging Techniques for Sleep-Related Neurodegeneration
Novel neuroimaging approaches are increasingly applied to understand how sleep disorders affect brain structures vulnerable to neurodegenerative processes. Neuromelanin-sensitive MRI (NM-MRI) has emerged as a particularly sensitive technique for visualizing degeneration in the substantia nigra and locus coeruleus—brain regions critically involved in sleep-wake regulation and profoundly affected in both sleep disorders and neurodegenerative conditions [52]. A 2021 meta-analysis determined that NM-MRI of the substantia nigra and locus coeruleus could distinguish between Parkinson's disease and controls with a pooled sensitivity of 89% and specificity of 83% [52], suggesting potential applications for detecting sleep-related neurodegeneration.
Other promising techniques include free water imaging, which quantifies changes in extracellular space associated with neuronal loss, and quantitative susceptibility mapping (QSM), which measures iron content in brain regions affected by neurodegenerative processes [52]. Iron-mediated cell death (ferroptosis) represents a particularly relevant pathway connecting sleep disorders to neurodegeneration, as excess iron promotes aggregation of alpha-synuclein fibrils—a key pathological feature in several synucleinopathies that commonly present with sleep disturbances [52]. These advanced imaging modalities offer sensitive measures of early pathological changes that may precede overt cognitive or motor symptoms in patients with chronic sleep disorders.
Table: Neuroimaging Biomarkers Relevant to Sleep Disorder Research
| Imaging Technique | Measured Parameter | Biological Significance | Relevance to Sleep Disorders |
|---|---|---|---|
| NM-MRI | Neuromelanin signal in SN/LC | Marker of catecholaminergic neuron integrity | Sleep-wake regulation dysfunction |
| Free Water Imaging | Extracellular water content | Indicator of neuronal loss/tissue degeneration | CSVD and neurodegenerative progression |
| QSM | Tissue iron concentration | Iron-related oxidative stress and ferroptosis | Neurodegenerative pathology linking sleep disorders |
| Cardiac MIBG Scintigraphy | Cardiac sympathetic innervation | Measure of autonomic dysfunction | REM sleep behavior disorder and synucleinopathies |
| DAT SPECT | Striatal dopaminergic terminal integrity | Presynaptic dopamine system function | Sleep-wake regulation and Parkinsonian disorders |
Integrated Experimental Protocols
Protocol 1: Transcriptomic Biomarker Discovery for Sleep Disorders
Objective: Identify circulating diagnostic biomarkers connecting sleep disorders and stroke using integrated bioinformatics and machine learning approaches.
Sample Preparation:
- Obtain transcriptome data from public repositories (e.g., GEO database); for sleep disorders, use dataset GSE208668 with RNA expression data from 17 SD patients and 25 healthy controls on platform GPL10904 [49].
- For validation cohorts, utilize independent datasets GSE22255 and GSE98566 [49].
- Perform gene symbol mapping according to respective platforms; for multiple matches, take the median and normalize expression matrix using log2(X + 1) method [49].
- Conduct quantile normalization using the "normalizeBetweenArrays" function in the "limma" package to reduce technical variations between samples [49].
Data Analysis Workflow:
- Differential Expression Analysis: Utilize limma package for DEG analysis with stringent cutoff criterion (adjusted p-value < 0.05 and absolute log2 fold change > 0.5) [49].
- Functional Enrichment: Conduct GO and KEGG enrichment analyses using clusterProfiler package to identify key biological processes and pathways [49].
- Co-expression Network Analysis: Apply WGCNA to identify gene modules co-expressed in sleep disorders and stroke, revealing shared mechanisms in inflammation, oxidative stress, and circadian regulation [49].
- Machine Learning Validation: Implement multiple algorithms (RandomForest, LASSO, and XGBoost) to identify high-confidence diagnostic biomarkers [49].
- Experimental Validation: Conduct animal studies to verify biomarker expression in experimental models [49].
The following diagram illustrates the integrated computational and experimental workflow for robust biomarker identification:
Protocol 2: Salivary Metabolomic Profiling for Sleep Quality Assessment
Objective: Identify salivary metabolic biomarkers for non-invasive assessment of chronic sleep disorders.
Sample Collection and Preparation:
- Recruit participants with good (PSQI-J ≤ 2) and poor (PSQI-J ≥ 6) sleep quality (n=50 per group) [50].
- Collect saliva samples and confirm chronic poor sleep status using OSA-MA questionnaire administered close to saliva collection date [50].
- Process samples using capillary electrophoresis–Fourier transform mass spectrometry (CE-FTMS) for metabolomic analysis [50].
- Identify and quantify 683 metabolites from saliva samples [50].
Data Processing and Statistical Analysis:
- Metabolite Detection: Identify 683 metabolites in saliva samples using CE-FTMS [50].
- Multivariate Analysis: Perform principal component analysis (PCA) to visualize group separation and partial least squares (PLS) for discriminative profiling [50].
- Differential Abundance Testing: Compare metabolite levels between groups using Mann-Whitney U tests with significance threshold of p < 0.05 [50].
- Pathway Analysis: Conduct metabolite set enrichment analysis (MSEA) to identify altered metabolic pathways [50].
- Predictive Modeling: Develop random forest classification model with leave-one-out cross validation, using stepwise forward selection based on mean decrease accuracy [50].
- Correlation Analysis: Assess relationships between metabolite levels and sleep parameters from OSA-MA questionnaires [50].
Statistical Considerations for High-Dimensional Biomarker Data
The analysis of high-dimensional biomarker data from transcriptomic and metabolomic studies requires careful statistical approaches to avoid false discoveries and ensure robust results. A comprehensive 2022 comparison of statistical methods for metabolomics data revealed that optimal approaches vary depending on sample size, number of metabolites assayed, and outcome type [53]. With an increasing number of study subjects, univariate methods (such as FDR correction) result in apparently higher false discovery rates due to substantial correlation between metabolites directly associated with the outcome and those not associated with the outcome [53].
For nontargeted metabolomics datasets including thousands of metabolite measures, sparse multivariate models (such as LASSO and sparse partial least squares) demonstrate greater selectivity and lower potential for spurious relationships [53]. When the number of metabolites is similar to or exceeds the number of study subjects—common in nontargeted metabolomics analysis of relatively small cohorts—sparse multivariate models exhibit the most robust statistical power with more consistent results [53]. For continuous outcomes, both LASSO and SPLS methods perform remarkably well, with SPLS slightly outperforming LASSO in terms of positive predictive value, negative predictive value, and number of false positives [53]. These statistical considerations are essential for designing robust biomarker discovery studies in sleep research.
The Scientist's Toolkit: Essential Research Reagents and Platforms
Table: Key Research Reagents and Platforms for Biomarker Discovery in Sleep Research
| Reagent/Platform | Application | Specific Function | Example Use |
|---|---|---|---|
| CE-FTMS Mass Spectrometry | Metabolomic profiling | High-resolution separation and detection of metabolites | Salivary metabolome analysis in sleep disorders [50] |
| Limma R Package | Transcriptomic analysis | Differential expression analysis for microarray/RNA-seq | Identifying DEGs in sleep disorder transcriptomes [49] |
| WGCNA R Package | Systems biology | Weighted gene co-expression network analysis | Identifying gene modules in sleep disorders and stroke [49] |
| clusterProfiler R Package | Functional enrichment | GO and KEGG pathway enrichment analysis | Pathway analysis of sleep disorder transcriptomes [49] |
| Random Forest Algorithm | Machine learning classification | Multivariate pattern recognition and prediction | Developing sleep quality prediction models [49] [50] |
| LASSO Regression | Feature selection | Sarse multivariate model with regularization | Selecting most predictive biomarkers from high-dimensional data [49] [53] |
| NM-MRI Sequences | Neuroimaging | Neuromelanin-sensitive magnetic resonance imaging | Quantifying integrity of locus coeruleus and substantia nigra [52] |
| QSM MRI Sequences | Neuroimaging | Quantitative susceptibility mapping for iron content | Measuring brain iron accumulation in sleep disorders [52] |
The integration of peripheral fluid biomarkers and neuroimaging correlates represents a transformative approach to understanding, diagnosing, and treating sleep disorders. Circulating transcripts, salivary metabolites, and inflammatory markers provide accessible windows into the pathophysiology of sleep disturbances, while advanced neuroimaging techniques reveal their structural consequences on the brain. Together, these biomarker modalities create a comprehensive framework for decoding the complex relationship between sleep disruption and neurological health.
For researchers and drug development professionals, these biomarker platforms offer powerful tools for patient stratification, target engagement assessment, and treatment monitoring. The experimental protocols and statistical considerations outlined in this review provide a roadmap for rigorous biomarker discovery and validation. As the field advances, the integration of multi-omics approaches with advanced neuroimaging will further unravel the biochemical pathways linking sleep disorders to neurological outcomes, paving the way for personalized interventions that preserve both sleep and brain health.
High-Throughput Screening for Novel Hypnotics and Wake-Promoting Agents
Sleep-wake disorders, encompassing conditions like insomnia, narcolepsy, and excessive daytime sleepiness (EDS), represent a significant and growing global health concern. Narcolepsy alone, a chronic neurological disorder characterized by EDS and cataplexy, affects approximately 170,000 Americans, with symptom onset often occurring in adolescence and diagnosis frequently delayed for up to a decade [54]. The biochemistry of sleep and wakefulness is governed by a complex interplay of neurocircuits and neurotransmitters. Wake-promoting systems include monoaminergic (e.g., histamine, norepinephrine) and cholinergic neurons, while sleep promotion is primarily mediated by inhibitory projections from the ventrolateral preoptic nucleus (VLPO) that release GABA and galanin to suppress wake-promoting nuclei [55]. A key regulator is the hypocretin/orexin system, located in the lateral hypothalamus, which stabilizes wakefulness; its loss is a primary cause of narcolepsy type 1 [54] [55]. This intricate neurobiological framework provides a rich source of molecular targets for therapeutic intervention. The high prevalence of these disorders, coupled with the limitations of existing treatments—such as the side effects of benzodiazepine receptor agonists and the abuse potential of amphetamines—has intensified the search for novel, targeted therapeutics acting on specific sleep-wake circuits, including orexin receptors, histamine H3 receptors, and adenosine systems [55]. High-throughput screening (HTS) has emerged as a powerful strategy to accelerate the discovery of such compounds, enabling the rapid evaluation of vast chemical libraries against these defined biological targets.
High-Throughput Screening Methodologies and Experimental Protocols
Core Principles of Quantitative High-Throughput Screening (qHTS)
Traditional HTS involves screening thousands of compounds at a single concentration, but this approach has been largely superseded by quantitative HTS (qHTS). qHTS performs multiple-concentration experiments in low-volume cellular systems (e.g., <10 μl per well in 1536-well plates), generating full concentration-response curves for each compound simultaneously [56]. This method offers significantly lower false-positive and false-negative rates compared to traditional single-concentration HTS. In a qHTS assay, large chemical libraries are screened against a specific target, and concentration-response data are fitted to pre-specified statistical models, most commonly the Hill equation (HEQN), to estimate key parameters such as AC50 (half-maximal activity concentration) and Emax (maximal response) for each compound [56]. However, the reliability of parameter estimation is highly dependent on the assay design, including the range of tested concentrations and the spacing of concentration points. Parameter estimates can be highly variable if the tested concentration range fails to capture at least one of the two asymptotes of the Hill curve [56].
Establishing a Cell-Based HTS System for Sleep-Wake Targets
The establishment of a robust, cell-based HTS system is a foundational step. A representative protocol for screening modulators of GABAA receptors, a key target for hypnotics, involves the following steps [57]:
- Generation of a Stable Cell Line: A CHO (Chinese Hamster Ovary) cell line stably expressing the human GABAA1 receptor (comprising α1, β2, and γ2L subunits) is generated. Using an induced expression strategy (e.g., the CHO-T-Rex system) can decrease cytotoxicity associated with constitutive expression.
- Functional Assay Development: A functional high-throughput screening assay is established based on measuring changes in membrane potential using a Fluorometric Imaging Plate Reader (FLIPR). This method detects the flux of ions through the activated receptor channel.
- Assay Validation and Optimization: The assay is optimized and validated using known tool compounds:
- Agonist: GABA (EC50 ~137 nM)
- Positive Allosteric Modulator: Diazepam (EC50 ~3.22 μM)
- Antagonists: Gabazine (IC50 ~0.16 μM) and Bicuculline (IC50 ~0.47 μM)
- Primary Screening: The validated assay is used to screen a compound library (e.g., 10,000 compounds). Active "hit" compounds are selected based on their activity in the membrane potential assay.
- Secondary Confirmation: Active hits are confirmed using a gold-standard, lower-throughput method such as patch-clamp electrophysiology to determine precise EC50/IC50 values and characterize pharmacological properties.
- In Vivo Validation: Promising compounds are advanced into animal models, such as the pentobarbital-induced sleep model in mice, to confirm hypnotic efficacy by measuring the reduction in sleep onset latency and increase in total sleep time [57] [58].
Data Analysis and Artifact Mitigation in qHTS
A critical challenge in qHTS is distinguishing true biological activity from assay artifacts, such as compound autofluorescence, chemical reactivity, and nonspecific cytotoxicity. A dedicated data analysis pipeline is required to address these issues [59]. One effective approach includes:
- Signal Noise Filtering: Implementing a signal noise-filtering and curation protocol to remove non-reproducible signals.
- Assay Interference Flagging: Using an assay interference flagging system to identify and label compounds with autofluorescence or other confounding properties.
- Activity Profiling: Adopting a weighted version of the area under the curve (wAUC) to quantify the total activity across the tested concentration range. Research on Tox21 qHTS assays has demonstrated that wAUC affords the best reproducibility (Pearson’s r = 0.91) compared to the point-of-departure (POD) concentration (r = 0.82) or the AC50 (r = 0.81) [59].
- Cytotoxicity Assessment: Characterizing and controlling for cytotoxicity, which is a major confounding factor affecting, on average, about 8% of compounds in Tox21 assays, compared to autofluorescence which affects less than 0.5% [59].
Table 1: Key Parameters for Analyzing qHTS Concentration-Response Data
| Parameter | Description | Biological Interpretation | Considerations |
|---|---|---|---|
| AC50 | The concentration that produces 50% of the maximal activity. | Potency of the compound. | Highly variable if concentration range does not capture asymptotes [56]. |
| Emax | The maximal efficacy of the compound. | Intrinsic activity of the compound. | Useful for comparing full vs. partial agonists [56]. |
| wAUC (Weighted Area Under the Curve) | Integrates the level of activity across the entire tested concentration range. | Overall activity and potency. | Shows superior reproducibility for profiling compounds in qHTS [59]. |
| POD (Point-of-Departure) | The lowest concentration where a statistically significant response is detected. | Threshold for biological activity. | Used in toxicological hazard assessment [59]. |
Key Molecular Targets and Recent Advances
Wake-Promoting Targets
The most promising wake-promoting targets currently under investigation include:
- Orexin 2 Receptor (OX2R) Agonists: The orexin/hypocretin system is a primary regulator of wakefulness stability. Loss of orexin-producing neurons is a key pathology in narcolepsy type 1. Consequently, OX2R agonists represent a targeted therapeutic strategy. For example, BP1.15205 is a highly potent (EC50 = 0.015 nM) and selective (>600-fold over OX1R) OX2R agonist. In preclinical models, it demonstrated significant, dose-dependent increases in wakefulness and reductions in cataplexy-like episodes, with a potential for once-daily dosing. A first-in-human study was planned for the second half of 2025 [54].
- Histamine H3 Receptor Inverse Agonists: The histaminergic system is a key wake-promoting pathway. Histamine H3 receptors function as autoreceptors that inhibit the release of histamine and other wake-promoting neurotransmitters. Pitolisant, an H3 receptor inverse agonist, is an approved wake-promoting agent that works by enhancing endogenous histamine release [60] [55].
- Dopamine and Norepinephrine Reuptake Inhibitors: Solriamfetol is a dual-acting dopamine and norepinephrine reuptake inhibitor (DNRI) approved for EDS in narcolepsy. A 2025 network meta-analysis found that solriamfetol 300 mg was highly effective in reducing Epworth Sleepiness Scale (ESS) scores and prolonging sleep latency on the Maintenance of Wakefulness Test (MWT) [60].
Sleep-Promoting (Hypnotic) Targets
For insomnia, research is moving beyond traditional GABAergic drugs to targets that may offer more natural sleep architecture with fewer side effects:
- Adenosine A1 Receptor Agonists: Adenosine is an endogenous sleep-promoter that accumulates in the brain during wakefulness. Agonists of the A1 receptor can directly inhibit wake-promoting neurons in the basal forebrain and lateral hypothalamus. Recent research on a novel compound, AR-001, demonstrated its hypnotic effect in a pentobarbital-induced sleep model. This effect was blocked by an A1 receptor antagonist (DPCPX), and immunohistochemistry showed that AR-001 increased neural activity in the sleep-promoting VLPO while decreasing activity in wake-promoting regions [58].
- GABAA Receptor Modulators: Despite the search for new targets, GABA remains the primary inhibitory neurotransmitter in the brain, and GABAA receptors are still a mainstay for hypnotic development. The HTS system described for GABAA1 modulators successfully identified novel positive allosteric modulators that increased sleep duration and quality in mouse models [57].
Table 2: Profile of Approved and Investigational Wake-Promoting Agents
| Drug (Example) | Mechanism of Action | Key Efficacy Findings (vs. Placebo) | Notable Adverse Effects |
|---|---|---|---|
| Solriamfetol (300 mg) | Dopamine & Norepinephrine Reuptake Inhibitor | MD in ESS: -4.74; MD in MWT latency: +10.82 min [60] | Anxiety, headache [60] |
| Pitolisant | Histamine H3 Receptor Inverse Agonist | Reduces ESS and MWT latency [60] | Nausea [60] |
| Sodium Oxybate (SXB) | GABAB Receptor Agonist | Reduces cataplexy and EDS [60] | High risk of gastrointestinal effects (diarrhea, vomiting) [60] |
| BP1.15205 | Orexin 2 Receptor (OX2R) Agonist | Preclinical: Increases wakefulness, suppresses cataplexy [54] | No adverse events in 3-month GLP toxicity study (pending histopathology) [54] |
| Modafinil/Armodafinil | Uncertain; may involve dopamine transport inhibition | Reduces EDS [60] [55] | Lower abuse potential than amphetamines [55] |
Visualizing Screening Workflows and Neurobiological Pathways
HTS Screening and Validation Workflow
The following diagram illustrates the integrated stages of a high-throughput screening campaign for sleep-wake therapeutics, from initial cell line preparation to final in vivo validation.
Key Neurobiological Pathways in Sleep and Wakefulness
This diagram provides a simplified overview of the major neuronal systems and molecular targets involved in regulating sleep and wakefulness, highlighting key sites of action for hypnotic and wake-promoting drugs.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful HTS campaigns for sleep-wake compounds rely on a standardized set of research tools and reagents. The following table details key components of the screening toolkit.
Table 3: Essential Research Reagent Solutions for HTS in Sleep-Wake Drug Discovery
| Reagent / Material | Function and Application in HTS |
|---|---|
| Stable Cell Lines (e.g., CHO-T-Rex) | Engineered to consistently express the human target receptor (e.g., GABAA1 α1β2γ2L, OX2R). Provides a reproducible and scalable system for screening [57]. |
| FLIPR & Membrane Potential Dye Kits | Enables high-throughput, functional measurement of ion flux through activated receptors (e.g., Cl- flux for GABAA) in living cells. Higher detection sensitivity and throughput than electrophysiology [57]. |
| qHTS Compound Libraries | Curated collections of thousands to millions of small molecules used for primary screening. Libraries are formatted for low-volume, multi-concentration testing in 1536-well plates [56] [59]. |
| Tool Compounds (Agonists/Antagonists) | Pharmacologically characterized reference compounds (e.g., GABA, Diazepam, DPCPX) used for assay validation, as internal controls, and for determining baseline assay performance (Z'-factor) [57] [58]. |
| Automated Liquid Handling Systems | Robotics for precise, nanoliter-volume compound and reagent dispensing across hundreds of assay plates. Essential for reproducibility and throughput [56]. |
| Data Analysis Pipeline Software | Custom or commercial software for curve fitting, calculating activity parameters (AC50, wAUC), and flagging assay artifacts like autofluorescence and cytotoxicity [56] [59]. |
The application of high-throughput screening in the discovery of novel hypnotics and wake-promoting agents has fundamentally accelerated the identification and optimization of compounds that act on specific sleep-wake circuits. The field is moving beyond serendipitous discovery towards a rational, target-driven approach, powered by robust qHTS methodologies, sophisticated data analysis pipelines to mitigate artifacts, and validated in vitro and in vivo models. Future directions will likely involve greater integration of machine learning models to analyze complex multi-parameter HTS data and predict in vivo efficacy [61], increased use of zebrafish models for high-throughput in vivo phenotypic screening [62], and a continued focus on novel mechanisms beyond the monoamine and GABA systems, particularly the orexin and adenosine pathways. As these technologies mature, they hold the promise of delivering a new generation of safer, more effective, and more targeted therapies for the millions of patients affected by sleep and wake disorders.
The regulation of sleep and wakefulness represents one of the most complex neurobiological processes, involving intricate interactions between multiple neurotransmitter systems and receptor networks. The biochemical orchestration of sleep-wake states relies on a precise balance between arousal-promoting and sleep-promoting nuclei within distinct brain regions. Understanding these receptor-specific mechanisms is fundamental to developing targeted therapeutics for sleep disorders, which affect approximately one-third of the global population and pose significant challenges to public health systems worldwide [2]. The emerging paradigm in sleep medicine has shifted from non-specific sedation to targeted modulation of specific wake-promoting pathways, with receptor-specific agonists and antagonists playing a pivotal role in this therapeutic transformation.
The historical development of sleep therapeutics began with chance discoveries of compounds that induced sedation, primarily through potentiation of widespread inhibitory neurotransmission. Traditional medications like benzodiazepines and non-benzodiazepine "Z-drugs" enhanced gamma-aminobutyric acid (GABA)-ergic signaling, providing symptomatic relief for insomnia but with significant limitations including tolerance, dependence, cognitive impairment, and disruption of natural sleep architecture [63] [64]. The discovery of the orexin (hypocretin) system in 1998 marked a revolutionary advancement in sleep neurobiology, revealing a highly specific wake-promoting system that could be selectively modulated to treat insomnia without the drawbacks associated with GABAergic drugs [63] [65]. This breakthrough exemplified the potential of receptor-specific approaches in developing more precise and safer therapeutics for sleep disorders.
Major Receptor Systems in Sleep-Wake Regulation
GABAergic Receptor System
The GABAergic system serves as the primary inhibitory neurotransmitter pathway in the central nervous system and plays a dominant role in sleep induction and maintenance. GABA is synthesized from glutamate through the catalytic action of glutamic acid decarboxylase (GAD), which represents a critical biochemical pathway governing the balance between excitatory and inhibitory signaling in the brain [66]. The ventrolateral preoptic nucleus (VLPO) releases GABA to inhibit wake-promoting centers such as the tuberomammillary nucleus, locus coeruleus, and raphe nuclei, thereby facilitating the transition to sleep [66].
GABA mediates its effects primarily through ionotropic GABAA receptors and metabotropic GABAB receptors. Upon binding to GABAA receptors, GABA triggers the influx of chloride ions (Cl-) into the postsynaptic neuron, causing neuronal hyperpolarization and reduced excitability [66]. This mechanism is particularly crucial for non-REM sleep generation. Pharmacological agents that potentiate GABAergic transmission include benzodiazepines, zolpidem, and barbiturates, which act as positive allosteric modulators of GABAA receptors [66]. While effective for sleep induction, these compounds lack specificity for sleep-related neural circuits and consequently produce side effects including cognitive impairment, tolerance, and risk of dependence [63].
Recent research has revealed complex interactions between the GABAergic system and other regulatory pathways. For instance, orexin-A has been demonstrated to inhibit GABA currents in spinal ventral horn neurons via both OX1R and OX2R receptors through a non-Ca2+-dependent PKC signaling pathway [67]. This interaction highlights the sophisticated cross-talk between wake-promoting and sleep-promoting systems and suggests potential mechanisms for fine-tuning sleep-related motor activity.
Orexin Receptor System
The orexin system has emerged as a cornerstone in the neurobiological understanding of wake regulation and a prime target for novel insomnia therapeutics. Orexin-A and orexin-B (also known as hypocretin-1 and hypocretin-2) are neuropeptides produced predominantly by neurons in the lateral hypothalamus that mediate their physiological effects through two G-protein-coupled receptors: orexin receptor type 1 (OX1R) and orexin receptor type 2 (OX2R) [63]. These receptors are widely distributed in brain regions implicated in arousal and vigilance, including the locus coeruleus, dorsal raphe nuclei, thalamus, and cerebral cortex [63].
The two orexin receptors exhibit differential functions in sleep and arousal regulation. OX2R is widely recognized as the primary mediator of sleep-wake stability through interactions with ventrolateral preoptic nucleus (VLPO) neurons and other sleep-active regions, while OX1R appears to play a more significant role in regulating reward, stress, and emotional processes [63] [68]. Dysregulation within the orexin system, especially persistent overactivation of orexin signaling, has been increasingly recognized as a contributing factor in chronic insomnia, while orexin deficiency is well-established as the causative factor in narcolepsy type 1 [63].
Dual orexin receptor antagonists (DORAs) represent a mechanistically distinct approach to insomnia treatment that functions by selectively dampening orexin-driven arousal, thereby facilitating sleep onset and maintenance without disrupting natural sleep architecture [63]. Unlike traditional sedative-hypnotics, DORAs promote sleep by temporarily reducing wake drive rather than by inducing widespread neuronal inhibition, resulting in a sleep pattern that more closely resembles physiological sleep [63].
Table 1: Clinically Approved Dual Orexin Receptor Antagonists (DORAs)
| Compound | Elimination Half-life | Primary Indications | Key Clinical Characteristics |
|---|---|---|---|
| Suvorexant | 12-16 hours | Insomnia (sleep onset and maintenance) | First FDA-approved DORA; moderate duration of action |
| Lemborexant | 17-19 hours (mean) | Insomnia (sleep onset and maintenance) | Demonstrated efficacy in elderly patients and those with Alzheimer's disease |
| Daridorexant | 8-9 hours | Insomnia (sleep onset and maintenance) | Balanced profile with minimal next-day residual effects |
| Vornorexant* | Notably short (comparable to zolpidem) | Insomnia (primarily sleep onset) | Investigational agent with shortest half-life among DORAs |
*Investigational compound not yet approved [69]
Melatonin Receptor System
The melatonin receptor system regulates circadian rhythms and helps signal the transition to sleep. Melatonin, a hormone secreted by the pineal gland, is synthesized from serotonin in a process regulated by the light-dark cycle [66]. It exerts its effects primarily through two G-protein-coupled receptors: MT1 and MT2. MT1 receptors mediate sleep onset and blood pressure regulation, while MT2 receptors are involved in phase-shifting effects and regulation of circadian rhythms [64].
Novel melatonin receptor agonists are being developed with enhanced receptor selectivity for MT1 and MT2 receptors, often incorporating prolonged-release mechanisms to mimic the physiological release of melatonin throughout the night [64]. This approach supports both sleep initiation and maintenance while maintaining circadian alignment. Additionally, research indicates that melatonin enhances the effect of GABA by increasing the expression and sensitivity of GABAA receptors, particularly in the hypothalamus, demonstrating integrative functions across neurotransmitter systems [66].
Adenosinergic Receptor System
Adenosine functions as a homeostatic sleep factor that accumulates during prolonged wakefulness as a byproduct of ATP metabolism [66]. During periods of prolonged neuronal activity or metabolic demand, ATP is hydrolyzed to AMP, which is subsequently converted into adenosine. As adenosine accumulates extracellularly, it binds to A1 receptors, especially in the basal forebrain, inhibiting wake-promoting neurons and promoting sleep onset [66].
The adenosinergic system provides a biochemical representation of sleep pressure, with increasing extracellular adenosine concentrations during wakefulness creating a homeostatic drive for sleep. Caffeine, a competitive antagonist of adenosine receptors, delays sleep onset and reduces total sleep time, highlighting adenosine's critical sleep-promoting role [66]. Research has shown that infusion of adenosine or A2A receptor agonists promotes NREM sleep, whereas A1 receptor knockout mice exhibit reduced sleep pressure [66].
Molecular Characterization of Drug-Receptor Interactions
Computational Analysis of Orexin Receptor Antagonists
Advanced computational methods have provided detailed insights into the molecular interactions governing drug-receptor binding. Recent studies investigating three OXR1 antagonists—daridorexant, lemborexant, and suvorexant—utilized an integrated approach combining molecular dynamics (MD) simulations, density functional theory (DFT) calculations, and the molecular fractionation with conjugate caps (MFCC) methodology to quantify interaction energies between ligands and key receptor residues [68].
The MFCC approach enabled precise quantification of interaction energies, revealing that residues such as GLU204, HIS216, and ASN318 play critical roles in stabilizing ligand-receptor interactions [68]. Daridorexant exhibited the strongest interaction energy, driven by hydrogen bonds and hydrophobic contacts, while lemborexant and suvorexant showed distinct stabilization patterns mediated by hydrophobic interactions [68]. These findings provide a robust molecular basis for the rational design of next-generation OXR1 antagonists with improved efficacy and safety profiles.
Quantum chemical calculations revealed that daridorexant and suvorexant had similar total energies (-1926.72 Ha and -1926.70 Ha, respectively), while lemborexant showed a different energy profile (-1503.62 Ha) [68]. The calculated HOMO-LUMO energy gaps were 4.86721 eV for daridorexant, 5.42455 eV for lemborexant, and 4.47390 eV for suvorexant, indicating differences in electronic stability and reactivity that may influence their pharmacological profiles [68].
Table 2: Quantum Chemical Properties of Orexin Receptor Antagonists
| Property | Daridorexant | Lemborexant | Suvorexant |
|---|---|---|---|
| Total Energy (Hartree) | -1926.72 | -1503.62 | -1926.70 |
| Binding Energy (Hartree) | -111.83 | -97.52 | -111.82 |
| Dipole Moment (Debye) | 3.81 | 3.58 | 3.30 |
| Cavity Volume (ų) | 3127.75 | 2977.15 | 3181.28 |
| HOMO-LUMO Gap (eV) | 4.87 | 5.42 | 4.47 |
| Surface Area (Ų) | 1384.73 | 1445.00 | 1475.70 |
Structural Dynamics of Receptor-Ligand Complexes
Molecular dynamics simulations have elucidated the dynamic behavior of orexin receptor-ligand complexes, providing insights into the conformational changes associated with receptor antagonism. These simulations have demonstrated that daridorexant, lemborexant, and suvorexant exhibit distinct binding modes and stabilization patterns within the orthosteric binding pocket of OX1R [68].
Daridorexant exhibited fifteen unique conformations during conformational space analysis, indicating moderate structural flexibility, while lemborexant presented nine distinct conformations, suggesting slightly lower flexibility. In contrast, suvorexant exhibited only a single conformation, highlighting its structural rigidity [68]. The radius of gyration was measured at 4.20 Å for daridorexant and 4.44 Å for lemborexant, indicating a slightly more elongated structure for the latter [68]. These structural differences contribute to variations in receptor binding kinetics and duration of action observed clinically.
Experimental Methodologies for Receptor Characterization
Electrophysiological Approaches
Electrophysiological techniques provide direct assessment of receptor function and modulation in neuronal systems. The whole-cell patch-clamp recording technique has been instrumental in characterizing the effects of neuropeptides and receptor ligands on ion channel function. A detailed methodology for investigating orexin-A effects on GABA currents in spinal ventral horn neurons illustrates this approach [67].
Protocol: Patch-Clamp Recording of GABA Currents
- Tissue Preparation: Select 7-12 day old neonatal Sprague-Dawley rats. Following anesthesia, isolate spinal cord segments containing lumbosacral enlargement and prepare 400-500 µm thick slices using a vibratome.
- Enzyme Digestion: Incubate slices with papain (0.18 g/30 mL artificial cerebrospinal fluid) for 40-60 minutes at 33°C to dissociate neurons.
- Mechanical Dissociation: Under microscope, retain ventral horn tissue and use polished Pasteur pipettes of different diameters to acutely mechanically separate individual neurons.
- Solution Preparation: Prepare artificial cerebrospinal fluid (ACSF) containing (in mmol/L): 124.0 NaCl, 24.0 NaHCO3, 5.0 KCl, 1.2 KH2PO4, 2.4 CaCl2, 1.3 MgSO4, 10.0 Glucose, oxygenated with 95% O2 + 5% CO2 to pH 7.35-7.45.
- Electrode Internal Solution: Prepare solution containing (in mmol/L): 0.5 EGTA, 120.0 K-gluconate, 20.0 KCl, 2.0 MgCl2, 20.0 HEPES, 2.0 Na2-ATP, 0.5 Na-GTP, adjusted to pH 7.26 with KOH.
- Recording Setup: Select neurons with large cell bodies, three-dimensional transparency, and multiple intact processes. Use perforated patch-clamp recording mode with amphotericin B as the perforating agent.
- Drug Application: Apply GABA receptor agonist γ-aminobutyric acid to evoke currents in spinal ventral horn neurons. Apply orexin-A to observe its modulatory effect on GABA currents.
- Pharmacological Analysis: Use receptor-selective antagonists including OX1R antagonist SB334867 (10 µmol/L), OX2R antagonist TCSOX229 (10 µmol/L), PKC inhibitor Bisindolylmaleimide IV (Bis-IV), PKC activator PMA, PKA inhibitor Rp-cAMP, and Ca2+ chelator BAPTA-AM to elucidate signaling mechanisms [67].
This methodology demonstrated that orexin-A significantly inhibits GABA-induced current amplitude in spinal ventral horn neurons by 67.48±12.50%, an effect mediated by both OX1R and OX2R through a non-Ca2+-dependent PKC signaling pathway [67].
Molecular Dynamics Simulation Protocols
Molecular dynamics simulations provide atomic-level insights into receptor-ligand interactions and dynamics. The following protocol outlines the integrated computational approach used to characterize orexin receptor antagonists [68]:
Protocol: Molecular Dynamics Simulation of Receptor-Ligand Complexes
- Ligand Preparation: Obtain three-dimensional structures of daridorexant, lemborexant, and suvorexant from PubChem database. Perform initial geometry optimization using Dreiding-like force field.
- Energy Minimization: Conduct energy minimization with CHARMm force field, version c44b2 in BIOVIA Discovery Studio, version 2022. Apply SmartMinimizer algorithm in solvent environment with distance-dependent dielectric.
- Conformational Analysis: Analyze conformational space using root-mean-square deviation (RMSD) calculations. For daridorexant, RMSD values range from 0.21 Å to 4.52 Å (mean: 2.71 Å) across fifteen conformations.
- Quantum Mechanical Optimization: Subject the most stable conformer of each ligand to quantum mechanical optimization using density functional theory (DFT) with generalized gradient approximation (GGA) exchange-correlation functional PBE and the 6-311++G basis set.
- System Preparation: Embed optimized ligand-receptor complexes in lipid bilayer mimicking neuronal membrane environment. Solvate system with TIP3P water model and neutralize with appropriate ions.
- Simulation Parameters: Perform molecular dynamics simulations using AMBER or CHARMM force fields. Maintain constant temperature (310 K) and pressure (1 atm) using Langevin dynamics and Berendsen barostat.
- Trajectory Analysis: Analyze 100-200 ns production trajectories using root-mean-square deviation (RMSD), root-mean-square fluctuation (RMSF), and interaction energy calculations via MFCC methodology.
- Binding Energy Calculations: Employ molecular mechanics Poisson-Boltzmann surface area (MM-PBSA) or molecular mechanics generalized Born surface area (MM-GBSA) methods to calculate binding free energies.
This integrated computational approach has revealed critical residues governing antagonist binding to OX1R and provided insights into the structural determinants of drug efficacy and duration of action [68].
Signaling Pathways and Neurotransmitter Interactions
The following diagram illustrates the core signaling pathways of major receptor systems involved in sleep-wake regulation and their key interactions:
Sleep-Wake Regulation Pathways: This diagram illustrates the core receptor systems governing sleep-wake states, highlighting targeted therapeutic interventions. Wake-promoting pathways (yellow) centered on orexin signaling activate multiple arousal systems, while sleep-promoting pathways (blue) involve GABAergic, adenosinergic, and melatonergic systems. Pharmacological interventions (red) selectively modulate these pathways: DORAs inhibit orexin signaling, GABAA drugs potentiate inhibitory neurotransmission, and melatonin agonists mimic circadian sleep signals. Critical interactions include adenosine enhancement of GABAergic tone and melatonin potentiation of GABA effects, demonstrating the integrated nature of sleep-wake regulation.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Sleep Receptor Research
| Reagent/Category | Specific Examples | Research Application | Mechanistic Insight |
|---|---|---|---|
| Selective Receptor Antagonists | SB334867 (OX1R), TCSOX229 (OX2R) | Receptor subtype pharmacology; pathway dissection | Elucidated orexin receptor subtype contributions to GABA current modulation [67] |
| Signal Transduction Modulators | Bisindolylmaleimide IV (PKC inhibitor), Rp-cAMP (PKA inhibitor) | Intracellular signaling pathway analysis | Identified PKC-dependent, Ca2+-independent mechanism of orexin-A effects on GABA currents [67] |
| Ion Channel Modulators | BAPTA-AM (Ca2+ chelator), Tetrodotoxin (Na+ channel blocker) | Ion flux measurement; neuronal excitability studies | Determined calcium dependence in receptor signaling pathways [67] |
| Computational Chemistry Tools | CHARMm/AMBER force fields, DFT calculations, MFCC methodology | Molecular dynamics simulations; binding energy calculations | Quantified interaction energies between DORAs and OX1R residues (GLU204, HIS216, ASN318) [68] |
| Electrophysiology Systems | Patch-clamp amplifiers, Microelectrode arrays | Neuronal circuit activity mapping; receptor function assessment | Measured orexin-A inhibition of GABA currents (67.48±12.50%) in spinal ventral horn neurons [67] |
Therapeutic Applications and Clinical Translation
Insomnia Therapeutics
The development of receptor-specific agonists and antagonists has fundamentally transformed the therapeutic landscape for insomnia. Dual orexin receptor antagonists represent the most significant advancement in insomnia pharmacotherapy in decades, offering targeted suppression of wake drive without widespread neuronal depression [69] [63]. Clinical trials have consistently demonstrated that DORAs significantly reduce sleep onset latency and improve sleep maintenance while preserving natural sleep architecture [63].
The emerging spectrum of DORAs with varying pharmacokinetic profiles enables more personalized treatment approaches aligned with patients' specific sleep complaints and metabolic characteristics [69]. For instance, the development of vornorexant, with a notably short half-life comparable to zolpidem, may address needs for sleep initiation without next-day residual effects, while longer-acting agents like lemborexant provide sustained efficacy for sleep maintenance difficulties [69].
Clinical trials extending up to one year in duration have demonstrated sustained efficacy of DORAs with favorable safety profiles. Nearly 60% of both patients and clinicians reported improvements in insomnia disorder with DORAs, with no instances of discontinuation due to treatment-related adverse events in some studies [63]. The low abuse potential, minimal risk of rebound effects, and limited impact on respiratory and cardiovascular function render DORAs particularly suitable for older adults and those with complex pharmacological regimens [63].
Implications for Neurodegenerative Disorders
Research has revealed compelling connections between sleep disruption and neurodegenerative diseases, particularly Alzheimer's disease (AD), creating new therapeutic opportunities for receptor-specific agents. The bidirectional relationship between sleep and AD pathology suggests that sleep disturbances may contribute to disease progression through impaired clearance of amyloid-β and tau proteins [70].
Dual orexin receptor antagonists show promise for mitigating AD sleep disturbances and potentially enhancing clearance of pathological protein aggregates [70]. Research in APP/PS1 mouse models of Alzheimer's disease has demonstrated that DORAs can improve sleep quality and restore diurnal fluctuations in interstitial fluid amyloid-β, suggesting potential disease-modifying effects beyond symptomatic treatment of sleep disturbances [70].
The therapeutic potential of DORAs in neurodegenerative conditions extends beyond amyloid pathology. Orexin receptors influence a broad range of neurobiological processes including dopaminergic, serotonergic, noradrenergic, and glutamatergic pathways, all of which are commonly dysregulated in neurodegenerative and mood disorders [63]. Suppression of orexin input to the locus coeruleus may reduce excessive noradrenergic tone, a key factor in the hyperarousal observed in both chronic insomnia and neurodegenerative diseases [63].
Future Directions and Emerging Targets
The future of receptor-specific sleep therapeutics includes several promising directions. Next-generation DORAs are being designed with optimized pharmacokinetic profiles to enhance efficacy for specific insomnia phenotypes while minimizing next-day residual effects [69] [64]. Additionally, novel multi-mechanism approaches are emerging, including compounds that combine orexin receptor antagonism with modulation of other neurotransmitter systems involved in anxiety and wakefulness [64].
Beyond the orexin system, researchers are exploring novel targets including circadian clock proteins (CLOCK, BMAL1, PERIOD genes), histamine H3 receptors, and adenosine A2A receptors [64]. The growing recognition of neuroinflammation in sleep disturbances has stimulated investigation of anti-inflammatory and immunomodulatory approaches for treatment-resistant insomnia [64].
The emerging understanding of the glymphatic system's role in clearing metabolic waste during sleep represents another frontier for therapeutic development [70]. While recent studies have questioned some aspects of glymphatic function during sleep, the fundamental relationship between sleep and clearance of neurotoxic proteins like amyloid-β remains well-established [70]. Therapeutic strategies that enhance this clearance function could have profound implications for preventing or slowing neurodegenerative disease progression.
Personalized sleep medicine represents the ultimate frontier, with pharmacogenomic approaches potentially enabling matching of specific receptor-targeted therapies to individual patients based on their genetic profile, sleep phenotype, and comorbid conditions [69]. As our understanding of the biochemical underpinnings of sleep and wakefulness continues to expand, so too will our ability to develop precisely targeted therapeutics that restore natural sleep without compromising daytime functioning or long-term health.
Circadian rhythms are intrinsic, approximately 24-hour biological cycles governed by a molecular clock within virtually every cell, orchestrating physiological processes from gene expression to behavior. These rhythms are generated by cell-autonomous programs of gene expression and are synchronized to environmental cues, primarily light, by the suprachiasmatic nucleus (SCN) in the hypothalamus, which acts as the master pacemaker [71]. The fundamental properties of circadian rhythms include their persistence in absence of external cues (self-sustenance), ability to entrain to environmental signals, stable periodicity under varying temperatures, and a period that closely mirrors the 24-hour solar day [71]. Understanding these rhythms has unveiled significant therapeutic potential, as nearly 50% of mammalian genes are expressed with 24-hour rhythms, and disruption of circadian timing is linked to numerous pathological conditions including cardiovascular, metabolic, and psychological disorders [72] [71]. Chronotherapy capitalizes on this temporal organization by aligning drug delivery with the body's endogenous rhythms to maximize efficacy and minimize adverse effects.
Molecular Mechanisms of the Circadian Clock
The circadian clock operates through interconnected transcriptional-translational feedback loops (TTFLs) at the cellular level. The core molecular machinery involves several key components:
- Transcriptional Activators: CLOCK and BMAL1 proteins form a heterodimer that activates transcription of period (PER) and cryptochrome (CRY) genes by binding to E-box enhancer elements [73].
- Transcriptional Repressors: PER and CRY proteins accumulate, form complexes, translocate to the nucleus, and inhibit CLOCK-BMAL1 activity, completing the primary negative feedback loop [73].
- Stabilizing Loops: Nuclear receptors REV-ERBα/β and RORα/β/γ form accessory loops that stabilize the core oscillator and regulate BMAL1 expression [73].
This molecular clock exists not only in the SCN but in virtually all peripheral tissues, where it governs local physiology and is synchronized by the SCN through neural and humoral signals [73]. The circadian system regulates diverse processes including sleep-wake cycles, hormone secretion, metabolism, immune function, and cellular proliferation, making it a critical target for therapeutic intervention [73].
The following diagram illustrates the core transcriptional-translational feedback loop of the molecular circadian clock:
Figure 1: Core Circadian Clock Feedback Loop. The molecular clock is driven by transcriptional-translational feedback loops. CLOCK:BMAL1 activates PER/CRY expression, whose proteins then suppress CLOCK:BMAL1 activity. Accessory loops involving nuclear receptors provide stability [73] [72].
Current Chronotherapeutic Drug Delivery Platforms
Multi-Pulse Drug Delivery Systems
Multi-pulse delivery systems provide controlled, pulsatile release mimicking natural body rhythms through advanced technologies. These systems employ stimuli-responsive mechanisms, artificial intelligence, and nanotechnology to achieve temporal control over drug release [74]. They are particularly valuable for chronic disease management where multiple daily dosing is required but patient compliance is challenging. By aligning drug plasma concentrations with circadian patterns of disease symptoms, these systems significantly enhance therapeutic efficacy while reducing side effects [74]. The integration of AI with personalized medicine enables customization of drug delivery profiles based on individual circadian patterns, further optimizing treatment outcomes [74].
Nanomaterial-Enabled Delivery Systems
Nanotechnology offers sophisticated solutions for circadian medicine through various material platforms:
- Liposomes: Spherical vesicles with phospholipid bilayers capable of encapsulating both hydrophilic and hydrophobic drugs, providing sustained release profiles.
- Polymeric Nanoparticles (PNPs): Biodegradable polymers that can be engineered for precise temporal drug release and targeted delivery to specific tissues [73].
- Mesoporous Silica Nanoparticles: High surface area materials with tunable pore sizes for controlled drug loading and release kinetics [73].
These nanoplatforms address key challenges in chronotherapy by enabling sustained release, targeted delivery to relevant tissues (such as the SCN or peripheral organs), and overcoming biological barriers [73]. Smart drug delivery systems (SDDSs) that respond to physiological cues such as temperature or pH changes represent particularly promising approaches for circadian-informed therapies [73].
Table 1: Nanomaterial Platforms for Chronotherapeutic Drug Delivery
| Material Type | Key Properties | Chronotherapeutic Advantages | Potential Applications |
|---|---|---|---|
| Liposomes | Phospholipid bilayers, biocompatible | Encapsulate diverse drug types, sustained release | Hormone therapy, anti-inflammatories |
| Polymeric Nanoparticles (PNPs) | Tunable degradation rates, surface functionalization | Precise temporal control, targeted delivery | Cancer therapy, metabolic disorders |
| Mesoporous Silica Nanoparticles | High surface area, uniform pore size | Controlled loading and release kinetics | Neurological disorders, inflammation |
| Stimuli-Responsive Systems | Respond to pH, temperature, or enzymes | Autonomous release aligned with physiological cues | Inflammatory conditions, cancer |
Chronogenetic Circuits for Programmable Drug Delivery
The emerging field of chronogenetics combines synthetic biology and tissue engineering to create autonomous, cell-based therapies that naturally align with circadian biology. Researchers have developed synthetic gene circuits that interface with core clock components to drive rhythmic production of therapeutic proteins [72] [75]. One pioneering approach utilized the Period2 (Per2) promoter to express interleukin-1 receptor antagonist (IL-1Ra) as an anti-inflammatory therapy for rheumatoid arthritis [72]. These circuits maintained robust circadian oscillations in vitro and in vivo, with a period of approximately 23.4 hours, and produced therapeutic levels of IL-1Ra with distinct daily rhythms, demonstrating approximately 2-fold changes between peak and trough protein abundance [72]. Subsequent advancements created programmable circuits using different circadian response elements (E'-boxes, D-boxes, RREs) to target three distinct phases within the daily period, enabling customized timing of therapeutic production [75].
The experimental workflow for developing and testing chronogenetic circuits involves:
Figure 2: Chronogenetic Circuit Development Workflow. Experimental pipeline for creating cell-based chronotherapies, from genetic circuit design to in vivo validation [72] [75].
Experimental Protocols and Methodologies
Development and Characterization of Chronogenetic Circuits
Circuit Construction and Lentiviral Transduction:
- Amplify circadian promoter elements (e.g., Per2 promoter, E'-boxes, D-boxes, RREs) via PCR and clone into lentiviral vectors upstream of therapeutic transgene (e.g., IL-1Ra) and reporter genes (e.g., luciferase) using Gibson Assembly [72] [75].
- Package lentiviral particles in HEK293T cells using standard packaging plasmids (psPAX2, pMD2.G) and concentrate via ultracentrifugation [72].
- Transduce murine induced pluripotent stem cells (miPSCs) at MOI 10-20 in the presence of 8 μg/mL polybrene, then select stable lines with appropriate antibiotics [72].
In Vitro Characterization of Circadian Rhythms:
- Differentiate transduced miPSCs into tissue-engineered cartilage pellets using TGF-β3-containing chondrogenic medium for 21-28 days [72] [75].
- Analyze circadian oscillations by continuous bioluminescence imaging (BLI) using cooled CCD cameras in time-lapse mode over 60-72 hours in sealed light-tight chambers [72].
- Quantify rhythm parameters (period, amplitude, phase) using Lumicycle Analysis or similar software with baseline subtraction and damped cosine curve fitting [72].
- Measure therapeutic protein production via ELISA of culture media collected every 3-6 hours over 72 hours; determine synthesis rates accounting for protein accumulation [72].
Assessment of Therapeutic Efficacy in Disease Models
Inflammatory Challenge and Protection Assays:
- Treat chronogenetic cartilage constructs with 10 ng/mL IL-1β to simulate inflammatory arthritis environment [72] [75].
- Assess circadian rhythm robustness by comparing peak-trough amplitude ratios 24 hours pre- and post-cytokine treatment using bioluminescence tracking [72].
- Evaluate cartilage protection via sulfated glycosaminoglycan (sGAG) content measurement using DMMB assay and collagen type II immunolabeling of sectioned constructs [72] [75].
- Quantify inflammatory damage by measuring NO release (Griess reagent) and PGE2 production (ELISA) in media [75].
In Vivo Implantation and Entrainment Studies:
- Implant engineered cartilage constructs subcutaneously in immunocompromised mice (e.g., C57BL/6 J) [72].
- Monitor in vivo circadian rhythms and entrainment to host via bioluminescence imaging for 36+ hours beginning 3 days post-implantation [72].
- Measure serum drug concentrations at multiple timepoints to verify circadian oscillations in therapeutic delivery [72].
- Assess functional outcomes in disease models (e.g., arthritis severity, joint inflammation) relative to control groups [72].
Quantitative Data and Research Findings
Table 2: Experimental Results from Chronogenetic Circuit Studies
| Parameter | In Vitro Performance | In Vivo Performance | Therapeutic Outcome |
|---|---|---|---|
| Circadian Period | 23.4 ± 0.35 hours [72] | Maintained circadian rhythms post-implantation [72] | N/A |
| IL-1Ra Production Rhythm | 22.5 ± 2.9 hour period, 2-fold peak-trough difference [72] | Serum concentrations showed circadian variation [72] | Protected cartilage from inflammatory damage [75] |
| Amplitude Stability | Maintained under IL-1 challenge (amplitude ratio similar to controls) [72] | Entrained to host light-dark cycles [72] | Resisted cytokine-induced circadian dysregulation [72] [75] |
| Phase Targeting Capability | Distinct phases achievable with E'-box, D-box, RRE elements [75] | Phase-locked to host SCN rhythms [72] | Therapeutic delivery aligned with symptom peaks (e.g., morning inflammation) [72] |
| Treatment Efficiency | N/A | N/A | 38% faster orthodontic tooth movement with circadian-force synchronization [76] |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Chronobiology and Chronotherapy Research
| Reagent/Category | Specific Examples | Research Application |
|---|---|---|
| Circadian Reporters | Per2-Luc, PER2::LUCIFERASE | Real-time monitoring of circadian rhythms in live cells and tissues [72] |
| Gene Circuit Components | E'-box, D-box, RRE promoter elements | Building chronogenetic circuits for phased therapeutic expression [75] |
| Delivery Vectors | Lentiviral vectors (psPAX2, pMD2.G) | Stable transduction of stem cells for engineered tissue therapies [72] [75] |
| Stem Cell Differentiation | TGF-β3, chondrogenic medium | Generation of tissue-engineered cartilage for implantation studies [72] [75] |
| Circadian Monitoring | Cooled CCD cameras, Lumicycle software | Continuous bioluminescence imaging and rhythm analysis [72] |
| Inflammatory Agents | IL-1β (10 ng/mL) | Simulating inflammatory disease environment for efficacy testing [72] [75] |
| Cartilage Matrix Assays | DMMB assay, collagen type II antibodies | Quantifying tissue health and protection from inflammatory damage [72] [75] |
Future Perspectives and Research Directions
The field of chronotherapeutic drug delivery is rapidly evolving toward increasingly sophisticated and personalized approaches. Several promising directions are emerging:
- Personalized Chronogenetic Circuits: Advanced synthetic biology approaches will enable creation of gene circuits that can be customized to an individual's circadian phenotype, potentially incorporating multiple therapeutic transgenes under control of different circadian phases for complex conditions [75].
- Closed-Loop Smart Delivery Systems: Integration of wearable technology that continuously monitors circadian biomarkers with automated drug delivery systems could create responsive therapeutic platforms that dynamically adjust dosing timing based on real-time physiological readouts [77].
- Nanomaterial Innovations: Next-generation stimuli-responsive nanomaterials that react to multiple circadian-linked physiological changes (melatonin levels, core body temperature, hormone concentrations) will enable more autonomous and precise chronotherapeutic delivery [73].
- Clinical Translation Challenges: Future research must address manufacturing scalability, long-term safety of implanted engineered cells, and development of circadian phenotyping protocols for treatment personalization [78] [72]. The clinical trial landscape faces challenges including regulatory complexities, diversity in participant recruitment, and the need for novel trial designs that account for circadian timing [78].
The integration of chronotherapeutic approaches into mainstream medicine holds significant promise for optimizing drug efficacy and safety across numerous therapeutic areas, fundamentally changing how treatments are temporally aligned with human biology.
Mechanisms of Sleep Pathologies and Intervention Strategies
Insomnia disorder is a prevalent condition characterized by chronic dissatisfaction with sleep quantity or quality, associated with significant daytime impairment and an elevated risk for numerous comorbid physical and mental health conditions [79]. The pathophysiology of insomnia is complex, but contemporary research has converged on several core neurobiological dysfunctions. This whitepaper elucidates the principal mechanisms underpinning chronic insomnia, focusing on the triad of hyperarousal, dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis, and deficits in GABAergic neurotransmission. These interconnected pathways form a self-reinforcing cycle that perpetuates sleep disruption and informs the development of novel therapeutic strategies. Understanding these mechanisms at a biochemical and circuit level is paramount for advancing targeted drug development in the field of sleep disorders.
Core Pathophysiological Mechanisms
The Hyperarousal Model
Hyperarousal is an overarching theme in insomnia pathophysiology, manifesting as heightened physiologic, cognitive, and cortical activation that interferes with the natural disengagement from the environment necessary for sleep onset and maintenance [79]. This state is not merely psychological but is detectable through objective measures.
- Neural Hyperarousal: Electroencephalographic (EEG) studies consistently show increased high-frequency beta and gamma activity in individuals with insomnia during both wakefulness and sleep, indicating a state of cortical hyperactivation [79]. This fast brainwave activity is antithetical to the synchronous, slow waves characteristic of restorative sleep.
- Cognitive and Affective Hyperarousal: Patients often report an inability to quiet the mind, with racing thoughts, worry, and rumination preventing sleep initiation. This cognitive arousal is frequently coupled with emotional hyperarousal, including heightened anxiety [80].
- Systemic Manifestations: The hyperarousal state extends beyond the central nervous system, often presenting as increased sympathetic nervous system tone and autonomic dysregulation [79].
HPA Axis Dysregulation
The HPA axis, the body's central stress response system, exhibits significant dysregulation in chronic insomnia. Under normal conditions, the diurnal rhythm of cortisol, the primary glucocorticoid, peaks in the morning and reaches a nadir at night, supporting the sleep-wake cycle. In insomnia, this rhythm is often flattened or phase-shifted.
- Elevated Nocturnal Cortisol: A hallmark of insomnia is the failure to suppress HPA axis activity at night. Elevated levels of cortisol and CRH (corticotropin-releasing hormone) during the usual sleep period contribute to the state of hyperarousal, directly interfering with sleep-promoting circuits [80].
- Bidirectional Reinforcement: The relationship between insomnia and HPA axis dysfunction is cyclical. While HPA hyperactivity disrupts sleep, the resulting sleep fragmentation and perceived stress of sleeplessness further activate the stress system, creating a vicious cycle [81] [80].
- Link to Comorbidities: Chronic HPA axis dysregulation and elevated cortisol levels are implicated in the well-established associations between insomnia and mood disorders, cognitive impairment, and metabolic diseases [50].
GABAergic Deficits
Gamma-aminobutyric acid (GABA) is the primary inhibitory neurotransmitter in the central nervous system and is crucial for promoting and maintaining sleep by countering the activity of wake-promoting systems.
- Reduced GABAergic Inhibition: A core deficit in GABAergic inhibition is believed to contribute significantly to the persistent hyperexcitability in neural circuits governing both sleep and anxiety [80]. Magnetic resonance spectroscopy (MRS) studies have reported decreased GABA levels in the brain of individuals with insomnia, particularly in the anterior cingulate and occipital cortex [79].
- Dysfunctional Sensory Gating: GABAergic interneurons, especially those containing parvalbumin, are critical for filtering sensory information and preventing hyperarousal. Their impaired function may lead to an increased awareness of environmental stimuli during sleep, contributing to the phenomenon of sleep state misperception [82].
- Interaction with the HPA Axis: GABA normally exerts an inhibitory tone on the HPA axis. Therefore, a deficit in GABAergic signaling can disinhibit HPA axis activity, leading to elevated cortisol release, which in turn can further disrupt GABA receptor function [83].
Table 1: Key Neurobiological Abnormalities in Insomnia
| Domain of Dysfunction | Key Findings | Technical/Methodological Evidence |
|---|---|---|
| Cortical Hyperarousal | Increased high-frequency (beta/gamma) EEG power during NREM sleep; altered default mode and salience network connectivity. | Quantitative EEG (qEEG); resting-state functional MRI (fMRI) [79]. |
| Autonomic Dysregulation | Increased heart rate, reduced heart rate variability, elevated sympathetic nervous system tone. | Polysomnography (PSG) with ECG analysis; heart rate variability (HRV) metrics [79]. |
| HPA Axis Dysregulation | Elevated nocturnal cortisol and CRH levels; flattened diurnal cortisol slope. | Radioimmunoassay/ELISA of serial saliva, plasma, or urine samples [80]. |
| GABAergic Deficit | Reduced GABA levels in cortical and limbic areas; altered GABA-A receptor density/function. | Magnetic Resonance Spectroscopy (MRS); positron emission tomography (PET) [79] [80]. |
Experimental Evidence and Quantitative Data
Research into the biomarkers of insomnia has yielded quantitative data that substantiates the core pathophysiological models. These findings not only provide objective measures for diagnosis but also point to underlying biochemical disturbances.
Metabolomic studies, which analyze the complete set of small-molecule metabolites, have revealed distinct profiles associated with poor sleep quality. For instance, a study analyzing salivary metabolites identified five metabolites—including glycerol and hippuric acid—that were significantly decreased in participants with poor sleep, while eight others, such as 2-hydroxybutyric acid (2HB), were increased. A random forest model using a panel of six salivary metabolites achieved a prediction accuracy of 0.866 for sleep quality [50]. This suggests that chronic sleep disturbance leaves a detectable metabolic signature, potentially related to disrupted energy metabolism.
Furthermore, proteomic analyses using Mendelian randomization have identified specific plasma proteins causally linked to insomnia risk. Elevated levels of TGFBI (Transforming Growth Factor Beta Induced) and PAM (Peptidylglycine Alpha-Amidating Monooxygenase) are associated with an increased risk of insomnia. The latter is particularly notable as PAM is an enzyme essential for the activation of numerous neuropeptides, suggesting a potential novel pathway for therapeutic intervention [84].
Table 2: Select Biomarkers Associated with Insomnia and Sleep Deprivation
| Biomarker Class | Specific Example | Change in Insomnia/Poor Sleep | Proposed Functional Implication |
|---|---|---|---|
| Metabolite | Glycerol [50] | Decreased | Potential indicator of altered central energy metabolism. |
| Metabolite | Hippuric Acid [50] | Decreased | Gut microbiome-derived compound; link to gut-brain axis. |
| Metabolite | 2-Hydroxybutyric Acid (2HB) [50] | Increased | Marker of oxidative stress and impaired energy metabolism. |
| Protein | PAM (Peptidylglycine Alpha-Amidating Monooxygenase) [84] | Increased | Disruption in neuropeptide amidation and signaling. |
| Protein | TGFBI (Transforming Growth Factor Beta Induced) [84] | Increased | Possible role in extracellular matrix and neural function. |
| Neuroendocrine | Nocturnal Cortisol [80] | Increased | HPA axis hyperarousal, direct inhibition of sleep-promoting circuits. |
Key Experimental Protocols and Methodologies
Understanding the evidence for these mechanisms requires insight into the experimental protocols used in both clinical and preclinical research.
Clinical Metabolomic Profiling
Objective: To identify non-invasive biomarkers for chronic sleep disorders by comparing metabolic profiles in saliva from individuals with good and poor sleep quality. Methodology:
- Participant Stratification: Participants are categorized based on standardized self-report questionnaires like the Pittsburgh Sleep Quality Index (PSQI), with a score of ≥6 indicating poor sleep.
- Sample Collection: Saliva is collected non-invasively from participants, offering a practical alternative to blood draws.
- Metabolite Analysis: Saliva samples are analyzed using high-throughput techniques like Capillary Electrophoresis–Fourier Transform Mass Spectrometry (CE-FTMS), which can identify hundreds of metabolites.
- Data Analysis and Modeling: Univariate statistics (e.g., Mann-Whitney U tests) identify individual metabolites that differ between groups. Machine learning models, such as random forest analysis, are then employed to build a predictive panel of biomarkers from the most significant metabolites [50].
Mendelian Randomization for Drug Target Identification
Objective: To identify plasma proteins with a causal relationship to insomnia risk for prioritization as novel drug targets. Methodology:
- Genetic Instrument Selection: Genome-wide association studies (GWAS) are used to identify genetic variants (single nucleotide polymorphisms, or SNPs) that are significantly associated with natural variation in plasma protein levels (pQTLs).
- Causal Inference: These protein-associated SNPs are used as instrumental variables in a Mendelian randomization (MR) analysis. MR tests whether genetically predicted higher levels of a specific protein are associated with a higher or lower odds of insomnia (using summary statistics from large insomnia GWAS).
- Validation and Sensitivity Analysis: Significant protein targets from the initial analysis are validated in independent datasets. Additional analyses, such as Bayesian co-localization, are performed to confirm that the same genetic variant influences both the protein and insomnia risk, strengthening the causal claim [84].
Preclinical Assessment of HPA Axis and GABAergic Function
Objective: To investigate the efficacy and mechanism of action of potential therapeutics in an animal model of stress-related pathology. Methodology (as exemplified in a PTSD study with relevance to insomnia):
- Disease Modeling: Mice are subjected to a paradigm like "single-prolonged stress with shock" (SPSS) to induce a PTSD-like state, which includes severe sleep disturbances and hyperarousal.
- Drug Administration: The test compound (e.g., Bojungikgi-tang) is administered orally over a set period (e.g., 14 days).
- Behavioral Phenotyping: Animals undergo a battery of tests: Open Field Test (for anxiety-like behavior), Forced Swimming Test (for depression-like behavior), Y-Maze (for spatial memory), and Contextual Fear Conditioning (for aversive memory).
- Molecular Endpoint Analysis: Post-behavioral testing, serum is collected to measure corticosterone levels via ELISA. Brain tissue (e.g., hippocampus) is analyzed via Western blot or immunohistochemistry for proteins related to GABAergic function (e.g., GAD67, vGAT, parvalbumin) and neuronal activity/plasticity (e.g., c-Fos, DCX) [83].
Research Reagent Solutions Toolkit
Table 3: Essential Research Tools for Investigating Insomnia Pathophysiology
| Reagent / Assay | Function in Research | Specific Application Example |
|---|---|---|
| Pittsburgh Sleep Quality Index (PSQI) | Validated self-report questionnaire for assessing subjective sleep quality and disturbances over a 1-month interval. | Stratifying research participants into "good" and "poor" sleeper groups for case-control studies [50]. |
| Capillary Electrophoresis–Fourier Transform Mass Spectrometry (CE-FTMS) | High-resolution platform for untargeted metabolomic analysis, capable of identifying a wide array of polar and ionic metabolites in biofluids. | Profiling salivary metabolites to discover biomarkers of chronic sleep disturbance [50]. |
| Magnetic Resonance Spectroscopy (MRS) | Non-invasive neuroimaging technique that measures the concentration of specific neurochemicals, such as GABA and glutamate, in vivo. | Quantifying GABA levels in the occipital or anterior cingulate cortex of individuals with insomnia compared to healthy controls [79]. |
| Enzyme-Linked Immunosorbent Assay (ELISA) | Sensitive biochemical assay used to quantitatively measure concentrations of specific proteins or hormones, such as cortisol, in serum, saliva, or plasma. | Determining diurnal cortisol patterns or stress-induced corticosterone levels in preclinical models [83]. |
| Polysomnography (PSG) with EEG | Gold-standard objective test for sleep, measuring brain waves (EEG), eye movements (EOG), muscle activity (EMG), and other physiological parameters. | Objectively quantifying sleep architecture and identifying EEG signatures of hyperarousal (e.g., high-frequency activity) [82]. |
Pathway and Workflow Visualizations
Core Pathophysiology of Insomnia
Drug Target Identification Workflow
The intertwined pathologies of hyperarousal, HPA axis dysregulation, and GABAergic deficits provide a robust neurobiological framework for understanding chronic insomnia. The evidence from neuroimaging, neuroendocrinology, genetics, and metabolomics consistently points to a state of 24-hour hyperarousal that disrupts normal sleep-wake cycles. This mechanistic understanding is crucial for moving beyond symptomatic treatment towards mechanism-based therapeutics. The identification of causal plasma proteins like PAM through Mendelian randomization and the characterization of distinct metabolic signatures of poor sleep represent the vanguard of this effort, offering new, biologically-grounded targets for drug development. Future research that further dissects these pathways and validates targets in dedicated clinical trials holds the promise of yielding more effective and personalized treatments for this debilitating disorder.
Narcolepsy Type 1 (NT1) is a chronic neurological disorder characterized by a primary disorganization of sleep-wake states. Its pathology is uniquely defined by the degeneration of orexin-producing neurons in the lateral hypothalamus, leading to a marked deficiency in the orexin neuropeptide in the cerebrospinal fluid (CSF) [85] [86]. This specific pathophysiology distinguishes NT1 from other sleep disorders and establishes it as a model disease for understanding the critical role of the orexin system in maintaining wakefulness and stabilizing sleep architecture.
The orexin system, comprising the neuropeptides orexin-A and orexin-B (also known as hypocretin-1 and hypocretin-2), is central to the regulation of sleep/wake cycles, energy homeostasis, and reward processes [87] [86]. Orexin neurons project extensively throughout the neuraxis, from the spinal cord to cortical regions, and act on two G-protein coupled receptors (GPCRs): orexin receptor 1 (OX1R) and orexin receptor 2 (OX2R) [87] [88]. Orexin-A binds with approximately equal affinity to both receptors, whereas orexin-B has a 5- to 10-fold higher affinity for OX2R [88] [86]. The widespread projection pattern of orexin neurons allows them to coordinate activity across diverse brain regions, making them a master regulator of behavioral state stability.
Molecular Basis of Orexin System Dysfunction
Orexin Neuron Degeneration
The orexin-producing neurons are localized exclusively in the lateral hypothalamus, perifornical area, and dorsomedial hypothalamus [86]. In NT1, a selective loss of these neurons is observed, which is believed to be caused by an autoimmune-mediated destruction process [89]. This neurodegeneration results in undetectable or significantly low levels of orexin-A in the CSF, a diagnostic hallmark of the condition [86]. The absence of orexin signaling disrupts the normal transition between sleep and wake states, leading to the signature symptoms of NT1: excessive daytime sleepiness (EDS), cataplexy (sudden loss of muscle tone triggered by emotions), sleep paralysis, hypnagogic/hypnopompic hallucinations, and disrupted nighttime sleep [85].
Orexin Receptor Distribution and Function
The two orexin receptors exhibit distinct distributions within the central nervous system, which informs their functional roles:
- OX1R Expression: More highly expressed in cortical regions, the bed nucleus of the stria terminalis, and the locus coeruleus [87]. This receptor is particularly implicated in the modulation of emotion, reward, and autonomic functions.
- OX2R Expression: More highly expressed in the nucleus accumbens, anterior pretectal nucleus, cerebral cortex, and tuberomammillary nucleus [87] [88]. This receptor is critically involved in promoting and stabilizing wakefulness, with strong projections to histaminergic neurons in the tuberomammillary nucleus that are essential for maintaining arousal.
The differential expression patterns and ligand affinities of these receptors provide the molecular basis for targeted therapeutic interventions, particularly the development of selective OX2R agonists to promote wakefulness without directly engaging emotional pathways mediated by OX1R.
Table 1: Orexin System Components and Their Characteristics
| Component | Characteristics | Role in NT1 |
|---|---|---|
| Orexin Neurons | Localized in lateral hypothalamus; extensive projections throughout CNS [87] | Selectively degenerated in NT1, causing orexin deficiency [86] |
| Orexin-A | 33-amino acid peptide; equal affinity for OX1R and OX2R [88] | Low/undetectable CSF levels are a diagnostic biomarker for NT1 [86] |
| Orexin-B | 28-amino acid peptide; higher affinity for OX2R [88] | Deficient in NT1, contributing to sleep-wake instability |
| OX1 Receptor | GPCR; enriched in cortex, locus coeruleus [87] | Less directly implicated in primary NT1 sleep-wake symptoms |
| OX2 Receptor | GPCR; enriched in TMN, nucleus accumbens, cortex [87] [88] | Primary target for novel therapeutics aiming to restore wakefulness |
Research Methodologies and Experimental Protocols
Assessment of Wakefulness and Sleepiness in Clinical Trials
Clinical research on NT1 therapeutics relies on standardized objective and subjective measures to quantify excessive daytime sleepiness and treatment efficacy.
- Maintenance of Wakefulness Test (MWT): An objective test that evaluates the ability to stay awake in a sleep-conducive environment. The protocol consists of four to five 40-minute sessions conducted at two-hour intervals in a quiet, dark room. Participants are instructed to try to remain awake while seated in a relaxed position. Sleep latency (time to fall asleep) is measured using electroencephalography (EEG), electromyography (EMG), and electrooculography (EOG). Longer sleep latencies indicate a better ability to maintain wakefulness [85].
- Epworth Sleepiness Scale (ESS): A subjective, self-administered questionnaire that assesses an individual's general level of daytime sleepiness. Patients rate their likelihood of dozing in eight different situations on a scale of 0-3. The total score ranges from 0 to 24, with higher scores indicating greater daytime sleepiness. A score of 10 or below is generally considered within the normal range [89].
- Modified Karolinska Sleepiness Scale (KSS): A simple subjective scale used to assess the current intensity of sleepiness on a 10-point scale, ranging from "1 - Extremely alert" to "10 - Extremely sleepy, falls asleep all the time" [85].
Cataplexy Assessment
- Weekly Cataplexy Rate (WCR): Patients maintain daily diaries to record the frequency and duration of cataplexy attacks. The change in the weekly rate from baseline to the study endpoint is a key secondary endpoint in clinical trials of NT1 therapeutics [89].
Experimental Workflow for Phase 3 Clinical Trials of Orexin Agonists
The following diagram illustrates the standardized protocol for recent Phase 3 clinical trials investigating orexin receptor agonists in NT1:
Quantitative Findings from Recent Clinical Trials
Recent Phase 3 clinical trials of novel orexin receptor agonists have demonstrated significant improvement across multiple efficacy endpoints in NT1 patients, as summarized in the table below.
Table 2: Efficacy Outcomes from Recent Orexin Agonist Phase 3 Clinical Trials (12-Week Data)
| Efficacy Measure | Intervention & Dose | Baseline Value | Change from Baseline | P-value vs. Placebo |
|---|---|---|---|---|
| MWT Sleep Latency (minutes) [89] | Oveporexton (2 mg BID) | ~6-8 min | Increase to >20 min* | <0.001 |
| E2086 (25 mg QD) [85] | Not specified | Significant prolongation | <0.0001 | |
| ESS Score [89] | Oveporexton (2 mg BID) | ~15-17 | Decrease to ≤10* | <0.001 |
| Weekly Cataplexy Rate [89] | Oveporexton (2 mg BID) | Variable | >80% median reduction | <0.001 |
| Narcolepsy Severity Scale [89] | Oveporexton (2 mg BID) | Not specified | >70% patients reached 'mild' severity | <0.001 |
| Patient Global Impression of Change [89] | Oveporexton (2 mg BID) | Not applicable | 97% reported improvement | <0.001 |
Note: BID = twice daily; QD = once daily. *Indicates value within normative range achieved by majority of patients.
Emerging Therapeutic Strategies: Orexin Receptor Agonists
The understanding of NT1 pathophysiology has led to a paradigm shift from symptomatic management to targeted replacement therapy. Instead of using broad-spectrum stimulants for EDS and antidepressants for cataplexy, the new therapeutic approach aims to restore the missing orexin signaling.
Mechanism of Orexin Agonism
Orexin receptor agonists are designed to selectively activate OX2R or both orexin receptors, thereby promoting wakefulness and reducing abnormal REM-sleep phenomena like cataplexy. The following diagram illustrates the cellular mechanism of action of these novel therapeutics:
Clinical Profile of Investigational Orexin Agonists
- Oveporexton (TAK-861): An investigational oral OX2R-selective agonist. In Phase 3 trials (FirstLight and RadiantLight), oveporexton demonstrated statistically significant improvement across all primary and secondary endpoints with twice-daily dosing (1mg and 2mg). It was generally well-tolerated, with the most common adverse events being insomnia, urinary urgency, and frequency. No treatment-related serious adverse events were reported [89].
- E2086: Eisai's novel selective orexin 2 receptor agonist. A Phase Ib clinical trial demonstrated that once-daily dosing (5mg, 10mg, 25mg) resulted in statistically significant longer sleep latencies on MWT compared to both placebo and modafinil. Common treatment-emergent adverse events included increased urinary frequency, nausea, dizziness, and urinary urgency, exhibiting a dose-dependent trend [85].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents for Orexin System and NT1 Investigation
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| Selective OX2R Agonists (e.g., Oveporexton, E2086, YNT-185) [85] [88] [89] | Activate OX2R signaling to promote wakefulness; potential NT1 therapeutics | In vivo proof-of-concept studies in narcolepsy models [88] |
| Dual Orexin Receptor Antagonists (DORAs; e.g., Suvorexant, Lemborexant) [85] [90] [91] | Block both OX1R and OX2R to promote sleep; tool compounds for probing orexin system function | Investigating the role of orexin in sleep initiation and maintenance [90] |
| Prepro-orexin Antibodies | Detect orexin precursor protein in tissue samples via immunohistochemistry | Quantifying orexin neuron population in post-mortem hypothalamic tissue [86] |
| CSF Orexin-A Immunoassay | Measure orexin-A concentration in cerebrospinal fluid | Clinical confirmation of NT1 diagnosis (low CSF orexin-A) [86] |
| Polysomnography (PSG) with EEG/EMG/EOG | Objective measurement of sleep architecture and wakefulness | Conducting the Maintenance of Wakefulness Test (MWT) in clinical trials [85] |
| Narcolepsy Severity Scale (NSS-CT) [89] | Comprehensive clinician-rated scale assessing NT1 symptom frequency and severity | Evaluating broad treatment efficacy in clinical trials beyond EDS and cataplexy |
The direct link between orexin neuron degeneration and the complex symptomatology of Narcolepsy Type 1 provides a powerful model for understanding the neurobiological basis of sleep-wake regulation. The emergence of orexin receptor agonists represents a transformative shift from symptomatic treatment to targeted mechanism-based therapy, directly addressing the underlying pathophysiology of NT1. These novel agents have demonstrated robust efficacy in restoring wakefulness and controlling cataplexy in late-stage clinical trials, heralding a new era of care for patients.
Future research directions include further elucidation of the autoimmune mechanisms triggering orexin neuron loss, the development of biomarkers for early diagnosis and disease monitoring, and exploration of orexin-based therapies for other neurological and psychiatric conditions where sleep-wake disturbances and orexin system dysregulation are implicated [87] [90] [91]. The orexin system continues to serve as a compelling example of how fundamental neurobiological research can directly catalyze the development of novel and powerful therapeutic strategies for complex brain disorders.
The escalating prevalence of metabolic diseases coincides with increasing instances of sleep disorders in modern societies. This whitepaper examines the intricate relationship between sleep disruption, adipose tissue physiology, and the regulatory role of melatonin. We synthesize evidence from clinical and preclinical studies demonstrating how sleep disturbances promote metabolic dysfunction through altered adipokine secretion, insulin resistance, and adipose tissue inflammation. Conversely, we explore how melatonin, a circadian hormone whose production is impaired by sleep disruption and light-at-night exposure, counteracts these effects by promoting brown adipose tissue (BAT) thermogenesis, browning of white adipose tissue (WAT), and improving systemic metabolism. This analysis provides researchers and drug development professionals with a comprehensive framework for understanding these interconnected pathways and identifies potential therapeutic targets for metabolic disorders rooted in sleep-wake cycle disruptions.
Sleep is a fundamental physiological process crucial for metabolic homeostasis, and its disruption has emerged as a significant risk factor for obesity and related metabolic disorders [92]. The global increase in sleep disorders, affecting an estimated 35.7% of the population, parallels the rising incidence of obesity, suggesting potential shared pathophysiological mechanisms [50]. Within this context, adipose tissue has been recognized not merely as a passive energy storage depot but as an active endocrine organ that responds to sleep-related signals [92]. Melatonin, the pineal hormone synthesized predominantly during nighttime darkness, serves as a critical link between circadian rhythms and energy metabolism [93] [94]. The significance of understanding this relationship is heightened by the recent discovery that functional brown adipose tissue (BAT) is retained in adult humans, providing a potential therapeutic target for combating obesity [94]. This technical review examines the molecular and clinical evidence linking sleep disruption to adipose tissue dysfunction and elucidates the potential of melatonin as a regulatory agent in metabolic health.
Metabolic Consequences of Sleep Disruption
Impact on Adipokine Secretion and Glucose Metabolism
Sleep disruption profoundly alters adipokine secretion patterns, particularly affecting leptin, a hormone critical for appetite regulation. Multiple controlled studies demonstrate that sleep restriction significantly reduces circulating leptin levels independently of body mass changes:
Table 1: Effects of Sleep Restriction on Leptin Levels in Controlled Studies
| Study Design | Sleep Intervention | Impact on Leptin | Associated Metabolic Changes |
|---|---|---|---|
| Acute sleep restriction [92] | 2 nights of insufficient sleep | 18% reduction | Increased hunger, preference for calorie-dense foods |
| Chronic partial sleep restriction [92] | 4 hours/night for 6 nights | 19% reduction | Impaired glucose tolerance, decreased insulin sensitivity |
| Extended sleep restriction [92] | 4 hours/night for 7 days | 33% reduction | Not specified |
| Total sleep deprivation [92] | 88 hours without sleep | Decreased amplitude and mean levels | Reversed after recovery sleep |
These leptin alterations occur through several mechanisms: (1) reduced insulin-stimulated glucose uptake and metabolism in adipocytes, which is necessary for leptin production [92]; (2) disruption of normal circadian leptin rhythms, which typically peak between midnight and early morning [92]; and (3) potential leptin resistance independent of changes in body mass index [92].
Concurrently, sleep disruption induces significant impairments in glucose metabolism. As little as two nights of insufficient sleep decreases the disposition index, a validated predictor of diabetes risk [92]. Six nights of sleep restriction (4 hours/night) impairs glucose tolerance, reduces acute insulin response to glucose, and decreases insulin sensitivity [92]. The combination of these effects significantly elevates diabetes risk.
White Adipose Tissue Transcriptomic Reprogramming
Sleep loss induces profound molecular changes in white adipose tissue, restructuring the transcriptome in ways that promote metabolic dysfunction. A rigorous study involving 15 healthy men examined morning-to-evening differences in WAT transcriptome under three conditions: normal sleep (8 hours), sleep restriction (4 hours), and total sleep deprivation [95].
Table 2: Sleep Loss Effects on WAT Transcriptome and Metabolic Parameters
| Parameter | Normal Sleep | Sleep Restriction | Sleep Deprivation |
|---|---|---|---|
| Transcriptome rhythmicity | Preserved morning-to-evening variations | Dramatically blunted | Further dampened |
| Core clock genes | Stable rhythmicity | Largely maintained | Mostly maintained |
| Metabolic pathways | Normal rhythmicity | Disrupted | Severely disrupted |
| Carbohydrate metabolism genes | Normal rhythmicity | Altered | Lost rhythmicity, morning upregulation |
| Clinical correlates | Normal metabolic parameters | Impaired glucose homeostasis | Increased retinol-binding protein 4 |
This transcriptional reprogramming occurs despite preservation of core clock gene rhythms, indicating that sleep loss uncovers metabolic pathways from local circadian control in WAT [95]. The findings suggest that sleep restriction preferentially affects metabolic genes over core circadian regulators, leading to increased carbohydrate turnover in adipose tissue and systemic metabolic impairments.
Systemic Inflammation and Adipose Tissue Insulin Resistance
Sleep disruption promotes a pro-inflammatory state in adipose tissue that contributes to systemic metabolic dysfunction. Chronic sleep fragmentation (SF) in mouse models induces visceral white adipose tissue (VWAT) inflammation characterized by increased infiltration of neutrophils and macrophages, particularly those with M1 polarity [96]. This inflammatory state is associated with:
- Increased expression of pro-inflammatory markers (LBP, IL-6, NGAL) in VWAT adipocytes and macrophages [96]
- Reduced insulin sensitivity evidenced by decreased p-AKT/AKT ratios in VWAT adipocytes [96]
- Elevated plasma inflammatory markers (LBP, NGAL, IL-6) [96]
These changes occur even in the absence of significant body weight changes, suggesting that sleep disruption itself directly promotes adipose tissue inflammation and insulin resistance [96]. The correlation between p-AKT/AKT ratios in mesenteric adipose tissues and plasma LBP/NGAL levels further supports the link between adipose tissue inflammation and metabolic dysfunction in sleep disruption [96].
Altered Gut Microbiota and Metabolic Phenotype
Emerging evidence indicates that sleep disruption significantly alters gut microbiota composition, which subsequently affects adipose tissue metabolism. Chronic sleep fragmentation in mice induces reversible changes in gut microbiota characterized by:
- Increased abundance of Firmicutes (20%) with concomitant decreases in Bacteroidetes (20%) and Actinobacteria (50%) [96]
- Expansion of highly fermentative families Lachnospiraceae and Ruminococcaceae [96]
- Suppression of Lactobacillaceae and Bifidobacteriaceae families [96]
These microbial changes promote systemic inflammation and adipose tissue insulin resistance through enhanced colonic epithelium barrier disruption [96]. Conventionalization of germ-free mice with SF-derived microbiota recapitulates these metabolic alterations, confirming the causal role of gut microbiota in SF-induced metabolic dysfunction [96].
Melatonin as a Regulatory Factor in Adipose Tissue Physiology
Melatonin Signaling and BAT Activation
Melatonin demonstrates significant potential in counteracting obesity by activating brown adipose tissue and promoting browning of white adipose tissue. A proof-of-concept study involving patients with melatonin deficiency demonstrated that melatonin replacement (3 mg daily for 3 months) increased BAT volume and activity as measured by positron emission tomography-MRI [93]. This clinical finding corroborates extensive preclinical evidence showing melatonin's effects on energy metabolism:
Table 3: Metabolic Effects of Melatonin Supplementation in Preclinical Models
| Parameter | Normal Chow + Melatonin | High-Fat Diet + Vehicle | High-Fat Diet + Melatonin |
|---|---|---|---|
| Body weight | No significant change | Significant increase | Attenuated weight gain |
| Fat mass | No significant change | Significant increase | Reduced |
| Adipocyte size | No significant change | Hypertrophy | Reduced size |
| BAT UCP1 expression | Increased | Decreased | Restored/Increased |
| Energy expenditure | No change or slight increase | Reduced | Prevented reduction |
| Insulin sensitivity | No significant change | Insulin resistance | Improved |
| Plasma lipids | Reduced TG | Elevated TG | Reduced TG |
Mechanistically, melatonin increases BAT thermogenesis and promotes WAT browning through FGF21-dependent pathways. In FGF21-/- mice, melatonin fails to protect against obesity, insulin resistance, and reduced energy expenditure, confirming the essential role of FGF21 in mediating melatonin's metabolic benefits [97].
Molecular Mechanisms of Melatonin Action
Melatonin regulates adipose tissue physiology through multiple interconnected mechanisms:
- Enhancing BAT thermogenesis: Melatonin increases UCP1 expression in brown adipocytes, enhancing uncoupled respiration and energy expenditure [97].
- Promoting white adipose tissue browning: Melatonin induces the appearance of beige adipocytes in WAT depots, characterized by increased mitochondrial content and UCP1 expression [98] [97].
- Modulating adipokine secretion: Melatonin regulates the secretion of adipokines involved in energy homeostasis and insulin sensitivity [98].
- Reducing adipose tissue inflammation: Melatonin exerts anti-inflammatory effects in adipose tissue, reducing macrophage infiltration and pro-inflammatory cytokine production [97].
- Shift in energy substrate utilization: Melatonin treatment increases fat utilization, as evidenced by decreased respiratory exchange ratio [97].
These multifaceted actions position melatonin as a significant regulator of energy metabolism through coordinated effects on different adipose tissue depots.
Experimental Models and Methodologies
Human Studies Protocol: Adipose Tissue Biopsy and Transcriptomic Analysis
Objective: To assess the acute effects of sleep curtailment on circadian regulation of white adipose tissue transcriptome in normal-weight men [95].
Participants: 15 healthy men aged 18-30 years.
Study Design: Randomized, balanced crossover design with three conditions:
- Regular sleep (8 hours: 11:00 PM - 7:00 AM)
- Sleep restriction (4 hours: 3:00 AM - 7:00 AM)
- Sleep deprivation (no sleep)
Methodological Details:
- Polysomnographic monitoring to verify sleep stages and duration
- WAT biopsy samples collected at 9:00 PM and 7:00 AM under each condition
- RNA sequencing for transcriptome profiling
- Analysis of core clock gene expression using qPCR
- Assessment of glucose homeostasis, lipid profile, and adipokines
Key Measurements:
- Morning-to-evening differences in WAT transcriptome
- Pathway enrichment analysis of differentially expressed genes
- Correlation between transcriptional changes and metabolic parameters
Animal Studies Protocol: Melatonin Supplementation in HFD Model
Objective: To assess the effects of melatonin on BAT activity and energy metabolism in high-fat diet-induced obese mice [97].
Animals: 8-week-old male C57BL/6 mice and FGF21-/- mice.
Study Groups (12-week treatment):
- Normal chow + vehicle
- Normal chow + melatonin (20 mg/kg, i.p.)
- High-fat diet + vehicle
- High-fat diet + melatonin (20 mg/kg, i.p.)
Methodological Details:
- Daily intraperitoneal injection of melatonin (20 mg/kg) or vehicle before dark
- Pair-feeding protocol to control food intake
- Indirect calorimetry for energy expenditure measurement
- Body composition analysis using EchoMRI
- Glucose and insulin tolerance tests
- Tissue collection for histology, gene expression, and protein analysis
Key Measurements:
- Body weight and adiposity
- BAT UCP1 expression and activity
- Insulin signaling (p-AKT/AKT ratios)
- Inflammatory markers in adipose tissue
- Energy expenditure and substrate utilization
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Reagents for Investigating Sleep-Melatonin-Adipose Tissue Axis
| Reagent/Category | Specific Examples | Research Application | Key Findings Enabled |
|---|---|---|---|
| Melatonin Formulations | Synthetic melatonin (Sigma-Aldrich #M5250) [97] | In vivo supplementation studies | Dose-dependent effects on BAT activation and metabolic parameters |
| Adipose Tissue Markers | UCP1 antibodies [97], Leptin ELISA | Histological and protein analysis | BAT activation status, adipokine secretion changes |
| Metabolic Assay Kits | Glucose, insulin, FGF21, triglyceride assays [97] | Metabolic phenotyping | Systemic glucose homeostasis, lipid metabolism |
| RNA Analysis Tools | TRIzol RNA isolation, SYBR Green qPCR [97] | Gene expression analysis | Transcriptional regulation in adipose tissue |
| Circadian Rhythm Assessment | Polysomnography equipment [95] | Sleep architecture monitoring | Correlation between sleep stages and metabolic parameters |
| Immunohistochemistry Reagents | F4/80 antibodies [97] | Macrophage infiltration analysis | Adipose tissue inflammation quantification |
| Metabolomic Analysis | CE-FTMS platforms [50] | Salivary metabolome profiling | Identification of sleep quality biomarkers |
| Body Composition Analyzers | EchoMRI systems [97] | Fat/lean mass quantification | Longitudinal body composition changes |
Integrated Pathophysiological Framework
The relationship between sleep disruption, melatonin signaling, and adipose tissue dysfunction can be visualized as a series of interconnected pathological pathways:
This integrated framework illustrates how sleep disruption initiates a cascade of events leading to metabolic disease, while also highlighting potential intervention points. The model emphasizes: (1) the central role of melatonin suppression in mediating sleep-related metabolic dysfunction; (2) the multifaceted impact on different adipose tissue depots; and (3) the bidirectional relationship between sleep disruption, adipose tissue dysfunction, and systemic metabolism.
The evidence synthesized in this review establishes a robust connection between sleep disruption, impaired melatonin signaling, and adipose tissue dysfunction in the pathogenesis of metabolic diseases. Key findings demonstrate that sleep restriction alters WAT transcriptome, reduces BAT activity, promotes adipose tissue inflammation, and disrupts adipokine secretion, collectively contributing to metabolic impairment. Melatonin emerges as a significant regulatory factor that counteracts these effects by enhancing BAT thermogenesis, promoting WAT browning, and improving insulin sensitivity through FGF21-dependent mechanisms.
Future research should focus on: (1) elucidating the precise molecular pathways through which melatonin regulates adipocyte differentiation and function; (2) establishing optimal dosing and timing of melatonin supplementation for metabolic benefits in humans; (3) investigating the interplay between melatonin, other circadian regulators, and adipose tissue metabolism; and (4) developing targeted therapies that specifically address the melatonin-adipose tissue axis for preventing and treating obesity and related metabolic disorders. The integration of sleep optimization and melatonin-related interventions may offer novel approaches for addressing the escalating global burden of metabolic diseases.
Magnesium (Mg²⁺) serves as a critical physiological modulator of neural excitability and synchronization necessary for sleep architecture. This whitepaper examines the essential mechanisms through which magnesium regulates ion homeostasis by targeting N-methyl-D-aspartate (NMDA) and γ-aminobutyric acid type A (GABA-A) receptors, thereby influencing sleep-wake cycles. We synthesize recent structural biology breakthroughs revealing specific magnesium-binding pockets on NMDA receptors alongside clinical evidence demonstrating the efficacy of magnesium supplementation in sleep disorders. The integration of molecular, structural, and clinical data establishes a compelling framework for targeting magnesium-dependent pathways in the development of novel therapeutics for sleep disorders.
Sleep is an active physiological state regulated by complex neurochemical circuits and molecular pathways. Disruption of ion homeostasis represents a fundamental mechanism underlying various sleep disorders, making the regulation of neuronal excitability a primary target for therapeutic intervention [99]. Among the minerals essential for neurological function, magnesium emerges as a crucial cation participating in over 300 enzymatic reactions and serving as a key regulator of neural excitability [99] [100].
Magnesium's role in sleep physiology extends beyond basic nutritional supplementation to direct receptor-level modulation of major neurotransmitter systems. As the second most abundant intracellular cation, magnesium regulates multiple aspects of neuronal function, including ion channel gating, neurotransmitter release, and receptor activation [101] [102]. This review examines the specific molecular mechanisms through which magnesium governs NMDA receptor and GABA-A receptor function, integrates structural biology insights with functional outcomes, and explores the translational potential for sleep disorder management within a biochemical research framework.
Mechanistic Insights: Magnesium's Regulation of Receptor Physiology
NMDA Receptor Modulation: Structural Foundations
The NMDA receptor represents a critical gateway for calcium influx and serves as a coincidence detector in synaptic plasticity. Magnesium's voltage-dependent block of this receptor is essential for preventing excitotoxicity and maintaining neural stability during sleep-wake transitions [103] [104].
Recent cryo-EM structural studies of GluN1-N2B NMDA receptors have identified three distinct magnesium-binding pockets with differential functions:
- Site I: Located at the selectivity filter formed by asparagine residues (QRN site), this site mediates the classical voltage-dependent magnesium block that controls calcium permeation [103] [104].
- Site II: Situated at the N-terminal domain (NTD) of the GluN2B subunit, this site consists of three acidic residues and is responsible for GluN2B-specific magnesium potentiation, which enhances receptor function under specific conditions [104].
- Site III: Also located at the GluN2B-NTD, this site overlaps with the zinc-binding pocket and contributes to allosteric inhibition of channel activity [104].
Table 1: Magnesium Binding Sites on NMDA Receptors
| Site | Location | Structural Features | Functional Role |
|---|---|---|---|
| Site I | Selectivity filter | Asparagine ring coordination | Voltage-dependent channel block |
| Site II | GluN2B-NTD | Three acidic residues | GluN2B-specific potentiation |
| Site III | GluN2B-NTD | Overlaps with Zn²⁺ pocket | Allosteric inhibition |
These structural insights explain how magnesium exerts multifaceted regulation over NMDA receptor function, balancing the fine line between necessary synaptic plasticity during wakefulness and potential excitotoxicity during sleep transitions.
GABAergic Enhancement: Pharmacological Potentiation
Beyond NMDA receptor blockade, magnesium directly enhances inhibitory neurotransmission through GABA-A receptors. Magnesium acts as an agonist of GABA-A receptors, triggering chloride influx that leads to neuronal hyperpolarization and inhibition [101]. Experimental evidence indicates that physiologically relevant magnesium concentrations (up to 1 mM) potentiate GABA responses on GABA-A receptors, while higher concentrations (10 mM) may exhibit inhibitory effects [101].
The GABAergic effects of magnesium are mechanistically distinct from benzodiazepines, as evidenced by suppression of magnesium's anxiolytic effects by flumazenil, a GABA-A receptor antagonist [101]. This suggests a unique binding site or mechanism separate from the classical benzodiazepine binding pocket. Through this dual action—inhibition of excitatory NMDA receptors and potentiation of inhibitory GABA-A receptors—magnesium creates a net inhibitory effect on central nervous system activity, promoting the conditions necessary for sleep initiation and maintenance.
Diagram 1: Magnesium's dual receptor modulation promotes sleep by reducing neuronal excitability.
Experimental Evidence and Clinical Correlations
Preclinical and Clinical Studies
The molecular mechanisms of magnesium-mediated sleep regulation are supported by substantial preclinical and clinical evidence. Animal studies have demonstrated that magnesium deficiency shortens sleep duration and reduces slow-wave sleep power, while magnesium supplementation enhances sleep quality and architecture [99] [101].
Table 2: Clinical Evidence for Magnesium in Sleep Regulation
| Study Design | Population | Intervention | Key Sleep Outcomes | Mechanistic Insights |
|---|---|---|---|---|
| RCT (Nielsen et al, 2010) [99] | Adults with sleep complaints (n=100) | 320 mg Mg citrate for 7 weeks | Improved PSQI scores | Enhanced sleep quality |
| RCT (Abbasi et al, 2012) [99] | Elderly with primary insomnia (n=46) | 500 mg elemental Mg (as oxide) for 8 weeks | Improved subjective and objective insomnia measures | Restoration of sleep architecture |
| RCT (Luo et al, 2024) [105] | Adults with self-reported sleep problems (n=80) | 1 g/day Mg L-threonate for 21 days | Improved deep and REM sleep; better daytime functioning | Improved neuronal function via bioavailable Mg |
| Cohort (CARDIA Study) [106] | Young adults (n=3,964) | Dietary Mg assessment | 36% reduced risk of short sleep (<7 hr) | Long-term sleep duration protection |
| Mendelian Randomization (Wang et al, 2025) [107] | European ancestry (408,074) | Genetic instrumental variable analysis | Causal protection against insomnia (OR=0.869) | Genetic evidence for causality |
Recent research utilizing magnesium-L-threonate (MgT), a form with enhanced brain bioavailability, demonstrates particularly promising results. In a randomized, double-blind, placebo-controlled trial with adults experiencing self-reported sleep problems, MgT supplementation significantly improved both subjective and objective sleep parameters [105]. The MgT group demonstrated improved deep sleep scores, REM sleep metrics, and daytime functioning compared to placebo, suggesting direct central nervous system effects [105].
Magnesium Deficiency and Sleep Pathology
The flip side of magnesium's sleep-promoting effects is evident in deficiency states. Hypomagnesemia is associated with various sleep pathologies, including insomnia, excessive daytime sleepiness, and specific sleep disorders such as restless legs syndrome/Willis-Ekbom disease (RLS/WED) [99]. Magnesium deficiency creates a state of neuronal hyperexcitability through impaired NMDA receptor blockade and reduced GABAergic inhibition, disrupting the balance necessary for normal sleep initiation and maintenance [99] [102].
Epidemiological evidence further supports this relationship. The CARDIA study, a large longitudinal analysis, found that higher magnesium intake was associated with better sleep quality and reduced risk of short sleep duration, particularly in individuals without depressive disorders [106]. Mendelian randomization studies providing genetic evidence for causal inference have confirmed that higher magnesium levels protect against insomnia risk (OR=0.869, 95% CI=0.763-0.990) [107].
Methodological Approaches in Magnesium-Sleep Research
Structural Biology Techniques
The elucidation of magnesium-binding sites on NMDA receptors employed sophisticated structural biology approaches. Key methodological workflows include:
Cryo-Electron Microscopy (cryo-EM) Workflow:
- Protein Expression: Recombinant human GluN1-1a and GluN2B subunits co-expressed in eukaryotic systems (e.g., Xenopus oocytes or mammalian cells) [104]
- Receptor Purification: Detergent solubilization followed by affinity and size-exclusion chromatography [104]
- Grid Preparation: Vitrification of purified receptors in magnesium-containing solutions [104]
- Data Collection: High-resolution imaging using modern cryo-EM detectors [104]
- Image Processing: Single-particle analysis to reconstruct 3D density maps [104]
- Model Building: Atomic model docking and refinement to identify magnesium coordination geometries [104]
Electrophysiological Validation:
- Two-Electrode Voltage Clamp (TEVC): Functional characterization of magnesium effects on recombinant receptors expressed in Xenopus oocytes [104]
- Current-Voltage (I-V) Curves: Assessment of voltage-dependent magnesium block across membrane potentials from -100 mV to +60 mV [104]
- Site-Directed Mutagenesis: Validation of binding site significance through point mutations of key residues (e.g., asparagine residues in QRN site) [104]
Diagram 2: Integrated structural and functional workflow for studying Mg²⁺ binding.
Clinical Trial Methodologies
Robust clinical investigation of magnesium's effects on sleep incorporates both subjective and objective measures:
Sleep Assessment Methods:
- Polysomnography (PSG): Gold standard objective measurement of sleep architecture, including deep sleep (N3) and REM sleep quantification [99]
- Actigraphy: Objective assessment of sleep-wake patterns using wrist-worn movement detectors [105]
- Standardized Questionnaires: Pittsburgh Sleep Quality Index (PSQI), Insomnia Severity Index (ISI), Epworth Sleepiness Scale (ESS) for subjective sleep quality assessment [99] [105] [106]
- Sleep Diaries: Daily participant recordings of sleep timing, quality, and daytime functioning [105]
Supplementation Protocols:
- Dosage Range: 320-500 mg elemental magnesium daily [99] [105]
- Formulations: Citrate, oxide, and L-threonate salts with differing bioavailability profiles [99] [105]
- Duration: Typically 3-12 weeks to assess both acute and sustained effects [99] [105]
Research Toolkit: Essential Reagents and Methodologies
Table 3: Essential Research Reagents for Magnesium-Sleep Investigations
| Reagent/Method | Specifications | Research Application |
|---|---|---|
| Recombinant NMDA Receptors | GluN1-N2A/GluN1-N2B subtypes | Structural and functional studies of subtype-specific Mg²⁺ effects |
| Magnesium Salts | MgCl₂, MgSO₄, Mg citrate, Mg L-threonate | Ionic studies, in vitro applications, and in vivo supplementation |
| Cryo-EM Equipment | High-end microscope (e.g., Titan Krios) with direct electron detectors | High-resolution structure determination of Mg²⁺-bound states |
| Two-Electrode Voltage Clamp | Oocyte expression system with voltage control | Electrophysiological characterization of voltage-dependent Mg²⁺ block |
| Site-Directed Mutagenesis Kits | Q5 Site-Directed Mutagenesis Kit or equivalent | Binding site validation through targeted residue substitution |
| Sleep Assessment Tools | Polysomnography, actigraphy, PSQI, ISI | Objective and subjective sleep quality quantification |
| Molecular Dynamics Software | GROMACS, NAMD, or AMBER | Simulation of Mg²⁺ binding dynamics and receptor interactions |
Implications for Therapeutic Development and Future Research
The precise mechanistic understanding of magnesium's action on NMDA and GABA receptors provides a robust foundation for targeted therapeutic development in sleep medicine. Several promising directions emerge:
Bioavailable Formulations: The enhanced efficacy of magnesium-L-threonate in improving sleep architecture, particularly deep and REM sleep, suggests that blood-brain barrier penetration is a critical factor for central nervous system effects [105]. Future formulations should prioritize optimized brain delivery.
Subtype-Selective NMDA Receptor Modulation: The identification of GluN2B-specific magnesium potentiation sites [104] opens possibilities for developing compounds that enhance magnesium's beneficial effects while minimizing side effects associated with broad NMDA receptor inhibition.
Chronotherapeutic Approaches: Given magnesium's role in regulating circadian rhythms and cellular biological clocks [99], timed supplementation strategies aligned with individual circadian phases may enhance efficacy for specific sleep disorders.
Biomarker-Driven Supplementation: Identification of biochemical markers predictive of magnesium responsiveness (e.g., specific NMDA receptor polymorphisms, magnesium deficiency indicators) could enable personalized treatment approaches for sleep disorders.
The accumulated evidence positions magnesium not merely as a nutritional supplement but as a fundamental modulator of neurochemical pathways essential for sleep physiology. Future research integrating structural biology, genetics, and chronobiology will further elucidate magnesium's role in sleep and facilitate the development of novel therapeutics targeting magnesium-dependent pathways for sleep disorders.
Sleep disorders are increasingly recognized as a critical public health issue linked to significant cognitive decline and an elevated risk of neurodegenerative diseases. This whitepaper delves into the core biochemical pathways underlying this relationship, with a specific focus on the dual roles of adenosine signaling and neuroinflammation. Evidence consolidates that sleep disruption triggers a cascade of events: the accumulation of adenosine, activation of microglial cells via specific receptor subtypes (e.g., A2A), sustained release of pro-inflammatory cytokines, astrocyte dysfunction, and impaired synaptic plasticity. These processes collectively drive cognitive impairment. Furthermore, a vicious cycle emerges where neuroinflammation and neurotransmitter imbalance reinforce each other, accelerating neuronal damage. This document provides a comprehensive technical guide for researchers and drug development professionals, summarizing key quantitative data, illustrating critical signaling pathways, detailing experimental methodologies, and cataloging essential research reagents. The goal is to furnish the scientific community with a structured framework to advance the development of targeted therapeutic interventions.
Sleep is a fundamental physiological process governed by a complex interplay of neurochemical systems, chief among them being the adenosinergic system. Adenosine is an endogenous purine nucleoside that functions as a key sleep-regulatory substance, with extracellular concentrations in the brain rising during prolonged wakefulness and declining during sleep [108]. This accumulation during wakefulness promotes sleep drive, or the homeostatic need for sleep, and is a crucial component of the body's sleep-wake cycle [109]. Beyond its role in energy metabolism as a breakdown product of ATP, adenosine exerts its effects through four G protein-coupled receptor subtypes—A1, A2A, A2B, and A3—which are distributed throughout the central nervous system (CNS) and on immune cells [110].
When sleep is disrupted, whether acutely or chronically, this finely tuned system is thrown into disequilibrium. The resultant adenosine signaling dysregulation is closely intertwined with the activation of the brain's innate immune response, leading to neuroinflammation. This state is characterized by the activation of microglia and astrocytes, leading to the release of pro-inflammatory cytokines such as TNF-α, and the initiation of oxidative stress pathways [111]. This neuroinflammatory milieu is detrimental to cognitive processes; it disrupts neurotransmitter homeostasis, impairs synaptic plasticity, and can lead to neuronal damage [111] [112]. Understanding the mechanistic links between adenosine, neuroinflammation, and cognitive dysfunction is therefore paramount for developing novel therapeutic strategies aimed at preserving brain health in individuals with sleep disorders.
Adenosine Signaling in Sleep Homeostasis and Pathology
Adenosine Origin, Metabolism, and Receptor Signaling
The intra- and extracellular concentrations of adenosine are tightly controlled by complex regulatory processes dependent on the metabolic state of neurons and astrocytes [108]. The primary source of extracellular adenosine is the catalytic product of 5′-ecto-nucleotidases (5′-ENs), which degrade ATP released alongside glutamate from activated astrocytes—a process termed "gliotransmission" that is triggered in proportion to sleep need [108]. Key enzymes and transporters regulate its availability: Adenosine kinase (AdK) phosphorylates adenosine to AMP, effectively controlling its concentration, while adenosine deaminase (ADA) breaks down adenosine to inosine. Bi-directional equilibrative nucleoside transporters (ENTs 1–4) shuttle adenosine between intra- and extracellular compartments [108].
Adenosine exerts its diverse physiological effects by signaling through its four receptor subtypes, which have distinct affinities, distributions, and downstream signaling pathways, as summarized in the table below.
Table 1: Adenosine Receptor Subtypes: Distribution, Signaling, and Function
| Receptor Subtype | G Protein Coupling | Primary CNS Distribution | Affinity for Adenosine | Key Functions in Sleep & Neuroinflammation |
|---|---|---|---|---|
| A1 Receptor | Gi/o | Cortex, Cerebellum, Hippocampus [110] | High (nM range) [110] | Inhibits neurotransmitter release; promotes sleep; generally anti-inflammatory [108] [110] |
| A2A Receptor | Gs/Golf | Striatum, Nucleus Accumbens, Olfactory Tubercle [110] | High (nM range) [110] | Promotes sleep drive; strongly pro-inflammatory; modulates dopamine signaling [113] [108] |
| A2B Receptor | Gs (also Gq) | Widely expressed at low levels (e.g., blood vessels) [110] | Low (µM range) [110] | Activated under high adenosine (stress); contributes to inflammation [110] |
| A3 Receptor | Gi/o (also Gq) | Various brain regions at low levels [110] | High (nM range in humans) [110] | Role in inflammation is complex and context-dependent [110] |
The Adenosine-Caffeine Interaction
Caffeine, the most widely consumed psychoactive substance, is a non-selective antagonist of adenosine receptors. Its wake-promoting and cognitive-enhancing effects are primarily achieved by blocking A1 and A2A receptors in the brain [108] [109]. By preventing adenosine from binding to its receptors, caffeine deters the brain's natural sleep-inducing and inflammatory signals, thereby promoting the release of wake-promoting neurotransmitters. Chronic caffeine consumption can lead to tolerance, likely due to the compensatory upregulation of adenosine receptors [108]. Furthermore, emerging evidence suggests that caffeine can influence the circadian clock, potentially by antagonizing adenosine receptors in the suprachiasmatic nucleus (SCN), thereby delaying circadian phases [108].
Neuroinflammatory Pathways Activated by Sleep Disruption
Sleep deprivation (SD) is a potent trigger for neuroinflammation, which is now established as a key mechanism linking sleep loss to cognitive impairment [111] [114]. The following diagram illustrates the core signaling pathways involved.
Figure 1: Core Signaling Pathways from Sleep Depruption to Cognitive Impairment
The pathways outlined above involve several key cellular and molecular events:
- Microglial Activation: Microglia, the resident immune cells of the CNS, are rapidly activated in response to SD. This activation is often mediated via the NF-κB pathway, leading to the production and release of pro-inflammatory cytokines like TNF-α and IL-1β [111] [113]. The A2A receptor plays a critical role in this process, as its genetic inactivation can markedly inhibit HH-induced microglial accumulation and TNF-α production [113].
- Astrocyte Dysfunction: Astrocytes, essential for neuronal support and synaptic maintenance, become dysfunctional under conditions of SD. This disrupts ionic homeostasis, neurotransmitter recycling, and the provision of metabolic substrates to neurons, further contributing to cognitive decline [111].
- Glymphatic System Dysfunction: The glymphatic system, a brain-wide perivascular network that facilitates the clearance of neurotoxic waste products, is primarily active during sleep. SD disrupts this clearance, leading to the accumulation of harmful metabolites like beta-amyloid, which in turn exacerbates neuroinflammation and neuronal damage [111].
- Oxidative Stress & Gut-Brain Axis: SD induces oxidative stress, overwhelming the brain's antioxidant defenses and leading to neuronal damage. Furthermore, SD can disrupt the gut-brain axis, altering gut microbiota and increasing intestinal permeability, which allows peripheral inflammatory molecules to enter the circulation and further potentiate neuroinflammation [111].
Quantitative Data and Experimental Evidence
Key Biochemical Alterations in Human Subjects
Human studies have consistently demonstrated that sleep disorders lead to measurable biochemical changes that underscore the link to metabolic and cognitive dysfunction.
Table 2: Biochemical Parameters in Young Adults with Disordered Sleep vs. Normal Sleep [115]
| Biochemical Parameter | Normal Sleep Group (Mean ± SD) | Disordered Sleep Group (Mean ± SD) | p-value |
|---|---|---|---|
| Fasting Blood Glucose (mg/dL) | 90.2 ± 8.4 | 102.6 ± 10.1 | < 0.001 |
| Insulin (µU/mL) | 7.5 ± 2.3 | 11.1 ± 3.0 | < 0.01 |
| HOMA-IR | Not Reported | Not Reported | < 0.01 |
| Cortisol | Not Reported | Not Reported | < 0.05 |
| Triglycerides | Not Reported | Not Reported | < 0.05 |
| Leptin | Not Reported | Significantly Lower | < 0.05 |
| HDL | Not Reported | Significantly Lower | < 0.05 |
Key In Vivo Experimental Models and Findings
Animal models have been instrumental in elucidating the causal mechanisms linking adenosine, neuroinflammation, and cognitive decline.
Table 3: Key Experimental Findings from Preclinical Models
| Experimental Model | Intervention / Genotype | Key Cognitive & Molecular Findings | Source |
|---|---|---|---|
| Mouse model of Acute Hypobaric Hypoxia (HH) | Wild-type vs. A2A Receptor Knock-out (KO) | HH caused spatial memory impairment in wild-type mice; A2AR KO attenuated this impairment and reduced microglial activation and hippocampal TNF-α production. | [113] |
| Rat/Mouse model of Sleep Deprivation (SD) | Pharmacological A2A receptor antagonism | Inhibition of A2AR signaling reduced SD-induced neuroinflammation (microglial activation, cytokine release) and associated cognitive deficits. | [111] |
| Mouse model of SD | Natural compounds (e.g., Phloretin) | Phloretin alleviated SD-induced cognitive impairment by reducing inflammation via the PPARγ/NF-κB signaling pathway. | [111] |
The Scientist's Toolkit: Essential Research Reagents and Models
This section catalogs critical reagents, models, and methodologies used in this field to facilitate experimental design.
Table 4: Essential Research Reagents and Experimental Tools
| Reagent / Model / Tool | Specific Example(s) | Primary Function in Research |
|---|---|---|
| Adenosine Receptor Agonists | CGS 21680 (A2A selective) | To probe the effects of specific receptor activation on sleep, inflammation, and behavior. |
| Adenosine Receptor Antagonists | Caffeine (non-selective), SCH 58261 (A2A selective), KW-6002 (Istradefylline) | To investigate the physiological role of adenosine tone and to test therapeutic potential. |
| Genetic Knock-out Models | A2A receptor global KO mice; Cell-specific conditional KO mice | To establish causal relationships between specific receptors and pathological phenotypes. |
| Sleep Deprivation Models | Modified Multiple Platform Method; Gentle Handling; Chronic Sleep Restriction setups | To experimentally induce sleep loss and study its physiological consequences. |
| Inflammation Assay Kits | ELISA for TNF-α, IL-1β, IL-6; IHC/IF antibodies for Iba1 (microglia), GFAP (astrocytes) | To quantify neuroinflammatory markers in tissue, CSF, or plasma. |
| Cognitive Behavioral Tests | Morris Water Maze, Y-Maze, Novel Object Recognition, Fear Conditioning | To assess learning, memory, and executive function in animal models. |
Detailed Experimental Protocols
To ensure reproducibility and provide a clear technical roadmap, this section outlines two foundational experimental protocols cited in the literature.
This protocol is designed to evaluate the contribution of A2A receptor-mediated neuroinflammation to cognitive impairment under acute hypobaric hypoxia, a condition that mimics sleep apnea-related hypoxia.
1. Animals and Groups:
- Use adult male wild-type (WT) and A2A receptor knock-out (A2AR KO) mice.
- Divide mice into four experimental groups: (1) WT Normoxia, (2) WT Hypoxia, (3) A2AR KO Normoxia, (4) A2AR KO Hypoxia (n=10-12 per group).
2. Hypobaric Hypoxia (HH) Exposure:
- Place the hypoxia groups in a hypobaric chamber.
- Simulate an altitude of 8000 meters (approx. 282 mmHg) for 7 consecutive days, 24 hours per day.
- Maintain control (normoxia) groups at normal atmospheric pressure.
3. Behavioral Testing (During HH Exposure):
- Morris Water Maze (MWM): Conduct over the final 5 days of HH to assess spatial learning and memory.
- Days 1-4: Training phase with 4 trials per day. Record escape latency to find the hidden platform.
- Day 5: Probe trial. Remove the platform and record time spent in the target quadrant, a measure of spatial memory retention.
- Open Field Test: Perform on a day separate from MWM training. Record total distance traveled and time spent in the center to assess locomotor activity and anxiety-like behavior.
4. Tissue Collection and Molecular Analysis:
- Euthanize animals following behavioral testing and perfuse transcardially with ice-cold PBS.
- Dissect the hippocampus and cortex.
- Immunohistochemistry: Fix brain sections and perform double-labeling immunofluorescence for A2A receptors (e.g., anti-A2AR antibody) and microglial marker Iba1. Use confocal microscopy to confirm A2AR expression on microglia.
- ELISA: Homogenize hippocampal tissue. Use a commercial mouse TNF-α ELISA kit to quantitatively measure the levels of this pro-inflammatory cytokine.
5. Data Analysis:
- Compare MWM performance and TNF-α levels between WT and A2AR KO mice under hypoxic conditions using two-way ANOVA followed by post-hoc tests.
- Correlate microglial activation markers with cognitive performance metrics.
The workflow of this integrated protocol is visualized below.
Figure 2: Experimental Workflow for A2A Receptor Role in Hypoxia
This protocol outlines the methodology for testing the efficacy of pharmacological agents, such as the natural compound Phloretin, in mitigating SD-induced cognitive impairment and neuroinflammation.
1. Animal Model and Sleep Deprivation:
- Use adult rodents (mice or rats).
- Induce chronic sleep deprivation for 5-7 days using a validated method like the Modified Multiple Platform Method (placing animals over water in an apparatus that forces movement upon muscle atonia).
2. Drug Administration:
- Administer the test compound (e.g., Phloretin) or vehicle via intraperitoneal injection or oral gavage daily, 30-60 minutes before the start of the SD period.
3. Cognitive and Molecular Phenotyping:
- Behavioral Testing: After the SD period, assess cognitive function using the Y-maze or Novel Object Recognition test to evaluate working memory and recognition memory.
- Molecular Analysis: Euthanize animals and collect brain tissue.
- Perform Western Blot or ELISA to measure the activation of key signaling pathways (e.g., phosphorylation of NF-κB p65) and levels of pro-inflammatory cytokines (IL-1β, TNF-α) in the prefrontal cortex and hippocampus.
- Use immunohistochemistry to assess microglial (Iba1) and astrocytic (GFAP) activation.
The evidence is compelling: adenosine signaling and neuroinflammation are central, interconnected pathways through which sleep disorders lead to cognitive dysfunction. The A2A receptor has emerged as a particularly promising therapeutic target, with genetic and pharmacological inhibition showing efficacy in preclinical models of sleep disruption and hypoxia [111] [113]. However, critical challenges remain. The limitations of recovery sleep in fully reversing molecular and cognitive deficits highlight the potential for irreversible damage after chronic sleep disruption [111]. Future research must focus on several key areas: 1) Deconvoluting the bidirectional relationship between neurotransmitter systems and neuroinflammation; 2) Exploring multi-axis regulation, including the gut-brain axis, in sleep disorders; 3) Developing clinically viable compounds that can cross the blood-brain barrier to target adenosinergic signaling without systemic side-effects [111] [110] [109]. The integration of emerging technologies—such as organoids, single-cell omics, and advanced neuroimaging—with well-defined preclinical protocols will be crucial for translating this mechanistic understanding into effective therapies for the growing population suffering from sleep-related cognitive decline.
Comparative Analysis of Therapeutic Mechanisms and Emerging Targets
GABAergic agents, primarily benzodiazepines (BZDs) and Z-drugs (non-benzodiazepine hypnotics, also known as BZRAs), represent a cornerstone in the pharmacotherapy of insomnia and anxiety disorders. These agents function by modulating the gamma-aminobutyric acid (GABA) system, the principal inhibitory neurotransmitter pathway in the central nervous system (CNS). Within the broader context of sleep biochemistry research, understanding the comparative efficacy, safety, and mechanistic profiles of these drugs is paramount for developing safer, more targeted therapeutics. Despite their widespread use, global prescribing trends reveal significant concerns regarding dependency, high-risk use, and varying national practices [116]. This whitepaper provides an in-depth technical analysis of BZDs and Z-drugs, synthesizing current evidence on their mechanisms, direct efficacy comparisons, and safety profiles to inform researchers and drug development professionals.
Mechanisms of Action: GABAergic Neurotransmission and Pharmacological Modulation
The therapeutic and adverse effects of BZDs and Z-drugs are inextricably linked to their action on the GABAergic system. GABA is the main inhibitory neurotransmitter in the mammalian CNS, with an estimated 60-75% of all synapses being GABAergic [117]. Its primary role is to counterbalance excitatory neurotransmission, thereby maintaining neuronal homeostasis.
The GABAergic System and Sleep-Wake Regulation
Sleep is an actively regulated process governed by complex neural circuits. The sleep-wake cycle is maintained by a delicate balance between arousal-promoting neurotransmitters (e.g., glutamate, noradrenaline, serotonin, acetylcholine, orexin, and dopamine) and sleep-promoting neurotransmitters, primarily GABA and adenosine [118]. GABAergic neurons, concentrated in the basal forebrain and anterior hypothalamus, become active during sleep, releasing GABA to inhibit brain regions that stimulate arousal, such as the tuberomammillary nucleus (TMN), raphe nucleus, and locus coeruleus [118] [119]. This inhibition is crucial for the initiation and maintenance of sleep, particularly non-rapid eye movement (NREM) sleep.
Molecular Pharmacology of GABAA Receptors
Both BZDs and Z-drugs exert their effects by binding to a specific site on the GABAA receptor (GABAAR), a ligand-gated ion channel. The GABAAR is a heteropentameric structure, typically composed of two α, two β, and one γ subunit (e.g., α1, β2, γ2) [1]. These subunits form a central pore that allows the passage of chloride ions (Cl⁻) upon activation.
- Receptor Binding and Allosteric Modulation: BZDs and Z-drugs are positive allosteric modulators. They bind to a site distinct from the endogenous GABA binding site, which is located at the interface between an α and γ subunit [1]. This binding does not directly open the ion channel but induces a conformational change that enhances the affinity of GABA for its binding site. When GABA subsequently binds, the frequency of channel-opening events increases, leading to a greater influx of Cl⁻ ions into the neuron [2] [120].
- Cellular and Systemic Effects: The increased Cl⁻ influx hyperpolarizes the postsynaptic neuron, moving its membrane potential further from the firing threshold. This hyperpolarization makes the neuron less responsive to excitatory stimuli, resulting in CNS depression [1]. This foundational mechanism underlies the anxiolytic, sedative-hypnotic, muscle-relaxant, and anticonvulsant properties of these drugs. The specific pharmacological profile—whether a compound is more sedating or more anxiolytic—is largely determined by its binding affinity and selectivity for GABAAR subtypes containing different α subunits [120].
Table 1: Key Subunits of the GABAA Receptor and Their Functional Roles
| Subunit | Primary Localization | Functional Role & Pharmacological Significance |
|---|---|---|
| α1 | Widespread, high density in cortex, thalamus | Mediates sedative and amnestic effects; primary target for Z-drugs [118] |
| α2 | Limbic system, hippocampus, amygdala | Mediates anxiolytic effects [118] |
| α3 | Cerebral cortex, reticular activating system | May influence arousal and muscle relaxation [118] |
| α5 | Hippocampus | Associated with memory processes and potential cognitive side effects [118] |
| β1-3 | Various brain regions | Structural components; the β-subunit contains the GABA binding site [1] |
| γ2 | Synaptic sites | Essential for benzodiazepine binding and fast inhibitory neurotransmission [1] |
The following diagram illustrates the molecular mechanism of GABAAR modulation by BZDs/Z-drugs, from synaptic binding to neuronal inhibition.
Comparative Efficacy Analysis: Bayesian Network Meta-Regression Evidence
Direct comparisons of pharmacotherapies for insomnia require robust methodologies to account for multiple interventions and confounding variables. A recent Bayesian network meta-regression (NMR) analysis of randomized controlled trials (RCTs) provides high-quality evidence for the comparative efficacy of various GABAergic agents and newer drugs, using objective polysomnography (PSG) data to minimize bias [121].
Experimental Protocol for Efficacy Evaluation
The cited NMR followed a rigorous protocol [121]:
- Data Sources & Search Strategy: Researchers retrieved relevant RCTs from PubMed, Embase, Scopus, Cochrane Library, Web of Science, and ClinicalTrials.gov from inception to April 15, 2025.
- Inclusion/Exclusion Criteria: The analysis included RCTs evaluating pharmacotherapies for insomnia disorder, reporting PSG outcomes like wake after sleep onset (WASO), latency to persistent sleep (LPS), total sleep time (TST), and sleep efficiency (SE).
- Statistical Analysis: A Bayesian NMR was performed using RStudio 4.4.2. The standard mean difference (SMD) was calculated for PSG outcomes. Analyses were adjusted for critical confounding factors, including follow-up period and patient age, to generate "time windows" of efficacy.
- Outcomes Measured: Primary efficacy endpoints were WASO, LPS, TST, and SE.
Efficacy Findings and Interpretation
The analysis included 15 studies evaluating 10 treatment regimens involving 2,408 patients. The following table synthesizes the key efficacy findings against placebo for the critical sleep maintenance parameter, WASO [121].
Table 2: Comparative Efficacy of Insomnia Pharmacotherapies on Wake After Sleep Onset (WASO) vs. Placebo (Bayesian NMR)
| Drug (Dose) | Standardized Mean Difference (SMD) | 95% Credible Interval | Clinical Interpretation |
|---|---|---|---|
| Daridorexant (25 mg/d) | -0.957 | -1.436 to -0.479 | Large, statistically significant improvement |
| Melatonin (6 mg/d) | -0.741 | -1.423 to -0.044 | Significant improvement (significance lost after follow-up adjustment) |
| Lemborexant (10 mg/d) | -0.624 | -0.894 to -0.355 | Medium-large, statistically significant improvement |
| Lemborexant (5 mg/d) | -0.612 | -0.880 to -0.342 | Medium-large, statistically significant improvement |
| Doxepin (3 mg/d) | -0.497 | -0.713 to -0.282 | Medium, statistically significant improvement |
| Dimdazenil (2.5 mg/d) | -0.388 | -0.608 to -0.166 | Small-medium, statistically significant improvement |
| Zolpidem (10 mg/d) | -0.348 | -0.610 to -0.068 | Small-medium, statistically significant improvement |
Key Interpretations from Efficacy Data:
- Superiority of Dual Orexin Receptor Antagonists (DORAs): Daridorexant and lemborexant demonstrated medium to large effect sizes, with daridorexant 25 mg showing the greatest improvement in WASO. The study concluded that for insomnia characterized by difficulty maintaining sleep, daridorexant 25 mg/d should be prioritized [121].
- Performance of Z-drugs: Zolpidem 10 mg showed a significant but smaller effect (SMD: -0.348) compared to several DORAs and even the antidepressant doxepin. Dimdazenil, a partial positive allosteric modulator, also showed a modest effect.
- The "Time Window" of Melatonin: Melatonin's significant superiority was sensitive to the follow-up period, demonstrating a specific "time window" of efficacy from the 10th to 40th weeks, highlighting the importance of adjusting for confounders in analysis [121].
- Subtype-Specific Recommendations: The study proposed a tailored strategy: for sleep-onset insomnia, lemborexant 10 mg/day or zolpidem 10 mg/day were recommended, while for overall poor sleep efficiency, lemborexant was the top choice [121].
Safety and Pharmacovigilance Profile
While efficacy is crucial, the risk-benefit profile of GABAergic agents is heavily influenced by their safety and tolerability. Evidence comes from both large-scale prescription pattern studies and pharmacovigilance analyses of adverse event databases.
Prescription Trends and High-Risk Use
An observational study of dispensings in Ireland (2014-2022) provides real-world data on utilization and risks [116]:
- Overall Use: The rate of BZD and Z-drug dispensings in Ireland decreased by 5% (from 1531 to 1474 per 1000 eligible population), a less pronounced decline than in England, which saw a 27% decrease. This suggests persistent, high-volume use in some regions [116].
- High-Risk Dispensings: The study noted that "High-risk dispensings of benzodiazepines and z-drugs decreased over the study period." This includes co-prescription with other sedatives or anticholinergics (increasing fall risk) and high-dose use (increasing dependency risk) [116].
- Chronic Use in Older Adults: Ireland has consistently had the second-highest rate of chronic BZD use among older adults among OECD countries, a indicator of potentially inappropriate prescribing, though this has also seen a recent decrease [116].
Analysis of Adverse Event Reporting
The previously cited NMR study also performed a pharmacovigilance investigation by leveraging the FDA Adverse Event Reporting System (FAERS) database [121].
- Nervous System Disorders: Safety signals for nervous system disorders (e.g., dizziness, somnolence) were detected for DORAs: suvorexant (IC025 = 0.212), lemborexant (IC025 = 0.221), and daridorexant (IC025 = 0.205). The antidepressant doxepin also showed a signal (IC025 = 0.066) [121].
- Dyspnea: Analysis of dyspnea (shortness of breath) revealed that eszopiclone (a Z-drug) had a significantly lower constituent ratio than daridorexant, melatonin, and zolpidem. Conversely, melatonin and zolpidem had a significantly higher reporting ratio of dyspnea than suvorexant [121].
- Dependence and Withdrawal: A primary danger of regular BZD/Z-drug use is dependence, which can develop after just two weeks of daily use. Withdrawal can be severe and, unlike opioid withdrawal, can be fatal. The risk is higher with more potent drugs, higher doses, and longer duration of use [120].
Table 3: Key Safety Considerations for GABAergic Agents
| Risk Category | Benzodiazepines | Z-Drugs | Comments and Evidence |
|---|---|---|---|
| Dependence & Withdrawal | High risk | High risk | Can occur after ~2 weeks of daily use. Withdrawal can be fatal [120]. |
| Cognitive Impairment | Debated link to dementia; clear short-term impairment | Similar risk profile | Conflicting data on long-term use and dementia [120]. |
| Fall Risk | Significantly increased, especially in elderly | Significantly increased | Due to sedative and muscle-relaxant effects [116] [120]. |
| Nervous System AEs | Common (drowsiness, dizziness) | Common (drowsiness, dizziness) | FAERS data shows signal for DORAs and doxepin [121]. |
| Respiratory Effects | - | Dyspnea reported with zolpidem | Eszopiclone had lower dyspnea reports than zolpidem [121]. |
| "As-Needed" Use | May reduce cumulative exposure and dependence | Suitable for sleep-onset insomnia | Research on PRN regimens is limited but clinically common [120]. |
Research Gaps and Future Directions
The current evidence base for GABAergic agents, while substantial, contains critical gaps that must be addressed through targeted research.
- As-Needed and Intermittent Dosing: There is a pronounced lack of research into the safety and efficacy of as-needed (PRN) and intermittent prescription, which is how these drugs are often used in practice for anxiety and situational insomnia [120]. This gap is problematic as PRN use could potentially optimize benefit-risk ratios by minimizing cumulative exposure.
- Long-Term Daily Use vs. Novel Agents: The debate around the safety and appropriateness of long-term daily BZD use for anxiety continues, with some authorities viewing the risks as unacceptable and others defending its use, particularly when non-pharmacological alternatives are unavailable [120]. More long-term, head-to-head trials comparing BZDs/Z-drugs with newer agents like DORAs are needed.
- Molecular and Systems-Level Mechanisms: Further research is needed to fully elucidate how modulation of specific GABAAR subtypes translates to global sleep architecture changes and to explore non-GABAergic pathways for sleep modulation, such as the gut-brain axis [119].
The Scientist's Toolkit: Key Research Reagents and Methodologies
Research into GABAergic agents relies on a suite of specialized reagents, assays, and methodologies. The following table details essential tools for investigating the mechanisms, efficacy, and safety of these compounds.
Table 4: Essential Research Reagents and Tools for GABAergic Agent Investigation
| Research Reagent / Tool | Function and Application in GABA Research |
|---|---|
| Radioligand Binding Assays | Used to quantify receptor binding affinity (Kd) and density (Bmax) of BZDs/Z-drugs for recombinant or native GABAARs using radioactively labeled ligands (e.g., [³H]Flunitrazepam). |
| Electrophysiology (Patch-Clamp) | The gold-standard functional assay for measuring chloride current kinetics in neurons or recombinant cells expressing GABAARs, directly testing the potency and efficacy of allosteric modulators. |
| Recombinant GABAAR Subtypes | Cell lines (e.g., HEK293, Xenopus oocytes) engineered to express specific combinations of human GABAAR subunits (e.g., α1β2γ2, α5β3γ2) to determine subunit selectivity and pharmacological profile of novel compounds. |
| Polysomnography (PSG) | Objective multi-parameter sleep study measuring EEG, EOG, and EMG. The primary method for assessing drug efficacy on sleep architecture (e.g., WASO, LPS, TST, SE) in clinical trials [121]. |
| Bayesian Network Meta-Regression | Advanced statistical methodology that integrates data from multiple RCTs to provide comparative efficacy and safety estimates while adjusting for confounders like follow-up time and age [121]. |
| FAERS Database | A publicly available pharmacovigilance database containing millions of adverse event reports. Used for post-market safety signal detection and disproportionality analysis [121]. |
| Animal Models (e.g., Kv1.1-/- mice) | Genetically modified or pharmacologically induced models of sleep disorders or GABA dysfunction used for in vivo efficacy and safety screening before human trials [118]. |
Benzodiazepines and Z-drugs remain potent tools for managing insomnia and anxiety through their action on the GABAergic system. However, evidence from recent network meta-analyses and pharmacovigilance studies indicates that newer agents like DORAs may offer superior efficacy for sleep maintenance, with a distinct, though not absent, adverse event profile. The significant risks of dependence, cognitive impairment, and falls associated with traditional GABAergic agents underscore the need for cautious, short-term prescribing and the exploration of PRN regimens. Future research must prioritize head-to-head trials, investigate intermittent dosing strategies, and continue the pursuit of novel mechanisms that target the sleep-wake cycle with greater precision and improved safety, thereby advancing the core mission of biochemistry and sleep disorder research.
The therapeutic landscape for sleep disorders has expanded significantly with the development of targeted pharmacological agents that modulate the melatonin signaling pathway. This whitepaper provides a comprehensive technical analysis comparing exogenous melatonin with synthetic melatonin receptor agonists (ramelteon, tasimelteon, agomelatine, and prolonged-release melatonin), examining their distinct pharmacokinetic properties, receptor binding affinities, molecular mechanisms of action, and clinical applications. Within the broader context of sleep biochemistry research, we present structured quantitative comparisons, detailed experimental methodologies, and visualizations of signaling pathways to inform drug development strategies and clinical trial design for researchers and pharmaceutical professionals.
Sleep-wake cycles are regulated by complex neurobiological systems, with the circadian timing system and sleep homeostasis representing two fundamental pillars. The suprachiasmatic nucleus (SCN) of the hypothalamus serves as the master circadian pacemaker, coordinating physiological rhythms with the 24-hour light-dark cycle. Melatonin (N-acetyl-5-methoxytryptamine), an indoleamine hormone secreted predominantly by the pineal gland during darkness, serves as a key hormonal output of the circadian system, transmitting timing information to distributed oscillators throughout the body [122] [123]. The decline in endogenous melatonin production associated with aging, particularly in individuals over 55, has been linked to sleep maintenance difficulties and poor sleep quality [122].
The therapeutic targeting of melatonin signaling represents a paradigm shift from conventional GABAergic hypnotics toward chronobiotic approaches that restore circadian organization. While exogenous melatonin supplements attempt to replace declining hormone levels, synthetic melatonin receptor agonists offer enhanced pharmacokinetic profiles and receptor selectivity. Understanding the distinctions between these therapeutic approaches is essential for advancing targeted treatments for circadian rhythm sleep-wake disorders (CRSWDs) and comorbid insomnia [124] [125].
Mechanisms of Action: Receptor Physiology and Signaling Pathways
Melatonin Receptor Subtypes and Distribution
Melatonin exerts its effects primarily through two high-affinity G protein-coupled receptors: MT1 (Mel1a) and MT2 (Mel1b). These receptors are expressed in various tissues, with highest concentration in the SCN, where they regulate circadian timing and sleep processes [125] [123].
- MT1 Receptors: Predominantly located in the SCN, hippocampus, substantia nigra, cerebellum, central dopaminergic pathways, and peripheral tissues including retina, ovary, testis, coronary arteries, and immune cells. MT1 receptor activation inhibits neuronal firing in the SCN and mediates vasoconstriction in peripheral arteries [125] [126].
- MT2 Receptors: Expressed in the SCN, retina, lung, heart, aorta, myometrium, immune cells, and adipose tissue. MT2 receptor activation phase-shifts circadian rhythms and mediates retinal neurotransmission and vasodilation [125] [123].
Preclinical studies indicate that MT2 receptors are critical for phase-shifting the circadian clock, while MT1 activation inhibits SCN activity, potentially affecting downstream sleep circuits [123]. Both receptors signal through Gαi/o proteins, inhibiting adenylate cyclase and reducing intracellular cAMP levels. Additional signaling pathways include inhibition of cAMP-responsive element binding protein (CREB) phosphorylation, activation of potassium channels, and inhibition of voltage-gated calcium channels [125].
Signaling Pathway Visualization
The following diagram illustrates the molecular signaling pathways of melatonin receptor activation:
Diagram 1: Melatonin receptor signaling pathways. MT1 and MT2 receptors couple primarily to Gαi/o proteins, inhibiting adenylate cyclase (AC) and reducing cAMP production. Additional signaling through Gαq activates phospholipase C (PLC), generating IP3 and DAG, which activate protein kinase C (PKC). MT1 activation also modulates potassium and calcium channels, reducing neuronal firing in the SCN. MT2 receptors primarily mediate circadian phase-shifting effects.
Comparative Pharmacokinetic Profiles
Structured Pharmacokinetic Comparison
Table 1: Comparative pharmacokinetic properties of exogenous melatonin and melatonin receptor agonists
| Compound | Receptor Selectivity | Bioavailability | Tmax (h) | Half-life (h) | Active Metabolites | Key Metabolic Pathways |
|---|---|---|---|---|---|---|
| Melatonin | MT1, MT2 (non-selective) | 1.8-15% [122] | 0.75-3 [122] | 0.5-1 [126] | None significant | Hepatic CYP1A2, 2C19 [122] |
| Ramelteon | MT1, MT2 (selective) | 1.8% [122] | 0.5-1.5 [122] | 1.0-2.6 (parent), 2-5 (M-II) [122] | M-II (potent) | Hepatic CYP1A2 [122] |
| Tasimelteon | MT1, MT2 (selective) | Not specified | 0.5-3 [123] | 0.9-1.7 [123] | Multiple | Hepatic CYP3A4, 2C9 [123] |
| Agomelatine | MT1, MT2 + 5-HT2C antagonism | <5% [124] | 1-2 [124] | 1-2 [124] | None significant | Hepatic CYP1A2, 2C9 [124] |
| Prolonged-release Melatonin | MT1, MT2 (non-selective) | ~15% [122] | 0.75 (fasting), 3 (fed) [122] | 3.5-4.0 [122] | None significant | Hepatic conjugation [122] |
Pharmacokinetic Interpretation
The pharmacokinetic data reveal significant differences between agents that inform their clinical application. Exogenous melatonin demonstrates variable absorption depending on formulation, with immediate-release preparations having rapid Tmax (0.75 hours) and short half-life (0.5-1 hour), making them suitable primarily for sleep onset difficulties [126]. The development of prolonged-release melatonin (Circadin) addresses this limitation with a Tmax of 3 hours under fed conditions and extended half-life of 3.5-4.0 hours, better mimicking physiological melatonin secretion patterns [122].
Synthetic agonists exhibit distinct profiles: ramelteon has low bioavailability (1.8%) due to extensive first-pass metabolism but generates an active metabolite (M-II) with longer half-life (2-5 hours) that circulates at 10- to 100-fold higher concentrations than the parent compound [122]. Tasimelteon demonstrates intermediate half-life (0.9-1.7 hours) suitable for both phase-shifting and sleep initiation [123]. Agomelatine shows similar pharmacokinetics to immediate-release melatonin but possesses dual mechanism of action through additional 5-HT2C receptor antagonism [124].
Clinical Efficacy and Applications
Structured Clinical Indications and Efficacy
Table 2: Clinical applications and efficacy evidence for melatonin and receptor agonists
| Compound | Approved Indications | Key Clinical Evidence | Specialized Applications |
|---|---|---|---|
| Melatonin | Dietary supplement (US), primary insomnia ≥55 years (EU as Circadin) [122] | Reduces sleep onset latency; improves sleep quality in elderly [122] [127] | Jet lag, shift work, children with neurodevelopmental disorders [126] |
| Ramelteon | Insomnia (sleep onset difficulty) [122] | Reduces sleep latency by 40 min (56%) at 6 months vs 30 min (43%) for placebo [122] | Delirium prevention, safe in mild sleep apnea and COPD [122] [126] |
| Tasimelteon | Non-24-hour sleep-wake disorder [123] | Phase-shifts circadian rhythm, decreases sleep latency when administered 5h before bedtime [123] | Blind individuals with non-24-hour disorder [123] |
| Agomelatine | Depression (with sleep disturbances) [124] | Improves sleep latency and quality in depressed patients [124] | Depression with comorbid insomnia, circadian rhythm disruption [124] |
| Prolonged-release Melatonin | Primary insomnia ≥55 years (EU) [122] | Improves sleep quality, morning alertness, and quality of life [122] | Long-term use (up to 3 months) in elderly insomnia [122] |
Critical Efficacy Analysis
Recent meta-analyses provide nuanced understanding of clinical efficacy. A 2025 systematic review and meta-analysis of 20 randomized controlled trials found that while melatonin and melatonin receptor agonists showed no statistically significant reduction in intensive care unit length of stay overall (p=0.298), significant benefits were observed in specific subgroups: coronary artery bypass graft surgery patients demonstrated reduced ICU length of stay (-0.47 days, 95% CI: -0.78 to -0.16, p=0.003) and COVID-19 patients showed reduced hospital length of stay (-3.90 days, 95% CI: -6.28 to -1.51, p=0.001) [128].
For primary insomnia, evidence supports the efficacy of melatonin receptor agonists particularly in sleep initiation. Ramelteon demonstrates sustained efficacy over 6 months with reduction in sleep onset latency of 40 minutes (56% from baseline) compared to 30 minutes (43%) for placebo [122]. The safety profile of these agents is generally favorable, with ramelteon being the only insomnia medication not classified as a controlled substance in the United States due to absence of abuse potential [122] [126].
Experimental Methodologies for Receptor Characterization
Receptor Binding Assays
Objective: To determine binding affinity (Ki) and selectivity of melatonin compounds for MT1 and MT2 receptors.
Protocol:
- Membrane Preparation: Harvest CHO-K1 or HEK-293 cells stably expressing human MT1 or MT2 receptors. Homogenize in ice-cold hypotonic buffer (5 mM Tris-HCl, 2 mM EDTA, pH 7.4) and centrifuge at 40,000 × g for 20 minutes. Resuspend membrane pellets in assay buffer (50 mM Tris-HCl, 5 mM MgCl2, 1 mM EDTA, pH 7.4) [125].
- Radioligand Binding: Incubate membrane preparations (50-100 μg protein) with [³H]-melatonin (0.1-10 nM) and increasing concentrations of test compounds (10⁻¹² to 10⁻⁵ M) in a total volume of 250 μL for 2-3 hours at 25°C [125].
- Separation and Detection: Rapidly vacuum filter through GF/B filters presoaked in 0.3% polyethyleneimine. Wash filters with ice-cold buffer and measure bound radioactivity by liquid scintillation counting [125].
- Data Analysis: Determine IC50 values using nonlinear regression. Calculate Ki values using Cheng-Prusoff equation: Ki = IC50 / (1 + [L]/KD), where [L] is radioligand concentration and KD is dissociation constant [125].
Functional Assays: GTPγS Binding
Objective: To assess functional efficacy through G-protein activation.
Protocol:
- Membrane Preparation: As in 5.1.
- GTPγS Binding: Incubate membranes with test compounds (10⁻¹² to 10⁻⁵ M) in assay buffer containing 0.1-0.5 nM [³⁵S]GTPγS, 10 μM GDP, and 100 mM NaCl for 60 minutes at 30°C [125].
- Detection: Terminate reactions by filtration through GF/B filters, wash, and measure bound radioactivity.
- Data Analysis: Calculate Emax (maximum efficacy) and EC50 (potency) using nonlinear regression. Compare to melatonin reference standard [125].
Phase Response Curve Determination
Objective: To characterize circadian phase-shifting properties of compounds.
Protocol:
- Animal Model: Use C57BL/6 mice (8-12 weeks) or Syrian hamsters maintained in constant darkness.
- Drug Administration: Inject test compounds at different circadian times (CT) determined by activity onset (CT0) or core body temperature rhythm.
- Phase Assessment: Monitor wheel-running activity or body temperature rhythm before and after treatment. Calculate phase shifts as difference in activity onset on cycles following treatment versus baseline.
- Data Analysis: Construct phase response curve by plotting magnitude and direction of phase shifts against CT of administration [123].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key research reagents for melatonin receptor research
| Reagent/Cell Line | Application | Key Features | Research Utility |
|---|---|---|---|
| CHO-MT1/HEK-MT1 | Receptor binding and signaling assays | Stably express human MT1 receptors | Determination of MT1 selectivity and potency |
| CHO-MT2/HEK-MT2 | Receptor binding and signaling assays | Stably express human MT2 receptors | Determination of MT2 selectivity and potency |
| [³H]-melatonin | Radioligand binding studies | High specific activity (>80 Ci/mmol) | Reference compound for competition binding |
| [³⁵S]GTPγS | Functional G-protein activation | Non-hydrolyzable GTP analog | Measurement of receptor activation and efficacy |
| 2-iodomelatonin | Reference agonist | High-affinity, metabolically stable | Positive control in functional assays |
| S26131/Luzindole | MT2-selective antagonist | 25-150-fold selectivity for MT2 over MT1 | Receptor subtype functional characterization |
| 4P-PDOT | MT2-selective antagonist | 300-fold selectivity for MT2 | Circadian phase-shifting mechanism studies |
Research Gaps and Future Directions
Despite advances in melatonin therapeutics, several research gaps remain. The precise individual contributions of MT1 versus MT2 receptors to specific sleep and circadian functions require further elucidation through development of more selective agonists and antagonists [125] [123]. Clinical evidence for melatonin agonists in certain CRSWDs, particularly delayed sleep-wake phase disorder, remains limited despite strong theoretical rationale [126] [123].
Future research directions should include:
- Development of receptor subtype-specific agonists to dissect MT1 versus MT2 functions
- Exploration of biased agonism to selectively target therapeutic pathways while minimizing side effects
- Personalization approaches based on circadian phenotype and genetic polymorphisms in melatonin receptors and metabolic enzymes
- Investigation of combinatory therapies targeting both circadian and sleep homeostatic systems
- Long-term safety and efficacy studies in vulnerable populations including children and elderly [124] [125] [123]
The integration of melatonin receptor agonists into treatment algorithms for comorbid conditions, particularly neurological disorders, cancer, and pain conditions, represents a promising research direction identified through bibliometric analysis of publication trends [129].
The strategic development of melatonin receptor agonists represents significant progress in sleep and circadian medicine, offering targeted alternatives to conventional hypnotics with improved safety profiles. The distinct pharmacokinetic and pharmacodynamic properties of these agents enable precise therapeutic applications, from sleep initiation to circadian rhythm realignment. Continued research into receptor subtype specificity, biased signaling, and personalized chronotherapeutic approaches will further enhance treatment precision for sleep and circadian disorders.
The discovery of the orexin (hypocretin) system has fundamentally reshaped the scientific understanding of sleep-wake regulation and created a new therapeutic paradigm for treating insomnia disorders. This hypothalamic neuropeptide system, consisting of orexin-A and orexin-B neuropeptides and their G-protein-coupled receptors OX1R and OX2R, functions as a critical stabilizer of wakefulness rather than a simple on/off switch [63] [130]. Orexin-producing neurons in the lateral hypothalamus project widely throughout the brain, integrating diverse physiological and environmental signals to coordinate arousal states [63]. In chronic insomnia, persistent overactivation of this orexin-driven arousal system underlies the characteristic difficulty with sleep initiation and maintenance [63]. Dual orexin receptor antagonists (DORAs) represent a mechanistically distinct class of insomnia therapeutics that target this specific pathophysiology by selectively blocking orexin receptors, thereby dampening wake-promoting signals without inducing broad neuronal suppression [131] [63]. This targeted approach maintains natural sleep architecture while minimizing the cognitive impairments and safety concerns associated with traditional GABAergic hypnotics [131] [63].
Neurobiological Basis of Orexin Signaling
Molecular Components of the Orexin System
The orexin system consists of two neuropeptides—orexin-A and orexin-B—derived from a common precursor polypeptide produced by neurons predominantly located in the lateral hypothalamus [63]. Orexin-A is a 33-amino acid peptide with four cysteine residues forming two intrachain disulfide bonds, while orexin-B comprises 28 amino acids and shares approximately 46% sequence identity with orexin-A [63]. These peptides bind to two G-protein-coupled receptors: orexin receptor 1 (OX1R) and orexin receptor 2 (OX2R) [63]. OX1R exhibits higher affinity for orexin-A, whereas OX2R binds both peptides with similar affinity [63]. The receptors demonstrate distinct signaling properties and downstream effects—OX1R predominantly modulates Na+/Ca2+ exchange, while OX2R enhances NMDA receptor transcription, suggesting potential influences on synaptic plasticity [63].
Neural Circuitry of Arousal Regulation
Orexin neurons project extensively throughout the brain, with particularly dense innervation of regions involved in arousal regulation, including the locus coeruleus (noradrenergic), dorsal raphe nucleus (serotonergic), tuberomammillary nucleus (histaminergic), and basal forebrain (cholinergic) [63]. This widespread connectivity positions the orexin system as a master coordinator that synchronizes the activity of multiple wake-promoting pathways [63]. The system integrates diverse inputs including metabolic cues, circadian signals, and emotional stimuli to determine arousal state transitions [63]. Under normal conditions, orexin neuronal activity peaks during active wakefulness, decreases during quiet wakefulness, and is virtually silent during sleep, particularly during rapid eye movement (REM) sleep [63].
Table 1: Key Components of the Orexin Signaling System
| Component | Type | Key Characteristics | Functional Significance |
|---|---|---|---|
| Orexin-A | Neuropeptide | 33-amino acids, two disulfide bonds | Higher affinity for OX1R; promotes wake stability |
| Orexin-B | Neuropeptide | 28-amino acids, linear structure | Equal affinity for OX1R and OX2R |
| OX1R | GPCR | Couples to Gq proteins; Na+/Ca2+ exchange | Modulates reward, addiction, and emotional arousal |
| OX2R | GPCR | Couples to Gq proteins; enhances NMDA receptor transcription | Primary mediator of sleep-wake transitions |
Pathophysiology of Orexin Dysregulation in Insomnia
In contrast to narcolepsy, where orexin deficiency causes unstable sleep-wake boundaries, chronic insomnia is characterized by persistent overactivity of the orexin system [63]. This hyperarousal state involves elevated orexin signaling during intended sleep periods, resulting in prolonged sleep latency, frequent awakenings, and non-restorative sleep [63]. Neuroimaging studies in insomnia patients demonstrate increased metabolic activity in orexin projection regions, consistent with this hyperarousal model [63]. The orexin system's role in translating motivational states into coordinated behaviors explains how stress and environmental cues can perpetuate insomnia through sustained orexin activation [130].
Pharmacological Profile of DORAs
Mechanism of Action: Targeted Arousal Modulation
DORAs function through competitive antagonism at both OX1R and OX2R receptors, selectively dampening orexin-driven arousal signals without potentiating inhibitory neurotransmission [131] [63]. This mechanism preserves natural sleep architecture by allowing sleep to occur through the normal physiological process of reduced arousal rather than through generalized neuronal suppression [131]. The targeted action contrasts sharply with GABAergic hypnotics, which non-selectively enhance inhibition throughout the central nervous system [131] [63]. By specifically modulating the wake-promoting system that is overactive in insomnia, DORAs reduce sleep latency and nocturnal awakenings while maintaining the normal cycling through sleep stages, including both non-REM and REM sleep [132] [63].
Comparative Pharmacokinetics of Approved DORAs
The clinical utility of DORAs is significantly influenced by their pharmacokinetic profiles, particularly elimination half-life, which affects both efficacy and next-day residual effects [69] [133] [134]. Currently, three DORAs are approved for clinical use—suvorexant, lemborexant, and daridorexant—with a fourth agent, vornorexant, in development with a notably shorter half-life [69] [133]. Vornorexant's rapid absorption and elimination (half-life comparable to zolpidem) positions it as a potential therapeutic option for sleep-onset difficulties with reduced risk of morning-after impairment [69] [133]. The expansion of pharmacokinetic diversity within the DORA class enables more precise matching of medication properties to individual sleep complaint patterns [69] [134].
Table 2: Pharmacokinetic Properties of Dual Orexin Receptor Antagonists
| Compound | Elimination Half-life (Hours) | Time to Peak Concentration (Hours) | Key Metabolic Pathways | Therapeutic Considerations |
|---|---|---|---|---|
| Suvorexant | 12-16 | 2-3 | CYP3A4 metabolism | Higher risk of next-day effects; evening administration crucial |
| Lemborexant | 17-19 | 1-2 | CYP3A4 metabolism, direct glucuronidation | Moderate duration; suitable for sleep maintenance |
| Daridorexant | 8-9 | 1.5-2 | CYP3A4 metabolism | Lower residual effects; balanced onset and duration |
| Vornorexant* | ~2-4 (estimated) | <1 (estimated) | Not fully characterized | Minimal next-day effects; primarily sleep initiation |
*Investigational compound
Clinical Efficacy and Safety Profiles
Effects on Sleep Architecture and Continuity
Clinical trials demonstrate that DORAs significantly improve key sleep parameters including sleep onset latency, wake after sleep onset, and total sleep time while preserving natural sleep architecture [135] [132] [63]. Polysomnographic studies reveal that DORAs primarily enhance the duration of rapid eye movement (REM) sleep while leaving non-REM sleep largely unchanged or slightly reduced [132]. This pattern contrasts with GABAergic agents, which typically suppress deeper sleep stages and disrupt normal sleep architecture [132] [63]. The preservation of natural sleep physiology likely underlies the reported improvements in morning alertness and cognitive performance despite increased total sleep time [132] [63].
Safety and Tolerability Across Populations
DORAs exhibit favorable safety profiles with low abuse potential, minimal withdrawal symptoms, and limited impact on respiratory function [136] [63]. Real-world data from the FDA Adverse Event Reporting System (FAERS) database show that DORAs have significantly lower rates of abuse-related adverse events (2.6%) compared to benzodiazepines (27.7%), Z-drugs (15.3%), and even non-scheduled medications like trazodone (22.7%) [136]. This low abuse liability, coupled with minimal cognitive impairment, makes DORAs particularly suitable for elderly patients, those with polypharmacy concerns, and individuals with history of substance use disorders [136] [132] [63]. The most common adverse effects are dose-dependent somnolence, nasopharyngitis, and headache, which are typically mild to moderate in severity [135] [132].
Special Population Considerations
In patients with Alzheimer's dementia, who often exhibit elevated cerebrospinal fluid orexin levels linked to cognitive decline and sleep efficiency reduction, suvorexant has demonstrated significant improvements in total sleep time without worsening cognitive impairment [70] [132]. Early research also suggests that suvorexant and lemborexant may help prevent delirium by reducing sleep disruptions, with several randomized trials showing promising results [132]. DORAs are generally not recommended in patients with narcolepsy due to their mechanism of action, and caution is advised in severe hepatic impairment [132].
Experimental Methodology for DORA Research
In Vitro Receptor Binding and Functional Assays
The characterization of DORA activity begins with comprehensive in vitro assays to determine binding affinity and functional antagonism at OX1R and OX2R. Radioligand binding experiments utilizing [³H]-labeled orexin-A in cell membranes expressing human recombinant orexin receptors provide quantitative assessment of binding affinity (Kd) and inhibitory constants (Ki) for test compounds [63]. Functional activity is typically evaluated using calcium flux assays in cells expressing OX1R or OX2R coupled to Gq proteins, measuring the concentration-dependent inhibition of orexin-induced calcium release [63]. Additional mechanistic studies include assessment of β-arrestin recruitment and receptor internalization to characterize biased signaling properties [63]. These assays establish the foundational pharmacodynamic profile prior to advancing to in vivo models.
In Vivo Sleep-Wake Assessment in Preclinical Models
Electroencephalogram (EEG) and electromyogram (EMG) telemetry in freely-moving rodents represents the gold standard for evaluating the effects of DORAs on sleep-wake architecture [63]. Animals are surgically implanted with wireless transmitters for continuous recording of cortical EEG and neck muscle EMG signals, allowing precise quantification of vigilance states (wakefulness, non-REM sleep, REM sleep) following compound administration [63]. Key parameters include sleep latency (time to persistent sleep), bout duration and number, time in each sleep stage, and spectral power within frequency bands [63]. Comparative studies typically include both active DORAs and reference compounds (e.g., zolpidem) to establish mechanistic differentiation [63].
Clinical Trial Design and Endpoint Assessment
Phase 2/3 clinical trials for DORAs employ polysomnography (PSG) and patient-reported outcomes as primary endpoints, typically conducted in specialized sleep laboratories under controlled conditions [135] [132]. Key PSG measures include latency to persistent sleep (LPS), wake after sleep onset (WASO), and total sleep time (TST) [135] [132]. Subjective measures include the Insomnia Severity Index (ISI) and sleep diaries capturing sleep onset, maintenance, and quality [63]. Next-day residual effects are assessed using digit symbol substitution tests, balance testing, and sleepiness scales [135] [132]. Long-term safety studies typically extend to 6-12 months, with particular attention to complex sleep behaviors, depression emergence, and tolerance development [135] [132].
Research Tools and Reagent Solutions
Table 3: Essential Research Reagents for Orexin System Investigation
| Reagent/Cell Line | Specific Application | Research Utility | Example Findings |
|---|---|---|---|
| CHO-K1/OX1R & OX2R cells | Receptor binding & signaling assays | Quantification of binding affinity (Kd, Ki) and functional potency (IC50) | Differential receptor occupancy requirements for sleep promotion [63] |
| Orexin-A & Orexin-B peptides | Receptor activation controls | Calcium flux, β-arrestin recruitment, and electrophysiological studies | Receptor-specific signaling bias and internalization kinetics [63] |
| Orexin promoter-reporter constructs | Transcriptional regulation studies | Identification of orexin expression modulators and circadian influences | Diurnal fluctuation in orexin production and release [63] |
| Selective OX1R (SORA-1) and OX2R (SORA-2) antagonists | Receptor subtype contribution analysis | Dissection of OX1R vs. OX2R roles in sleep-wake regulation | OX2R primarily mediates sleep-wake transitions [63] |
| Orexin neuron-specific transgenic models (orexin-Cre) | Circuit mapping and manipulation | Optogenetic/chemogenetic control of orexin neuronal activity | Orexin neuron stimulation promotes wakefulness; inhibition facilitates sleep [63] |
Signaling Pathway Visualization
Emerging Research Directions and Therapeutic Extensions
Potential Applications in Neurodegenerative Disorders
Growing evidence links sleep disruption with Alzheimer's disease (AD) pathophysiology through multiple mechanisms, including impaired clearance of tau and amyloid-β (Aβ) aggregates via the glymphatic system [70]. DORAs represent a promising therapeutic approach to mitigate AD progression by enhancing sleep-dependent clearance processes [70]. Preclinical models demonstrate that suvorexant improves sleep and reduces Aβ pathology, while clinical studies show it is well-tolerated in AD patients without worsening cognitive impairment [70] [132]. The bidirectional relationship between sleep and AD pathology creates a vicious cycle where sleep disturbances exacerbate Aβ accumulation, which in turn further disrupts sleep—a cycle that DORAs may help interrupt [70].
Investigations for Substance Use Disorders
Recent research has explored DORA applications in opioid use disorder (OUD), where the orexin system is hyperactive during withdrawal and contributes to drug craving and relapse [130]. Clinical studies demonstrate that suvorexant, when combined with buprenorphine/naloxone taper, improves sleep and reduces drug craving during initial abstinence [130]. The orexin system's role in "motivational activation" explains how drug-associated stimuli trigger orexin-mediated arousal states that drive drug-seeking behavior [130]. DORAs may disrupt this maladaptive learning by attenuating the hyperarousal associated with withdrawal and cue-induced craving [130].
Novel Compound Development
The development of vornorexant, with its notably short half-life comparable to zolpidem, represents a significant advancement in expanding the clinical utility of DORAs [69] [133]. This pharmacokinetic profile enables more targeted treatment of sleep initiation difficulties while minimizing next-day residual effects [69] [133] [134]. Ongoing research focuses on optimizing receptor occupancy kinetics and exploring selective orexin receptor antagonists (SORAs) that preferentially target OX2R, which appears to play a predominant role in sleep-wake regulation [63]. These refined therapeutic tools will enable more precise dissection of orexin receptor functions and potentially expand the therapeutic applications beyond insomnia.
Dual orexin receptor antagonists represent a paradigm shift in insomnia treatment, moving from non-selective neuronal suppression to targeted modulation of the wake-promoting system that is overactive in chronic insomnia. Their distinct mechanism of action preserves natural sleep architecture while minimizing the cognitive impairments, abuse potential, and safety concerns associated with traditional GABAergic hypnotics. The expanding pharmacokinetic diversity within the DORA class, exemplified by the development of short-acting agents like vornorexant, enables more precise matching of therapeutic properties to individual sleep complaints. Beyond primary insomnia, emerging research suggests potential applications in neurodegenerative disorders and substance use disorders where sleep disruption and orexin system dysregulation contribute to disease pathophysiology. As research continues to elucidate the complex neurobiology of the orexin system, DORAs stand as a compelling example of how targeted neuropharmacology can yield therapeutics with improved efficacy and safety profiles.
Adenosine is a primordial biological signaling molecule and an endogenous sleep-regulatory substance that operates through four G protein-coupled receptors: A1, A2A, A2B, and A3 [108] [9]. For decades, the most recognized manipulation of the adenosinergic system has been through caffeine, a non-selective adenosine receptor antagonist that promotes wakefulness by blocking A1 and A2A receptors [108] [137]. However, recent scientific advances have revealed that adenosine's therapeutic potential extends far beyond this antagonistic relationship, offering novel opportunities for treating sleep disorders, cancer, depression, and circadian rhythm disruptions [13] [138] [139]. The adenosine pathway represents a sophisticated regulatory system where extracellular adenosine concentrations are tightly controlled by enzymes including adenosine kinase (AdK) and adenosine deaminase (ADA), as well as equilibrative nucleoside transporters (ENTs) [108]. This complex regulatory network enables adenosine to function as a key homeostatic accumulator, with levels increasing during prolonged wakefulness and decreasing during sleep [108] [9]. The evolving understanding of receptor-specific effects, allosteric modulators, and pathway interactions now positions adenosine-based therapeutics as a promising frontier for developing treatments with potentially fewer side effects and novel mechanisms of action compared to conventional approaches.
Adenosine Receptor Subtypes and Their Therapeutic Targeting
Receptor Distribution, Signaling, and Selective Targeting
The four adenosine receptor subtypes exhibit distinct distributions throughout the body and couple to different intracellular signaling pathways, enabling highly specific physiological effects [108] [140] [9]. The A1 and A3 receptors primarily couple to Gi proteins, inhibiting adenylate cyclase and reducing intracellular cAMP levels, while A2A and A2B receptors couple to Gs proteins, stimulating adenylate cyclase and increasing cAMP production [138] [140]. Beyond these canonical pathways, adenosine receptors can also activate phospholipase C and modulate MAPK/ERK signaling, creating complex signaling networks that vary by cell type and context [140] [139].
Table 1: Adenosine Receptor Subtypes, Distribution, and Therapeutic Applications
| Receptor Subtype | G-protein Coupling | Key Tissue Distribution | Therapeutic Applications | Representative Ligands |
|---|---|---|---|---|
| A1 | Gi/o inhibitor | Brain (cortex, hippocampus, cerebellum), heart, kidneys | Sleep enhancement, cerebroprotection, anti-arrhythmic, antinociception | CPA (agonist), AR-001 (agonist), DPCPX (antagonist) |
| A2A | Gs stimulator | Brain (striatum, nucleus accumbens), immune cells, blood vessels | Cancer immunotherapy, Parkinson's disease, inflammation | CGS21680 (agonist), istradefylline (antagonist) |
| A2B | Gs stimulator | Lung, bladder, immune cells, fibroblasts | Inflammation, diabetes, cancer | N/A (less selectively targeted) |
| A3 | Gi/o inhibitor | Brain (low levels), testes, lung, kidneys, immune cells | Cancer, inflammation, neuroprotection | IB-MECA (agonist), CI-IB-MECA (agonist) |
The pursuit of receptor-selective compounds has become a major focus in adenosine pharmacology. For A1 receptors, which are highly expressed in the brain, heart, and kidneys, agonists show promise for sleep disorders and cerebroprotection, while antagonists may benefit renal function and cognitive enhancement [140] [58] [9]. The recently identified compound AR-001 exemplifies this targeted approach, demonstrating hypnotic effects through selective A1 receptor agonism [58]. A2A receptors, concentrated in the striatum and immune cells, represent valuable targets for cancer immunotherapy and Parkinson's disease [138]. A3 receptors, with their unique distribution and xanthine-insensitive properties, offer opportunities for cancer treatment and neuroprotection, though their lower abundance in the brain presents challenges for central nervous system applications [140].
Quantitative Data on Receptor Binding Affinities
The development of selective adenosine receptor ligands requires careful optimization of binding affinities to minimize off-target effects. Recent research has yielded compounds with significantly improved selectivity profiles.
Table 2: Binding Affinities (Ki, μM) of Selected Adenosine Receptor Ligands
| Compound | A1 Receptor | A2A Receptor | A3 Receptor | Primary Receptor Selectivity |
|---|---|---|---|---|
| CPA | 0.00059 | 0.462 | 0.24 | A1 agonist |
| CGS21680 | 2.6 | 0.015 | 0.584 | A2A agonist |
| CI-IB-MECA | 0.82 | 0.47 | 0.00033 | A3 agonist |
| IB-MECA | 0.054 | 0.056 | 0.0011 | A3 agonist |
| Caffeine | ~10 | ~10 | ~100 | Non-selective antagonist |
The data reveal remarkable progress in developing subtype-selective agonists, particularly for the A3 receptor where CI-IB-MECA demonstrates approximately 2,500-fold selectivity over A1 receptors and 1,400-fold selectivity over A2A receptors [140]. This selectivity is crucial for minimizing side effects that have plagued earlier adenosine-based therapeutics, such as the arteriopathy, nausea, and cardiovascular effects observed with earlier generation compounds [140].
Experimental Approaches for Evaluating Adenosine-Based Therapeutics
In Vivo Sleep Studies Using Pentobarbital-Induced Models
The pentobarbital-induced sleep model in mice provides a well-established methodology for evaluating the sleep-enhancing potential of adenosine receptor agonists [13] [58]. This approach measures two primary endpoints: sleep latency (time to sleep onset) and sleep duration (total sleep time), offering quantifiable metrics for comparing therapeutic efficacy.
Detailed Protocol:
- Animals and Housing: Utilize ICR mice (18-20 g) housed under controlled conditions (23 ± 2°C, 50% ± 5% relative humidity, 12-hour light/dark cycle) with ad libitum access to food and water [13].
- Test Substance Administration: Orally administer the investigational compound (e.g., PUTRE at 200-400 mg/kg/day) or positive control (diazepam at 2 mg/kg/day) once daily for two weeks [13].
- Pentobarbital Challenge: Forty-five minutes after the final administration, intraperitoneally inject pentobarbital (45 mg/kg) to induce sleep [13].
- Sleep Parameter Measurement:
- Sleep Latency: Measure the time from pentobarbital injection to loss of the righting reflex.
- Sleep Duration: Record the time between loss and recovery of the righting reflex.
- Exclusion Criteria: Exclude animals maintaining the righting reflex for >15 minutes post-injection [13].
- Receptor Mechanism Validation: To confirm adenosine receptor involvement, co-administer selective antagonists (e.g., DPCPX for A1 receptors at 5 mg/kg/day or caffeine at 10 mg/kg/day) with the test compound [13].
This methodology successfully demonstrated the sleep-enhancing effects of Phlomoides umbrosa Turczaninow root extract (PUTRE), which significantly decreased sleep latency and increased sleep duration in a manner blocked by A1 receptor antagonism [13]. Similar approaches have validated the hypnotic effects of novel compounds like AR-001 through A1 receptor mechanisms [58].
In Vitro Signaling Pathway Analysis
Elucidating the intracellular signaling pathways activated by adenosine receptors requires sophisticated in vitro approaches that can dissect complex molecular interactions.
Detailed Protocol:
- Cell Culture: Utilize appropriate cell lines (e.g., U2OS cells for circadian studies, CHO cells transfected with specific adenosine receptors) maintained under standard conditions [139].
- Receptor Expression Profiling: Quantify receptor mRNA and protein levels using RT-PCR, Western blotting, or immunocytochemistry to establish baseline expression patterns [139].
- Pathway Activation Assays:
- Transcription Factor Screening: Employ barcoded reporter luciferase libraries (e.g., BC-STARPROM) to identify DNA response elements activated by adenosine receptor modulation [139].
- Calcium Imaging: Monitor intracellular Ca2+ fluxes using fluorescent indicators (e.g., Fura-2) following receptor stimulation [139].
- Phosphoprotein Analysis: Detect phosphorylated signaling intermediates (e.g., ERK1/2) via Western blotting or phospho-specific immunoassays [139].
- Gene Silencing Approaches: Use siRNA-mediated knockdown of specific pathway components (e.g., FOS, JUN, ERK1) to confirm their necessity in adenosine-mediated effects [139].
- Circadian Rhythm Assessment: In cells expressing PER2::Luc reporters, monitor bioluminescence rhythms to evaluate the impact of adenosine receptor modulators on circadian period length [139].
These methods revealed that adenosine receptor antagonists activate a Ca2+-ERK-AP-1 pathway that regulates PER2 expression and circadian timing, independent of the traditional cAMP-CREB pathway activated by adenosine receptor agonists [139]. This sophisticated signaling mechanism explains how adenosine integrates both sleep history and light exposure to modulate circadian entrainment.
Signaling Pathways and Molecular Mechanisms
Adenosine receptor signaling engages multiple interconnected pathways that regulate diverse physiological processes from sleep to circadian entrainment. The following diagram illustrates the key molecular pathways through which adenosine and its receptors modulate sleep regulation and circadian timing:
Diagram Title: Adenosine Signaling in Sleep and Circadian Regulation
This integrated signaling network demonstrates how adenosine coordinates both sleep homeostasis through the A1 receptor in sleep/wake regulatory regions like the VLPO and basal forebrain [9], and circadian timing through transcriptional regulation of clock genes in SCN neurons [139]. The recently discovered Ca2+-ERK-AP-1 pathway activated by adenosine receptor antagonists provides a molecular explanation for how caffeine and other antagonists can modulate circadian rhythms independently of their effects on sleep pressure [139]. This complex interplay enables adenosine to function as a key integrator of sleep history and light signaling, fine-tuning circadian phase according to sleep-wake state.
The Scientist's Toolkit: Essential Research Reagents
Advancing adenosine-based therapeutics requires specialized research tools for probing receptor function, measuring adenosine dynamics, and evaluating physiological responses.
Table 3: Essential Research Reagents for Adenosine Pathway Investigation
| Reagent/Category | Specific Examples | Research Applications | Key Functions |
|---|---|---|---|
| Selective Agonists | CPA (A1), CGS21680 (A2A), IB-MECA (A3), AR-001 (A1) | Receptor-specific activation, sleep studies, circadian research | Mimic endogenous adenosine effects at specific receptor subtypes |
| Selective Antagonists | DPCPX (A1), SCH58261 (A2A), MRS1754 (A2B), MRS1523 (A3) | Receptor mechanism validation, blockade of endogenous signaling | Inhibit receptor activation to determine pathway specificity |
| Genetic Tools | Adora1-/-, Adora2a-/- knockout mice, siRNA/shRNA knockdown systems | Pathway necessity studies, receptor-specific function analysis | Enable genetic dissection of receptor contributions to physiological effects |
| Adenosine Sensors | GRABAdo1.0 (genetically encoded sensor), mutant controls | Real-time adenosine monitoring in vivo and in vitro | Detect spatial and temporal dynamics of adenosine release with high resolution |
| Behavioral Models | Pentobarbital-induced sleep, chronic restraint stress, forced swim test | Sleep efficacy assessment, antidepressant activity evaluation | Provide quantitative metrics of therapeutic effects in whole organisms |
| Circadian Reporters | PER2::Luc cell lines, PER2::Luc SCN slices | Circadian rhythm monitoring, period length assessment | Enable real-time tracking of circadian oscillations in response to interventions |
These research tools have been instrumental in recent discoveries, such as using GRABAdo1.0 sensors to demonstrate ketamine-induced adenosine surges in the medial prefrontal cortex [141] and employing PER2::Luc reporters to identify adenosine's role in circadian entrainment [139]. The combination of selective pharmacological agents with genetic approaches and advanced sensors provides a comprehensive toolkit for deconstructing the complex roles of adenosine in health and disease.
Emerging Therapeutic Applications and Clinical Perspectives
Sleep and Circadian Rhythm Disorders
Beyond traditional sedative-hypnotics, adenosine-based approaches offer potentially more physiological solutions for sleep disorders. Natural extracts like Phlomoides umbrosa Turczaninow root extract (PUTRE) demonstrate sleep-enhancing effects through A1 receptor-specific agonist activity, reducing sleep latency and increasing sleep duration in preclinical models [13]. Similarly, the synthetic compound AR-001 promotes sleep by activating A1 receptors in the ventrolateral preoptic nucleus (VLPO) while suppressing activity in wake-promoting regions like the basal forebrain and lateral hypothalamus [58]. Perhaps most intriguingly, adenosine signaling integrates with the circadian system, with A1/A2A receptor antagonists like caffeine modulating light-induced phase shifts and directly regulating PER1 and PER2 expression in the suprachiasmatic nucleus [139]. This intersection between sleep homeostasis and circadian regulation positions adenosine therapeutics as promising candidates for shift work disorder, jet lag, and other circadian sleep-wake rhythm disorders.
Cancer Immunotherapy
The adenosine pathway represents an emerging frontier in cancer immunotherapy, particularly through A2A receptor antagonism [138] [142]. Tumors often create immunosuppressive microenvironments by accumulating extracellular adenosine, which activates A2A receptors on natural killer cells and T cells to suppress their cytotoxic functions [138]. This mechanism also promotes the formation of immunosuppressive cell types like myeloid-derived suppressor cells and regulatory T cells [138]. Targeted approaches including small-molecule A2A receptor antagonists and monoclonal antibodies against the CD39/CD73/A2A receptor axis are currently in clinical development, both as monotherapies and in combination with anti-PD-1/PD-L1 immunotherapies [138] [142]. The clinical trial landscape for adenosine-targeted therapies has expanded significantly since 2011, with numerous programs directed at A2A receptors, CD73, and CD39 [142].
Rapid-Acting Antidepressants
Recent groundbreaking research has identified adenosine signaling as central to the mechanisms of rapid-acting antidepressants including ketamine and electroconvulsive therapy [141]. Genetically encoded adenosine sensor experiments reveal that both interventions induce strong adenosine surges in mood-regulatory regions like the medial prefrontal cortex and hippocampus [141]. Genetic or pharmacological disruption of A1 and A2A receptors abolishes the therapeutic effects of these treatments, establishing the essential role of adenosine signaling in their antidepressant efficacy [141]. Leveraging this mechanism, researchers have developed ketamine derivatives that enhance adenosine signaling with improved antidepressant efficacy and reduced side effects [141]. Furthermore, acute intermittent hypoxia, a non-pharmacological intervention involving controlled oxygen reduction, increases brain adenosine levels and produces similar antidepressant effects, offering a potentially scalable, noninvasive therapeutic approach for major depressive disorder [141].
The landscape of adenosine-based therapeutics has expanded dramatically beyond the familiar caffeine antagonism to encompass receptor-specific agonists, allosteric modulators, and pathway-targeted interventions. The sophisticated understanding of adenosine's dual role in sleep homeostasis and circadian entrainment, its immunomodulatory functions in the tumor microenvironment, and its newly discovered centrality in rapid-acting antidepressant mechanisms reveals a signaling system of remarkable versatility and therapeutic potential. As research continues to elucidate the complex signaling networks downstream of adenosine receptors and develop increasingly selective pharmacological tools, the clinical applications of adenosine-based therapeutics will continue to grow. The ongoing clinical trials targeting the adenosine pathway in cancer, the development of natural and synthetic A1 receptor agonists for sleep disorders, and the exploration of adenosine-mediated antidepressants represent just the beginning of harnessing this ancient biological signaling system for modern therapeutic challenges.
Within the expanding field of sleep biochemistry, the investigation of natural compounds and repurposed nutritional agents represents a promising frontier for developing novel therapeutic interventions. Sleep disorders, which affect a significant portion of the global population, are increasingly recognized as complex biochemical disturbances affecting neural circuitry, neurotransmitter balance, and circadian regulation [99]. Within this context, L-theanine and magnesium have emerged as two compounds with particular mechanistic interest and clinical potential. This whitepaper provides a comprehensive technical analysis of these agents, focusing on their synergistic mechanisms of action, clinical evidence profiles, and methodological considerations for preclinical research. The overarching thesis posits that targeted nutritional supplementation addressing multiple sleep-regulatory pathways simultaneously may offer enhanced efficacy over single-target approaches, potentially bridging gaps in current pharmacotherapeutic strategies for sleep disorders.
Biochemical Mechanisms of Action
Neuropharmacological Targets of L-Theanine
L-theanine (γ-glutamylethylamide), a non-proteinogenic amino acid predominantly found in Camellia sinensis, exerts its sleep-promoting effects through multiple neuropharmacological mechanisms. Structurally analogous to glutamate, L-theanine acts as a competitive antagonist at glutamatergic receptors, including the ionotropic AMPA and NMDA receptors, thereby reducing excitatory neurotransmission [143]. Simultaneously, L-theanine facilitates GABAergic inhibition by increasing GABA concentrations in the brain and potentially modulating GABA-A receptor function [143] [144]. This dual action creates a neurochemical environment conducive to sleep initiation and maintenance.
Research indicates that L-theanine administration increases alpha brain wave activity, which is associated with a state of wakeful relaxation that often precedes sleep onset [144]. Additionally, L-theanine influences monoaminergic systems, with studies reporting increased levels of dopamine and serotonin in specific brain regions following administration [143] [145]. These neurotransmitters play complex roles in sleep-wake regulation, particularly in the modulation of sleep architecture and mood states that influence sleep quality.
Magnesium's Role in Sleep Regulation
Magnesium, an essential mineral cofactor in over 300 enzymatic reactions, regulates sleep through both direct and indirect mechanisms. As a natural NMDA receptor antagonist, magnesium modulates glutamatergic excitability and calcium influx into neurons [99] [143]. Magnesium also activates GABA receptors, enhancing the activity of this primary inhibitory neurotransmitter system [99].
Beyond neurotransmitter regulation, magnesium influences circadian processes by supporting melatonin synthesis and regulating cellular biological clocks [99] [106]. Magnesium deficiency has been demonstrated to reduce plasma melatonin concentrations, disrupting normal sleep-wake cycles [99]. Furthermore, magnesium plays a role in maintaining cellular energy balance and mitochondrial function, both of which are crucial for sleep homeostasis [99]. The mineral also helps regulate the hypothalamic-pituitary-adrenal axis, with supplementation shown to reduce cortisol levels, particularly in individuals with magnesium deficiency [146].
Synergistic Mechanisms of Combined Administration
Emerging research suggests that magnesium and L-theanine may exhibit synergistic effects when administered in combination. Novel Mg-L-theanine complexes have demonstrated enhanced efficacy compared to either compound alone in preclinical models [143] [145]. These complexes appear to boost slow-wave brain activity, increase GABA receptor expression, and elevate key neurotransmitters including serotonin and melatonin more effectively than isolated components [143].
The molecular synergy likely stems from complementary mechanisms: while both compounds modulate glutamatergic and GABAergic systems, they target different aspects of these pathways. Magnesium serves as an ionic modulator of receptor function, while L-theanine acts as a direct receptor ligand and neurotransmitter precursor. This multi-target approach aligns with contemporary understanding of sleep as a complex physiological process requiring coordinated modulation of multiple systems.
Table 1: Key Neurochemical Effects of L-Theanine and Magnesium
| Neurochemical Parameter | L-Theanine Effects | Magnesium Effects | Synergistic Outcomes |
|---|---|---|---|
| GABAergic Activity | Increases GABA concentration; modulates GABA-A receptors [143] | Activates GABA receptors through allosteric modulation [99] | Enhanced receptor expression and neurotransmitter levels [143] |
| Glutamatergic Activity | Competitive NMDA/AMPA receptor antagonism [143] | Non-competitive NMDA receptor blockade; reduces calcium influx [99] | Additive inhibition of excitatory neurotransmission |
| Monoaminergic Systems | Increases dopamine, serotonin levels [143] [145] | Limited direct effect; modulates receptor sensitivity | Potentiated serotonin increase; improved mood-sleep interface |
| Brain Wave Patterns | Increases alpha wave activity [144] | Promotes delta wave synchronization [146] | Enhanced slow-wave power and sleep depth [143] |
| Circadian Regulation | Limited direct effect | Supports melatonin synthesis; regulates cellular clocks [99] | Improved sleep architecture and timing |
Clinical Evidence and Efficacy Profiles
Human Clinical Trials and Meta-Analyses
Recent systematic reviews and meta-analyses provide compelling evidence for the efficacy of both L-theanine and magnesium in improving sleep parameters. A 2024 meta-analysis of 18 randomized controlled trials (N=897 participants) found that L-theanine significantly improved subjective sleep onset latency (SMD=0.15, 95% CI [0.01, 0.29], p=0.04), subjective daytime dysfunction (SMD=0.33, 95% CI [0.16, 0.49], p<0.001), and overall subjective sleep quality scores (SMD=0.43, 95% CI [0.04, 0.83], p=0.03) [147].
Magnesium supplementation has demonstrated particular efficacy in specific populations. In older adults with insomnia, magnesium supplementation significantly reduced sleep onset latency and improved sleep efficiency [106] [146]. A 2021 systematic review and meta-analysis focusing on older adults found that magnesium supplementation reduced the time required to fall asleep, with researchers suggesting that divided daily dosing (up to three times daily) may optimize outcomes for insomnia symptoms [146].
Population Studies and Cross-Sectional Evidence
Large-scale epidemiological studies provide additional insights into the relationship between these compounds and sleep health. A cross-sectional study of 20,585 participants from NHANES data found a graded dose-response relationship between magnesium deficiency score (MDS) and sleep troubles, as well as sleep disorders [148]. Specifically, a significant positive association was observed between MDS and sleep apnea (OR=3.01, 95% CI [1.37, 6.62]), though no association was found with restless legs syndrome or insomnia [148].
The CARDIA study, a longitudinal analysis of 3,964 participants, demonstrated that higher magnesium intake was associated with reduced likelihood of short sleep (<7 hours) (Q4 vs. Q1: OR=0.64, 95% CI [0.51, 0.81], ptrend=0.012) and borderline association with better sleep quality (Q4 vs. Q1: OR=1.23, 95% CI [0.999, 1.50], ptrend=0.051) [106]. This association persisted among participants without depressive disorders but not among those with depressive disorders, suggesting potential effect modification by mental health status.
Specific Sleep Parameter Improvements
Table 2: Clinically Relevant Sleep Parameter Improvements from Supplementation
| Sleep Parameter | L-Theanine Efficacy | Magnesium Efficacy | Clinical Implications |
|---|---|---|---|
| Sleep Onset Latency | Significant improvement in meta-analysis (SMD=0.15) [147] | Reduced in older adults with insomnia [146] | Both agents address difficulty falling asleep |
| Sleep Quality Scores | Significant improvement in overall quality (SMD=0.43) [147] | Borderline association in longitudinal study [106] | Subjective sleep perception improved |
| Wake After Sleep Onset | Reduced in combination with caffeine [149] | Not specifically reported | L-theanine may protect against sleep fragmentation |
| Sleep Duration | Not primarily affected | Reduced risk of short sleep (<7h) (OR=0.64) [106] | Magnesium may help achieve sufficient sleep quantity |
| Daytime Functioning | Significant improvement (SMD=0.33) [147] | Improved in deficient populations [99] | Next-day functional benefits |
Experimental Models and Methodologies
Preclinical Sleep Assessment Models
Animal models remain essential for elucidating the mechanistic underpinnings of sleep-regulating compounds. The caffeine-induced sleep disturbance model represents a validated approach for evaluating potential therapeutic agents. In this model, animals receive caffeine (typically 7.5 mg/kg intraperitoneally) to induce wakefulness, followed by test compounds to assess their ability to counteract this effect [143] [145]. Electrocorticography (ECoG) recordings then quantify changes in brain electrical activity, including spike frequency, amplitude, and power spectral analysis across different frequency bands.
The pentobarbital-induced sleep test provides another established methodology for preliminary screening of sedative-hypnotic properties. This model evaluates sleep latency (time to sleep onset) and sleep duration following subhypnotic doses of pentobarbital (typically 30 mg/kg) [143]. Test compounds that reduce sleep latency or prolong sleep duration are considered to possess sleep-promoting properties. This model has demonstrated that Mg-L-theanine complexes significantly improve both parameters even after caffeine administration, suggesting potent effects [143].
Human Sleep Study Methodologies
Human studies employ various assessment tools to evaluate sleep quality and architecture. Polysomnography (PSG) represents the gold standard, providing objective measures of sleep architecture, including sleep stages, wake after sleep onset (WASO), and sleep efficiency [149]. Recent studies have incorporated additional measures such as cerebral blood flow monitoring via near-infrared spectroscopy (NIRS) to investigate hemodynamic changes associated with sleep [149].
Subjective assessment tools include standardized questionnaires such as the Pittsburgh Sleep Quality Index (PSQI), Insomnia Severity Index (ISI), and Epworth Sleepiness Scale (ESS) [99]. These instruments provide valuable patient-reported outcomes that complement objective measures. For example, a study examining theanine's ability to counteract caffeine-induced sleep disturbances used electroencephalography to measure sleep stages and demonstrated that theanine (50 mg) suppressed caffeine-induced increases in WASO time [149].
Diagram 1: Neurochemical Pathways in Sleep Regulation. This diagram illustrates the antagonistic relationship between caffeine-induced wakefulness and the sleep-promoting effects of L-theanine and magnesium through their actions on multiple neurotransmitter systems and sleep-regulatory pathways.
Research Reagent Solutions and Technical Tools
Essential Research Materials
Table 3: Key Reagents and Experimental Tools for Sleep Research
| Reagent/Tool | Specifications & Functions | Research Applications |
|---|---|---|
| L-Theanine | High-purity (>98%) Suntheanine; water-soluble; doses 50-200mg in human studies [149] | Human clinical trials; biochemical assays; neurotransmitter studies |
| Mg-L-Theanine Complexes | Novel compounds with 8% or 18% Mg²+ content; enhanced blood-brain barrier permeability [143] [145] | Preclinical models; mechanistic studies; synergy investigations |
| Caffeine | 7.5 mg/kg (i.p.) for rodent models; 30-50mg for human challenges [143] [149] | Induction of sleep disturbances; antagonism studies |
| Pentobarbital | 30 mg/kg (i.p.) for sleep tests in rodents [143] | Screening for sedative-hypnotic properties |
| Electrocorticography (ECoG) | PowerLab systems with 1000Hz sampling; band-pass filter 0.5-500Hz [143] | Brain electrical activity monitoring; power spectral analysis |
| Western Blot Assays | GABA-A receptor subunits, serotonergic receptors [143] [145] | Protein expression quantification in brain tissue |
| ELISA Kits | Dopamine, serotonin, melatonin, cortisol quantification [143] | Neuroendocrine and neurotransmitter measurements |
Diagram 2: Experimental Workflow in Sleep Research. This diagram outlines the integrated methodological approaches used in preclinical and clinical sleep research, from model systems to analytical techniques and data integration.
Discussion and Research Implications
The accumulating evidence for L-theanine and magnesium as sleep-modulating agents presents several compelling implications for both basic research and clinical applications. From a mechanistic perspective, these compounds exemplify the potential of multi-target approaches to sleep regulation, contrasting with single-receptor targeting characteristic of many pharmaceutical hypnotics. The synergistic effects observed with Mg-L-theanine complexes suggest that strategic combination of compounds with complementary mechanisms may yield enhanced efficacy while potentially minimizing side effects through lower required doses of individual components [143] [145].
From a translational perspective, the favorable safety profiles of both L-theanine and magnesium support their consideration as potential alternatives or adjuncts to conventional sleep medications, particularly for mild-to-moderate sleep disturbances. The FDA classifies L-theanine as "generally recognized as safe" at doses up to 250 mg per serving [144], while magnesium supplements are generally well-tolerated, with gastrointestinal symptoms being the most commonly reported adverse effect at higher doses [150]. This safety profile is particularly relevant given concerns regarding dependency and cognitive side effects associated with benzodiazepines and other sedative-hypnotics [143].
Future research directions should include more comprehensive dose-response studies to establish optimal dosing regimens for both individual and combined administration. Additionally, research exploring the effects of these compounds on specific sleep disorders beyond primary insomnia—such as sleep disturbances associated with neurological conditions, shift work, or aging—would expand our understanding of their therapeutic potential. The development of more bioavailable forms, such as magnesium L-threonate and Mg-L-theanine complexes, represents another promising avenue for enhancing efficacy [143] [146].
In conclusion, L-theanine and magnesium exemplify the promising intersection of nutritional science and sleep medicine. Their diverse mechanisms of action, supportive clinical evidence, and favorable safety profiles position them as valuable agents in the growing arsenal of evidence-based approaches to sleep optimization. Further research elucidating their precise molecular targets and synergistic potential will continue to advance both our fundamental understanding of sleep biochemistry and our ability to address sleep disturbances effectively.
Conclusion
The biochemistry of sleep is a complex, multi-system process orchestrated by a precise interplay of neurotransmitters, hormones, and intracellular signaling pathways. Disruptions in these systems—from GABAergic inhibition and adenosinergic homeostasis to melatonin signaling and orexinergic tone—underpin the pathophysiology of diverse sleep disorders. The future of sleep medicine lies in leveraging this mechanistic understanding to develop highly targeted, personalized therapies. Promising directions include exploiting the adenosine system for novel hypnotics, harnessing the metabolic benefits of melatonin, targeting orexin for narcolepsy and insomnia, and investigating the therapeutic potential of minerals like magnesium. Advancing molecular diagnostics and embracing circadian principles in drug delivery will be paramount for the next generation of sleep pharmacotherapies, ultimately aiming to restore not just sleep, but overall metabolic and neurological health.
References
- 1. Influence of glutamatergic and GABAergic ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2023.1213971/full]
- 2. GABA and its receptors' mechanisms in the treatment of ... [https://www.sciencedirect.com/science/article/pii/S2405844024166960]
- 3. Noradrenaline and acetylcholine shape functional connectivity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12585100/]
- 4. The Neurobiological Basis of Sleep and Sleep Disorders - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6230548/]
- 5. Sleep Disorders: Pathogenesis and Therapeutic Interventions [https://pmc.ncbi.nlm.nih.gov/articles/PMC11892028/]
- 6. GABA mechanisms and sleep [https://pubmed.ncbi.nlm.nih.gov/11983310/]
- 7. Elevated peripheral glutamate and upregulated expression ... [https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2024.1436024/full]
- 8. Metabotropic glutamate receptor function and regulation of ... [https://www.sciencedirect.com/science/article/abs/pii/S0074774222001416]
- 9. Functions and mechanisms of adenosine and its receptors ... [https://www.sciencedirect.com/science/article/pii/S1389945724000534]
- 10. Adenosine and P1 receptors: Key targets in the regulation ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2023.1098976/full]
- 11. Role of adenosine in the pathophysiology and treatment of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12454791/]
- 12. Special Issue “Molecular Research on Adenosine Receptors [https://www.mdpi.com/1422-0067/26/23/11298]
- 13. Improvement of sleep disorders through the adenosine A ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11904018/]
- 14. Integrative sleep management: from molecular pathways to ... [https://link.springer.com/article/10.1007/s00210-025-04183-y]
- 15. The monoaminergic system is a bilaterian... | Nature Communications [https://www.nature.com/articles/s41467-023-39030-2?error=cookies_not_supported&code=139c5166-22de-4601-a756-4da5cd387543]
- 16. New Players on the Monoaminergic Field: Relevance to the Mental... [https://www.frontiersin.org/research-topics/52442/new-players-on-the-monoaminergic-field-relevance-to-the-mental-disorder/articles]
- 17. A population-based observational study using statistical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12261605/]
- 18. The prevalence and risk factors of sleep disturbances ... [https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2025.1595303/full]
- 19. Monoamine transporters: structure, intrinsic dynamics and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6712983/]
- 20. Mapping neurotransmitter systems to the structural and ... [https://www.nature.com/articles/s41593-022-01186-3]
- 21. Brain Monoaminergic Neurons: Distribution and... | SpringerLink [https://link.springer.com/chapter/10.1007/978-1-4613-2315-0_2]
- 22. Certain Antidepressants Increase REM Sleep Without Atonia [https://consultqd.clevelandclinic.org/certain-antidepressants-increase-rem-sleep-without-atonia]
- 23. The regulatory role of AP-2β in monoaminergic ... [https://cellandbioscience.biomedcentral.com/articles/10.1186/s13578-022-00891-7]
- 24. Melatonin: An Overview on the Synthesis Processes and ... [https://www.mdpi.com/2077-0472/15/3/273]
- 25. Melatonin: beyond circadian regulation - exploring its ... [https://www.sciencedirect.com/science/article/abs/pii/S1087079225000760]
- 26. Melatonin biosynthesis and regulation in reproduction [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1630164/full]
- 27. Endocrine regulation of circadian rhythms [https://www.nature.com/articles/s44323-025-00024-6]
- 28. Circadian Rhythm Disruptions and Cardiovascular Disease ... [https://www.mdpi.com/1467-3045/47/8/664]
- 29. Molecular Links Between Circadian Rhythm Disruption ... [https://pubmed.ncbi.nlm.nih.gov/40363695/]
- 30. Melatonin-Mediated Circadian Rhythm Signaling Exhibits ... [https://www.mdpi.com/2218-273X/15/2/226]
- 31. A focus on melatonin, melanin-concentrating hormone and ... [https://pubmed.ncbi.nlm.nih.gov/40887101/]
- 32. The role of melatonin and circadian rhythms in ... [https://www.sciencedirect.com/science/article/pii/S1871402125000190]
- 33. Sleep disorders impact hormonal regulation - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12315459/]
- 34. Orexin A stimulates cortisol secretion from human ... [https://pubmed.ncbi.nlm.nih.gov/11158046/]
- 35. Leptin and ghrelin dynamics: unraveling their influence on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11196531/]
- 36. Ghrelin – Physiological Functions and Regulation - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5819073/]
- 37. Stress, motivation and the gut-brain axis - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC6252147/]
- 38. The relationship between the leptin/ghrelin ratio and meals ... [https://nutritionj.biomedcentral.com/articles/10.1186/s12937-018-0427-x]
- 39. The role of leptin and ghrelin in the regulation of appetite ... [https://www.sciencedirect.com/science/article/pii/S0196978125000282]
- 40. advances, limitations, and future directions for drug discovery [https://pubmed.ncbi.nlm.nih.gov/40583776/]
- 41. Genetic and Immunologic Aspects of Sleep and Sleep Disorders [https://pmc.ncbi.nlm.nih.gov/articles/PMC3734884/]
- 42. A Review on Genetics of Sleep Disorders - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3939950/]
- 43. Alterations in sleep-activity cycles and clock gene expression ... [https://translationalneurodegeneration.biomedcentral.com/articles/10.1186/s40035-025-00492-7]
- 44. Article NKCC1 inhibition improves sleep quality and EEG ... [https://www.sciencedirect.com/science/article/pii/S258900422500481X]
- 45. Sleep & Vigilance States for Drug Discovery [https://synapcell.com/sleep-vigilance-states/]
- 46. Post-sleep electroencephalographic slowing correlates ... [https://www.sciencedirect.com/science/article/abs/pii/S1389945725004289]
- 47. Association of Novel EEG Biomarkers of Sleep Depth and ... [https://pubmed.ncbi.nlm.nih.gov/39887059/]
- 48. Chronic sleep curtailment expediates brain aging by activating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11870557/]
- 49. Exploring common circulating diagnostic biomarkers for sleep ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12611825/]
- 50. Potential non-invasive biomarkers of chronic sleep ... [https://www.nature.com/articles/s41598-025-95403-1]
- 51. Analysis of the correlation between peripheral blood ... [https://www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2025.1538028/full]
- 52. Neuroimaging and fluid biomarkers in Parkinson's disease ... [https://www.nature.com/articles/s41467-024-49949-9]
- 53. Quantitative Comparison of Statistical Methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9227835/]
- 54. Release Details - Harmony Biosciences [https://ir.harmonybiosciences.com/news-releases/news-release-details/harmony-biosciences-presents-preclinical-data-demonstrating]
- 55. Wake-Promoting - an overview [https://www.sciencedirect.com/topics/chemistry/wake-promoting]
- 56. Quantitative high-throughput screening data analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC4375054/]
- 57. Establishment of a High Throughput Screening System for ... [https://pubmed.ncbi.nlm.nih.gov/35762541/]
- 58. Hypnotic effect of AR-001 through adenosine A1 receptor [https://www.sciencedirect.com/science/article/pii/S0014299924005363]
- 59. A Data Analysis Pipeline Accounting for Artifacts in Tox21 ... [https://www.sciencedirect.com/science/article/pii/S2472555222072525]
- 60. Comparative efficacy of new wake-promoting agents for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12613485/]
- 61. Application of machine learning for high-throughput tumor ... [https://pubmed.ncbi.nlm.nih.gov/38685558/]
- 62. Modeling sleep and neuropsychiatric disorders in zebrafish [https://www.sciencedirect.com/science/article/abs/pii/S0959438816302380]
- 63. Targeting the Orexin System in the Pharmacological ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12429101/]
- 64. What drugs are in development for Sleep Initiation and ... [https://synapse.patsnap.com/article/what-drugs-are-in-development-for-sleep-initiation-and-maintenance-disorders]
- 65. The importance of ligand gated ion channels in sleep and ... [https://www.sciencedirect.com/science/article/abs/pii/S0006295223001235]
- 66. Biochemical Mechanisms of Sleep Regulation [https://sciintl.scione.com/cms/fulltext.php?id=319]
- 67. Orexin-A inhibits γ-aminobutyric acid current of neonatal rat ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8214956/]
- 68. Molecular Insights into the Interaction of Orexin 1 Receptor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12250729/]
- 69. Dual orexin receptor antagonists in insomnia: Toward a ... [https://www.sciencedirect.com/science/article/pii/S0091305725001649]
- 70. Dual orexin receptor antagonists as promising therapeutics ... [https://www.nature.com/articles/s44323-025-00025-5]
- 71. Circadian rhythms revealed: unraveling the genetic ... [https://www.frontiersin.org/journals/sleep/articles/10.3389/frsle.2025.1544945/full]
- 72. A synthetic chronogenetic gene circuit for programmed ... [https://www.nature.com/articles/s41467-025-56584-5]
- 73. Nanomaterial-enabled drug delivery systems for circadian ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra04137f]
- 74. Multi-pulse chronotherapeutic approaches for circadian ... [https://pubmed.ncbi.nlm.nih.gov/40823912/]
- 75. Programmable chronogenetic gene circuits for self ... [https://www.sciencedirect.com/science/article/pii/S0168365925005796]
- 76. Molecular Mechanisms Of Chronotherapy In Orthodontic ... [https://www.sciencedirect.com/science/article/pii/S0020653925046386]
- 77. 2025 Chronobiology Conference GRC [https://www.grc.org/chronobiology-conference/2025/]
- 78. The biggest challenges clinical trials will face in 2025 [https://www.clinicaltrialsarena.com/features/clinical-trials-challenges-expect-2025/]
- 79. The Pathophysiology of Insomnia - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC4388122/]
- 80. Sleep disturbances in generalized anxiety Disorder [https://www.sciencedirect.com/science/article/abs/pii/S138994572500214X]
- 81. Pathophysiological Insights and Multimodal Interventions in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12474699/]
- 82. Neurobiological mechanisms of sleep state misperception ... [https://www.sciencedirect.com/science/article/abs/pii/S1087079225000498]
- 83. Bojungikgi-Tang Modulates the Hypothalamic–Pituitary– ... [https://www.mdpi.com/2227-9059/13/12/2846]
- 84. Identification of potential drug targets for insomnia by ... [https://www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2024.1380321/full]
- 85. Eisai Presents Clinical Study Results of Novel Orexin ... [https://www.eisai.com/news/2025/news202562.html]
- 86. Orexin/Hypocretin: A Neuropeptide at the Interface of Sleep ... [https://www.sciencedirect.com/science/article/abs/pii/S0031699724008330]
- 87. Neurobiology of the Orexin System and Its Potential Role in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8870430/]
- 88. Orexinergic System in Neurodegenerative Diseases - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8416170/]
- 89. Takeda Presents Orexin Data from Landmark Oveporexton ... [https://www.takeda.com/newsroom/newsreleases/2025/takeda-orexin-data-oveporexton-phase-3-narcolepsy-world-sleep-2025/]
- 90. possible therapeutic targets for psychiatric disorders [https://pubmed.ncbi.nlm.nih.gov/40153060/]
- 91. Targeting the orexin/hypocretin system for the treatment of ... [https://www.sciencedirect.com/science/article/pii/S0091302223000146]
- 92. The impact of sleep disturbances on adipocyte function and LIPID... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3031100/]
- 93. Melatonin Increases Brown Adipose Tissue Volume and ... [https://pubmed.ncbi.nlm.nih.gov/30765337/]
- 94. Significance and application of melatonin in the regulation ... [https://scholars.uthscsa.edu/en/publications/significance-and-application-of-melatonin-in-the-regulation-of-br/]
- 95. (PDF) Sleep Loss Disrupts Morning-to-Evening Differences in Human... [https://www.academia.edu/144888809/Sleep_Loss_Disrupts_Morning_to_Evening_Differences_in_Human_White_Adipose_Tissue_Transcriptome]
- 96. Chronic Sleep Alters Gut Microbiota, Induces Systemic and... Disruption [https://www.nature.com/articles/srep35405?error=cookies_not_supported&code=58adb77f-f871-4eff-a03b-15f14a660a86]
- 97. Suppression of obesity by melatonin through increasing ... [https://www.nature.com/articles/s41387-022-00222-2]
- 98. Elucidating the Regulatory Role of Melatonin in Brown, ... [https://www.sciencedirect.com/science/article/pii/S216183132200268X]
- 99. The Mechanisms of Magnesium in Sleep Disorders - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12535714/]
- 100. The Role of Magnesium in Depression, Migraine ... [https://www.mdpi.com/2072-6643/17/13/2216]
- 101. GABA and Magnesium: A Powerful Duo for Brain Health ... [https://troscriptions.com/blogs/main/gaba-magnesium?srsltid=AfmBOor7F9kRrDia4nbKAJIb10mPiPYm2_TWAJd6Xi6Vu5Z-nGIoPQNu]
- 102. Neuroprotective effects of magnesium: implications for ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2024.1406455/full]
- 103. Structural insights into the diverse actions of magnesium ... [https://pubmed.ncbi.nlm.nih.gov/40010346/]
- 104. Structural insights into the diverse actions of magnesium ... [https://www.sciencedirect.com/science/article/abs/pii/S0896627325000479]
- 105. Magnesium-L-threonate improves sleep quality and ... [https://www.sciencedirect.com/science/article/pii/S2590142724000193]
- 106. Association of magnesium intake with sleep duration and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8996025/]
- 107. Causal relations of trace elements and nutrients with insomnia [https://pmc.ncbi.nlm.nih.gov/articles/PMC12643735/]
- 108. Adenosine, caffeine, and sleep–wake regulation: state of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9541543/]
- 109. Adenosine and Sleep: Understanding Your Sleep Drive [https://www.sleepfoundation.org/how-sleep-works/adenosine-and-sleep]
- 110. Adenosine Receptors in Neuroinflammation and ... [https://www.mdpi.com/2073-4409/14/20/1585]
- 111. Sleep deprivation-induced cognitive impairment [https://www.sciencedirect.com/science/article/pii/S0014488625002833]
- 112. Neuroinflammation, Sleep, and Circadian Rhythms - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8981587/]
- 113. A2A receptor involves in Adenosine -mediated... neuroinflammation [https://pubmed.ncbi.nlm.nih.gov/29501623/]
- 114. Research hotspots and trends in the interaction mechanisms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12391283/]
- 115. impact of sleep disorders on biochemical parameters [https://jrbbr.com/article/view/360]
- 116. Changes in benzodiazepine, z-drug, and other sedative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12541714/]
- 117. Effects of Oral Gamma-Aminobutyric Acid (GABA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7527439/]
- 118. The role of the GABAergic system on insomnia - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11025592/]
- 119. Progress in Research on the Mechanism of GABA ... [https://www.mdpi.com/2304-8158/14/22/3856]
- 120. research into as-needed and intermittent benzodiazepines ... [https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2025.1569416/full]
- 121. Efficacy and Safety of Insomnia Pharmacotherapies [https://www.sciencedirect.com/science/article/pii/S1389945725005234]
- 122. Therapeutic Effects of Melatonin on Sleep and... Receptor Agonists [https://pmc.ncbi.nlm.nih.gov/articles/PMC4200764/]
- 123. Comparative Review of Approved Melatonin Agonists for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5108473/]
- 124. The Efficacy of Melatonergic Receptor Agonists Used in ... [https://pubmed.ncbi.nlm.nih.gov/41011705/]
- 125. The Efficacy of Melatonergic Receptor Agonists Used in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12472680/]
- 126. Melatonin Receptor Agonist - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/melatonin-receptor-agonist]
- 127. Exogenous Melatonin and Sleep Quality: A Scoping ... [https://pubmed.ncbi.nlm.nih.gov/41014554/]
- 128. The effect of exogenous melatonin and ... [https://pubmed.ncbi.nlm.nih.gov/40920753/]
- 129. Status of research on the application of melatonin ... [https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2025.1640198/full]
- 130. Found in translation: orexin receptor antagonism for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12552597/]
- 131. Overview of DORA Class Medications for the Treatment ... [https://www.neurologylive.com/view/overview-of-dora-class-medications-for-the-treatment-of-insomnia]
- 132. Orexin Receptor Antagonists for Insomnia in Patients with ... [https://www.mypcnow.org/fast-fact/orexin-receptor-antagonists-for-insomnia-in-patients-with-serious-illness/]
- 133. Current and novel dual orexin receptor antagonists for the ... [https://pubmed.ncbi.nlm.nih.gov/41273062/]
- 134. Dual orexin receptor antagonists in insomnia: Toward a ... [https://pubmed.ncbi.nlm.nih.gov/41138922/]
- 135. Efficacy and safety profiles of FDA-approved dual orexin ... [https://pubmed.ncbi.nlm.nih.gov/41109037/?utm_source=FeedFetcher&utm_medium=rss&utm_campaign=None&utm_content=10guX6I3SqrbUee4KRM8bS1RWRleWx6NPy1KJihLhedBd8bsna&fc=None&ff=20251101152122&v=2.18.0.post22+67771e2]
- 136. Idorsia | Media release [https://www.idorsia.com/investors/news-and-events/media-releases/media-release-details?id=3523547]
- 137. Adenosine, caffeine, and sleep-wake regulation: state of the science and perspectives - PubMed [https://pubmed.ncbi.nlm.nih.gov/35575450/]
- 138. Therapeutic potential of adenosine receptor modulators in ... [https://pubs.rsc.org/en/content/articlelanding/2025/ra/d5ra02235e]
- 139. Adenosine integrates light and sleep signalling for the ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8035342/]
- 140. A3-adenosine receptors: design of selective ligands and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4167349/]
- 141. Adenosine signalling drives antidepressant actions of ... [https://www.nature.com/articles/s41586-025-09755-9]
- 142. The Adenosine Pathway Clinical Trial Landscape Infographic [https://beacon-intelligence.com/infographic/adenosine-pathway-clinical-trial-landscape/]
- 143. A Novel Theanine Complex, Mg-L-Theanine Improves Sleep ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9017334/]
- 144. L-Theanine for Sleep [https://www.sleepfoundation.org/sleep-aids/l-theanine-for-sleep]
- 145. A Novel Theanine Complex, Mg-L-Theanine Improves ... [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2022.874254/full]
- 146. Why magnesium should be part of your sleep routine [https://sfihealth.com/news/why-magnesium-should-be-part-of-your-sleep-routine]
- 147. The effects of L-theanine consumption on sleep outcomes [https://www.sciencedirect.com/science/article/pii/S1087079225000292]
- 148. Association between magnesium deficiency score and ... [https://www.sciencedirect.com/science/article/abs/pii/S0165032724007183]
- 149. Theanine maintains sleep quality in healthy young women by ... [https://pubs.rsc.org/en/content/articlehtml/2023/fo/d3fo01247f]
- 150. Natural Sleep Aids: Which Are the Most Effective? [https://www.sleepfoundation.org/sleep-aids/natural-sleep-aids]